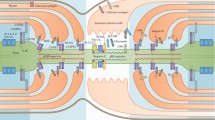
Abstract
Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) is considered to be an immune-mediated heterogeneous disease involving cellular and humoral immunity. In recent years, autoantibodies against nodal/paranodal protein neurofascin155 (NF155), neurofascin186 (NF186), contactin-1 (CNTN1), and contactin-associated protein 1 (CASPR1) have been identified in a small subset of patients with CIDP, which disrupt axo-glial interactions at nodes/paranodes. Although CIDP electrodiagnosis was made in patients with anti-nodal/paranodal component autoantibodies, macrophage-induced demyelination, the characteristic of typical CIDP, was not observed. Apart from specific histopathology, the pathogenic mechanisms and clinical manifestations of CIDP with autoantibody are also distinct. We herein compared pathogenesis, histopathology, clinical manifestations, and therapeutic response in CIDP with autoantibody vs. CIDP without autoantibody. CIDP with autoantibodies should be considered as an independent disease entity, not a subtype of CIDP due to many differences. They possibly should be classified as CIDP-like chronic nodo-paranodopathy, which can better characterize these disorders, help diagnose and make the most effective therapeutic decisions.


Similar content being viewed by others
References
Dyck PJ, Lais AC, Ohta M, Bastron JA, Okazaki H, Groover RV (1975) Chronic inflammatory polyradiculoneuropathy. Mayo Clin Proc 50(11):621–637
McCombe PA, Pollard JD, McLeod JG (1987) Chronic inflammatory demyelinating polyradiculoneuropathy. A clinical and electrophysiological study of 92 cases. Brain 110(Pt 6):1617–1630
Dyck P, Tracy JA (2018) History, diagnosis, and management of chronic inflammatory demyelinating polyradiculoneuropathy. Mayo Clin Proc 93(6):777–793
Koike H, Katsuno M, Sobue G (2018) Deciphering the mechanism and spectrum of chronic inflammatory demyelinating polyneuropathy using morphology. Clin Exp Neuroimmunol 9:35–46
Rodriguez Y, Vatti N, Ramirez-Santana C, Chang C, Mancera-Paez O, Gershwin ME, Anaya JM (2019) Chronic inflammatory demyelinating polyneuropathy as an autoimmune disease. J Autoimmun 102:8–37
Mathey EK, Park SB, Hughes RA, Pollard JD, Armati PJ, Barnett MH, Taylor BV, Dyck PJ, Kiernan MC, Lin CS (2015) Chronic inflammatory demyelinating polyradiculoneuropathy: from pathology to phenotype. J Neurol Neurosurg Psychiatry 86(9):973–985
Dyck PJ, O'Brien PC, Oviatt KF, Dinapoli RP, Daube JR, Bartleson JD, Mokri B, Swift T, Low PA, Windebank AJ (1982) Prednisone improves chronic inflammatory demyelinating polyradiculoneuropathy more than no treatment. Ann Neurol 11(2):136–141
Querol L, Devaux J, Rojas-Garcia R, Illa I (2017) Autoantibodies in chronic inflammatory neuropathies: diagnostic and therapeutic implications. Nat Rev Neurol 13(9):533–547
Koike H, Kadoya M, Kaida K, Ikeda S, Kawagashira Y, Iijima M, Kato D, Ogata H, Yamasaki R, Matsukawa N, Kira J, Katsuno M, Sobue G (2017) Paranodal dissection in chronic inflammatory demyelinating polyneuropathy with anti-neurofascin-155 and anti-contactin-1 antibodies. J Neurol Neurosurg Psychiatry 88(6):465–473
Doppler K, Appeltshauser L, Wilhelmi K, Villmann C, Dib-Hajj SD, Waxman SG, Mäurer M, Weishaupt A, Sommer C (2015) Destruction of paranodal architecture in inflammatory neuropathy with anti-contactin-1 autoantibodies. J Neurol Neurosurg Psychiatry 86(7):720–728
Doppler K, Appeltshauser L, Villmann C, Martin C, Peles E, Krämer HH, Haarmann A, Buttmann M, Sommer C (2016) Auto-antibodies to contactin-associated protein 1 (caspr) in two patients with painful inflammatory neuropathy. Brain 139(10):2617–2630
Kanda T (2013) Biology of the blood-nerve barrier and its alteration in immune mediated neuropathies. J Neurol Neurosurg Psychiatry 84(2):208–212
Mizisin AP, Weerasuriya A (2011) Homeostatic regulation of the endoneurial microenvironment during development, aging and in response to trauma, disease and toxic insult. Acta Neuropathol 121(3):291–312
Kanda T, Numata Y, Mizusawa H (2004) Chronic inflammatory demyelinating polyneuropathy: decreased claudin-5 and relocated ZO-1. J Neurol Neurosurg Psychiatry 75(5):765–769
Shimizu F, Sawai S, Sano Y, Beppu M, Misawa S, Nishihara H, Koga M, Kuwabara S, Kanda T (2014) Severity and patterns of blood-nerve barrier breakdown in patients with chronic inflammatory demyelinating polyradiculoneuropathy: correlations with clinical subtypes. PLoS ONE 9(8):e104205
Sommer C, Koch S, Lammens M, Gabreels-Festen A, Stoll G, Toyka KV (2005) Macrophage clustering as a diagnostic marker in sural nerve biopsies of patients with CIDP. Neurology 65(12):1924–1929
Diederich JM, Staudt M, Meisel C, Hahn K, Meinl E, Meisel A, Klehmet J (2018) Neurofascin and compact myelin antigen-specific T cell response pattern in chronic inflammatory demyelinating polyneuropathy subtypes. Front Neurol 9:171
Csurhes PA, Sullivan AA, Green K, Pender MP, McCombe PA (2005) T cell reactivity to P0, P2, PMP-22, and myelin basic protein in patients with Guillain-Barre syndrome and chronic inflammatory demyelinating polyradiculoneuropathy. J Neurol Neurosurg Psychiatry 76(10):1431–1439
Kieseier BC, Tani M, Mahad D, Oka N, Ho T, Woodroofe N, Griffin JW, Toyka KV, Ransohoff RM, Hartung HP (2002) Chemokines and chemokine receptors in inflammatory demyelinating neuropathies: a central role for IP-10. Brain 125(Pt 4):823–834
Shimizu F, Oishi M, Sawai S, Beppu M, Misawa S, Matsui N, Miyashiro A, Maeda T, Takeshita Y, Nishihara H, Sano Y, Sato R, Kaji R, Kuwabara S, Kanda T (2019) Increased IP-10 production by blood-nerve barrier in multifocal acquired demyelinating sensory and motor neuropathy and multifocal motor neuropathy. J Neurol Neurosurg Psychiatry 90(4):444–450
Moser B, Loetscher P (2001) Lymphocyte traffic control by chemokines. Nat Immunol 2(2):123–128
Mathey EK, Pollard JD, Armati PJ (1999) TNF alpha, IFN gamma and IL-2 mRNA expression in CIDP sural nerve biopsies. J Neurol Sci 163(1):47–52
Allard DE, Wang Y, Li JJ, Conley B, Xu EW, Sailer D, Kimpston C, Notini R, Smith CJ, Koseoglu E, Starmer J, Zeng XL, Howard JJ, Hoke A, Scherer SS, Su MA (2018) Schwann cell-derived periostin promotes autoimmune peripheral polyneuropathy via macrophage recruitment. J Clin Invest 128(10):4727–4741
Gillan L, Matei D, Fishman DA, Gerbin CS, Karlan BY, Chang DD (2002) Periostin secreted by epithelial ovarian carcinoma is a ligand for alpha(V)beta(3) and alpha(V)beta(5) integrins and promotes cell motility. Cancer Res 62(18):5358–5364
Johansson MW, Annis DS, Mosher DF (2013) Alpha(M)beta(2) integrin-mediated adhesion and motility of IL-5-stimulated eosinophils on periostin. Am J Respir Cell Mol Biol 48(4):503–510
Kiefer R, Kieseier BC, Stoll G, Hartung HP (2001) The role of macrophages in immune-mediated damage to the peripheral nervous system. Prog Neurobiol 64(2):109–127
Kiefer R, Dangond F, Mueller M, Toyka KV, Hafler DA, Hartung HP (2000) Enhanced b7 costimulatory molecule expression in inflammatory human sural nerve biopsies. J Neurol Neurosurg Psychiatry 69(3):362–368
Kuchroo VK, Das MP, Brown JA, Ranger AM, Zamvil SS, Sobel RA, Weiner HL, Nabavi N, Glimcher LH (1995) B7–1 and B7–2 costimulatory molecules activate differentially the Th1/Th2 developmental pathways: application to autoimmune disease therapy. Cell 80(5):707–718
Koike H, Nishi R, Ikeda S, Kawagashira Y, Iijima M, Katsuno M, Sobue G (2018) Ultrastructural mechanisms of macrophage-induced demyelination in CIDP. Neurology 91(23):1051–1060
Vallat JM, Mathis S, Vegezzi E, Richard L, Duchesne M, Gallouedec G, Corcia P, Magy L, Uncini A, Devaux J (2020) Antibody- and macrophage-mediated segmental demyelination in chronic inflammatory demyelinating polyneuropathy: clinical, electrophysiological, immunological and pathological correlates. Eur J Neurol 27(4):692–701
Vural A, Doppler K, Meinl E (2018) Autoantibodies against the node of ranvier in seropositive chronic inflammatory demyelinating polyneuropathy: diagnostic, pathogenic, and therapeutic relevance. Front Immunol 9:1029
Koga M (2018) autoantibodies in chronic inflammatory demyelinating polyneuropathy. Brain Nerve 70(4):395–403
Delmont E, Manso C, Querol L, Cortese A, Berardinelli A, Lozza A, Belghazi M, Malissart P, Labauge P, Taieb G, Yuki N, Illa I, Attarian S, Devaux JJ (2017) Autoantibodies to nodal isoforms of neurofascin in chronic inflammatory demyelinating polyneuropathy. Brain 140(7):1851–1858
Rasband MN, Peles E (2015) The nodes of ranvier: molecular assembly and maintenance. Cold Spring Harb Perspect Biol 8(3):a020495
Davis JQ, Lambert S, Bennett V (1996) Molecular composition of the node of Ranvier: identification of ankyrin-binding cell adhesion molecules neurofascin (mucin+/third FNIII domain−) and NrCAM at nodal axon segments. J Cell Biol 135(5):1355–1367
Susuki K, Chang KJ, Zollinger DR, Liu Y, Ogawa Y, Eshed-Eisenbach Y, Dours-Zimmermann MT, Oses-Prieto JA, Burlingame AL, Seidenbecher CI, Zimmermann DR, Oohashi T, Peles E, Rasband MN (2013) Three mechanisms assemble central nervous system nodes of Ranvier. Neuron 78(3):469–482
Stathopoulos P, Alexopoulos H, Dalakas MC (2015) Autoimmune antigenic targets at the node of Ranvier in demyelinating disorders. Nat Rev Neurol 11(3):143–156
Ho TS, Zollinger DR, Chang KJ, Xu M, Cooper EC, Stankewich MC, Bennett V, Rasband MN (2014) A hierarchy of ankyrin-spectrin complexes clusters sodium channels at nodes of Ranvier. Nat Neurosci 17(12):1664–1672
Thaxton C, Pillai AM, Pribisko AL, Dupree JL, Bhat MA (2011) Nodes of Ranvier act as barriers to restrict invasion of flanking paranodal domains in myelinated axons. Neuron 69(2):244–257
Desmazieres A, Zonta B, Zhang A, Wu LM, Sherman DL, Brophy PJ (2014) Differential stability of PNS and CNS nodal complexes when neuronal neurofascin is lost. J Neurosci 34(15):5083–5088
Sherman DL, Tait S, Melrose S, Johnson R, Zonta B, Court FA, Macklin WB, Meek S, Smith AJ, Cottrell DF, Brophy PJ (2005) Neurofascins are required to establish axonal domains for saltatory conduction. Neuron 48(5):737–742
Pillai AM, Thaxton C, Pribisko AL, Cheng JG, Dupree JL, Bhat MA (2009) Spatiotemporal ablation of myelinating glia-specific neurofascin (Nfasc NF155) in mice reveals gradual loss of paranodal axoglial junctions and concomitant disorganization of axonal domains. J Neurosci Res 87(8):1773–1793
Thaxton C, Pillai AM, Pribisko AL, Labasque M, Dupree JL, Faivre-Sarrailh C, Bhat MA (2010) In vivo deletion of immunoglobulin domains 5 and 6 in neurofascin (Nfasc) reveals domain-specific requirements in myelinated axons. J Neurosci 30(14):4868–4876
Martinez-Martinez L, Lleixa MC, Boera-Carnicero G, Cortese A, Devaux J, Siles A, Rajabally Y, Martinez-Pineiro A, Carvajal A, Pardo J, Delmont E, Attarian S, Diaz-Manera J, Callegari I, Marchioni E, Franciotta D, Benedetti L, Lauria G, de la Calle MO, Juarez C, Illa I, Querol L (2017) Anti-NF155 chronic inflammatory demyelinating polyradiculoneuropathy strongly associates to HLA-DRB15. J Neuroinflammation 14(1):224
Ogata H, Isobe N, Zhang X, Yamasaki R, Fujii T, Machida A, Morimoto N, Kaida K, Masuda T, Ando Y, Kuwahara M, Kusunoki S, Nakamura Y, Matsushita T, Kira JI (2019) Unique HLA haplotype associations in IgG4 anti-neurofascin 155 antibody-positive chronic inflammatory demyelinating polyneuropathy. J Neuroimmunol 339:577139
Kuwahara M, Suzuki H, Oka N, Ogata H, Yanagimoto S, Sadakane S, Fukumoto Y, Yamana M, Yuhara Y, Yoshikawa K, Morikawa M, Kawai S, Okazaki M, Tsujimoto T, Kira JI, Kusunoki S (2018) ELectron microscopic abnormality and therapeutic efficacy in chronic inflammatory demyelinating polyneuropathy with anti-neurofascin155 immunoglobulin G4 antibody. Muscle Nerve 57(3):498–502
Vallat JM, Yuki N, Sekiguchi K, Kokubun N, Oka N, Mathis S, Magy L, Sherman DL, Brophy PJ, Devaux JJ (2017) Paranodal lesions in chronic inflammatory demyelinating polyneuropathy associated with anti-neurofascin 155 antibodies. Neuromuscul Disord 27(3):290–293
Manso C, Querol L, Lleixa C, Poncelet M, Mekaouche M, Vallat JM, Illa I, Devaux JJ (2019) Anti-neurofascin-155 IgG4 antibodies prevent paranodal complex formation in vivo. J Clin Invest 129(6):2222–2236
Querol L, Nogales-Gadea G, Rojas-Garcia R, Diaz-Manera J, Pardo J, Ortega-Moreno A, Sedano MJ, Gallardo E, Berciano J, Blesa R, Dalmau J, Illa I (2014) Neurofascin IgG4 antibodies in CIDP associate with disabling tremor and poor response to IVIg. Neurology 82(10):879–886
Devaux JJ, Miura Y, Fukami Y, Inoue T, Manso C, Belghazi M, Sekiguchi K, Kokubun N, Ichikawa H, Wong AH, Yuki N (2016) Neurofascin-155 IgG4 in chronic inflammatory demyelinating polyneuropathy. Neurology 86(9):800–807
Klehmet J, Staudt M, Diederich JM, Siebert E, Meinl E, Harms L, Meisel A (2017) Neurofascin (NF)155- and NF186-Specific T cell response in a patient developing a central pontocerebellar demyelination after 10 years of CIDP. Front Neurol 8:724
Querol L, Nogales-Gadea G, Rojas-Garcia R, Martinez-Hernandez E, Diaz-Manera J, Suarez-Calvet X, Navas M, Araque J, Gallardo E, Illa I (2013) Antibodies to contactin-1 in chronic inflammatory demyelinating polyneuropathy. Ann Neurol 73(3):370–380
Manso C, Querol L, Mekaouche M, Illa I, Devaux JJ (2016) Contactin-1 IgG4 antibodies cause paranode dismantling and conduction defects. Brain 139(Pt 6):1700–1712
Labasque M, Hivert B, Nogales-Gadea G, Querol L, Illa I, Faivre-Sarrailh C (2014) Specific contactin N-glycans are implicated in neurofascin binding and autoimmune targeting in peripheral neuropathies. J Biol Chem 289(11):7907–7918
Cortese A, Lombardi R, Briani C, Callegari I, Benedetti L, Manganelli F, Luigetti M, Ferrari S, Clerici AM, Marfia GA, Rigamonti A, Carpo M, Fazio R, Corbo M, Mazzeo A, Giannini F, Cosentino G, Zardini E, Curro R, Gastaldi M, Vegezzi E, Alfonsi E, Berardinelli A, Kouton L, Manso C, Giannotta C, Doneddu P, Dacci P, Piccolo L, Ruiz M, Salvalaggio A, De Michelis C, Spina E, Topa A, Bisogni G, Romano A, Mariotto S, Mataluni G, Cerri F, Stancanelli C, Sabatelli M, Schenone A, Marchioni E, Lauria G, Nobile-Orazio E, Devaux J, Franciotta D (2020) Antibodies to neurofascin, contactin-1, and contactin-associated protein 1 in CIDP: Clinical relevance of IgG isotype. Neurol Neuroimmunol Neuroinflamm. https://doi.org/10.1212/NXI.0000000000000639
Fitzgerald M (2005) The development of nociceptive circuits. Nat Rev Neurosci 6(7):507–520
Ng JK, Malotka J, Kawakami N, Derfuss T, Khademi M, Olsson T, Linington C, Odaka M, Tackenberg B, Pruss H, Schwab JM, Harms L, Harms H, Sommer C, Rasband MN, Eshed-Eisenbach Y, Peles E, Hohlfeld R, Yuki N, Dornmair K, Meinl E (2012) Neurofascin as a target for autoantibodies in peripheral neuropathies. Neurology 79(23):2241–2248
Vallat JM, Mathis S, Magy L, Bounolleau P, Skarzynski M, Heitzmann A, Manso C, Devaux J, Uncini A (2018) Subacute nodopathy with conduction blocks and anti-neurofascin 140/186 antibodies: an ultrastructural study. Brain 141(7):e56
Yan W, Nguyen T, Yuki N, Ji Q, Yiannikas C, Pollard JD, Mathey EK (2014) Antibodies to neurofascin exacerbate adoptive transfer experimental autoimmune neuritis. J Neuroimmunol 277(1–2):13–17
Vital C, Vital A, Lagueny A, Ferrer X, Fontan D, Barat M, Gbikpi-Benissan G, Orgogozo JM, Henry P, Brechenmacher C, Bredin A, Desbordes P, Ribiere-Bachelier C, Latinville D, Julien J, Petry KG (2000) Chronic inflammatory demyelinating polyneuropathy: immunopathological and ultrastructural study of peripheral nerve biopsy in 42 cases. Ultrastruct Pathol 24(6):363–369
Listed NA (1991) Research criteria for diagnosis of chronic inlammatory demyelinating polyneuropathy (CIDP). Report from an Ad Hoc Subcommitteeof the American Academy of Neurology AIDS Task Force. Neurology 41(5):617–618. https://doi.org/10.1212/WNL.41.5.617
Van den Bergh PY, Hadden RD, Bouche P, Cornblath DR, Hahn A, Illa I, Koski CL, Leger JM, Nobile-Orazio E, Pollard J, Sommer C, van Doorn PA, van Schaik IN (2010) European Federation of Neurological Societies/Peripheral Nerve Society guideline on management of chronic inflammatory demyelinating polyradiculoneuropathy: report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society—first revision. Eur J Neurol 17(3):356–363
Ogata H, Yamasaki R, Hiwatashi A, Oka N, Kawamura N, Matsuse D, Kuwahara M, Suzuki H, Kusunoki S, Fujimoto Y, Ikezoe K, Kishida H, Tanaka F, Matsushita T, Murai H, Kira J (2015) Characterization of IgG4 anti-neurofascin 155 antibody-positive polyneuropathy. Ann Clin Transl Neurol 2(10):960–971
Boyle ME, Berglund EO, Murai KK, Weber L, Peles E, Ranscht B (2001) Contactin orchestrates assembly of the septate-like junctions at the paranode in myelinated peripheral nerve. Neuron 30(2):385–397
Sun XY, Takagishi Y, Okabe E, Chishima Y, Kanou Y, Murase S, Mizumura K, Inaba M, Komatsu Y, Hayashi Y, Peles E, Oda S, Murata Y (2009) A novel Caspr mutation causes the shambling mouse phenotype by disrupting axoglial interactions of myelinated nerves. J Neuropathol Exp Neurol 68(11):1207–1218
Miura Y, Devaux JJ, Fukami Y, Manso C, Belghazi M, Wong AH, Yuki N (2015) Contactin 1 IgG4 associates to chronic inflammatory demyelinating polyneuropathy with sensory ataxia. Brain 138(Pt 6):1484–1491
Fujita A, Ogata H, Yamasaki R, Matsushita T, Kira JI (2018) Parallel fluctuation of anti-neurofascin 155 antibody levels with clinico-electrophysiological findings in patients with chronic inflammatory demyelinating polyradiculoneuropathy. J Neurol Sci 384:107–112
Hashimoto Y, Ogata H, Yamasaki R, Sasaguri T, Ko S, Yamashita K, Xu Z, Matsushita T, Tateishi T, Akiyama S, Maruyama S, Yamamoto A, Kira JI (2018) Chronic inflammatory demyelinating polyneuropathy with concurrent membranous nephropathy: an anti-paranode and podocyte protein antibody study and literature survey. Front Neurol 9:997
Garg N, Park SB, Yiannikas C, Vucic S, Howells J, Noto YI, Mathey EK, Pollard JD, Kiernan MC (2018) Neurofascin-155 IGG4 neuropathy: pathophysiological insights, spectrum of clinical severity and response to treatment. Muscle Nerve 57(5):848–851
Bhat MA, Rios JC, Lu Y, Garcia-Fresco GP, Ching W, St MM, Li J, Einheber S, Chesler M, Rosenbluth J, Salzer JL, Bellen HJ (2001) Axon-glia interactions and the domain organization of myelinated axons requires neurexin IV/Caspr/Paranodin. Neuron 30(2):369–383
Kieseier BC, Mathey EK, Sommer C, Hartung HP (2018) Immune-mediated neuropathies. Nat Rev Dis Primers 4(1):31
Viala K, Maisonobe T, Stojkovic T, Koutlidis R, Ayrignac X, Musset L, Fournier E, Leger JM, Bouche P (2010) A current view of the diagnosis, clinical variants, response to treatment and prognosis of chronic inflammatory demyelinating polyradiculoneuropathy. J Peripher Nerv Syst 15(1):50–56
Illes Z, Blaabjerg M (2017) Cerebrospinal fluid findings in Guillain–Barre syndrome and chronic inflammatory demyelinating polyneuropathies. Handb Clin Neurol 146:125–138
Shibuya K, Sugiyama A, Ito S, Misawa S, Sekiguchi Y, Mitsuma S, Iwai Y, Watanabe K, Shimada H, Kawaguchi H, Suhara T, Yokota H, Matsumoto H, Kuwabara S (2015) Reconstruction magnetic resonance neurography in chronic inflammatory demyelinating polyneuropathy. Ann Neurol 77(2):333–337
Lewis RA (2017) Chronic inflammatory demyelinating polyneuropathy. Curr Opin Neurol 30(5):508–512
Saperstein DS, Amato AA, Wolfe GI, Katz JS, Nations SP, Jackson CE, Bryan WW, Burns DK, Barohn RJ (1999) Multifocal acquired demyelinating sensory and motor neuropathy: the Lewis–Sumner syndrome. Muscle Nerve 22(5):560–566
Lewis RA, Sumner AJ, Brown MJ, Asbury AK (1982) Multifocal demyelinating neuropathy with persistent conduction block. Neurology 32(9):958–964
Nobile-Orazio E (2014) Chronic inflammatory demyelinating polyradiculoneuropathy and variants: where we are and where we should go. J Peripher Nerv Syst 19(1):2–13
Katz JS, Saperstein DS, Gronseth G, Amato AA, Barohn RJ (2000) Distal acquired demyelinating symmetric neuropathy. Neurology 54(3):615–620
Ikeda S, Koike H, Nishi R, Kawagashira Y, Iijima M, Katsuno M, Sobue G (2019) Clinicopathological characteristics of subtypes of chronic inflammatory demyelinating polyradiculoneuropathy. J Neurol Neurosurg Psychiatry 90(9):988–996
Puwanant A, Herrmann DN (2012) Multifocal acquired demyelinating sensory and motor neuropathy. Neurology 79(16):1742
Franques J, Chapon F, Devaux J, Mathis S (2017) Teaching NeuroImages: Cranial nerve hypertrophy in IgG4 anti-neurofascin 155 antibody-positive polyneuropathy. Neurology 88(7):e52
Painous C, Lopez-Perez MA, Illa I, Querol L (2018) Head and voice tremor improving with immunotherapy in an anti-NF155 positive CIDP patient. Ann Clin Transl Neurol 5(4):499–501
Cortese A, Devaux JJ, Zardini E, Manso C, Taieb G, Carra DC, Merle P, Osera C, Romagnolo S, Visigalli N, Piscosquito G, Salsano E, Alfonsi E, Moglia A, Pareyson D, Marchioni E, Franciotta D (2016) Neurofascin-155 as a putative antigen in combined central and peripheral demyelination. Neurol Neuroimmunol Neuroinflamm 3(4):e238
Burnor E, Yang L, Zhou H, Patterson KR, Quinn C, Reilly MM, Rossor AM, Scherer SS, Lancaster E (2018) Neurofascin antibodies in autoimmune, genetic, and idiopathic neuropathies. Neurology 90(1):e31–e38
Taieb G, Le Quintrec M, Pialot A, Szwarc I, Perrochia H, Labauge P, Devaux JJ (2019) "neuro-renal syndrome" related to anti-contactin-1 antibodies. Muscle Nerve 59(3):E19–E21
Reid RA, Bronson DD, Young KM, Hemperly JJ (1994) Identification and characterization of the human cell adhesion molecule contactin. Brain Res Mol Brain Res 21(1–2):1–8
Hu W, Xin Y, He Z, Zhao Y (2018) Association of neurofascin IgG4 and atypical chronic inflammatory demyelinating polyneuropathy: A systematic review and meta-analysis. Brain Behav 8(10):e01115
Bunschoten C, Jacobs BC, Van den Bergh P, Cornblath DR, van Doorn PA (2019) Progress in diagnosis and treatment of chronic inflammatory demyelinating polyradiculoneuropathy. Lancet Neurol 18(8):784–794
Roggenbuck JJ, Boucraut J, Delmont E, Conrad K, Roggenbuck D (2018) Diagnostic insights into chronic-inflammatory demyelinating polyneuropathies. Ann Transl Med 6(17):337
Hughes RA, Mehndiratta MM, Rajabally YA (2017) Corticosteroids for chronic inflammatory demyelinating polyradiculoneuropathy. Cochrane Database Syst Rev 11:CD002062
Eftimov F, Winer JB, Vermeulen M, de Haan R, van Schaik IN (2013) Intravenous immunoglobulin for chronic inflammatory demyelinating polyradiculoneuropathy. Cochrane Database Syst Rev 12:CD001797
Mehndiratta MM, Hughes RA, Pritchard J (2015) Plasma exchange for chronic inflammatory demyelinating polyradiculoneuropathy. Cochrane Database Syst Rev 8:CD003906
van Schaik IN, Bril V, van Geloven N, Hartung HP, Lewis RA, Sobue G, Lawo JP, Praus M, Mielke O, Durn BL, Cornblath DR, Merkies I (2018) Subcutaneous immunoglobulin for maintenance treatment in chronic inflammatory demyelinating polyneuropathy (PATH): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Neurol 17(1):35–46
Mahdi-Rogers M, Brassington R, Gunn AA, van Doorn PA, Hughes RA (2017) Immunomodulatory treatment other than corticosteroids, immunoglobulin and plasma exchange for chronic inflammatory demyelinating polyradiculoneuropathy. Cochrane Database Syst Rev 5:CD003280
Hughes R, Dalakas MC, Merkies I, Latov N, Leger JM, Nobile-Orazio E, Sobue G, Genge A, Cornblath D, Merschhemke M, Ervin CM, Agoropoulou C, Hartung HP (2018) Oral fingolimod for chronic inflammatory demyelinating polyradiculoneuropathy (FORCIDP Trial): a double-blind, multicentre, randomised controlled trial. Lancet Neurol 17(8):689–698
Kadoya M, Kaida K, Koike H, Takazaki H, Ogata H, Moriguchi K, Shimizu J, Nagata E, Takizawa S, Chiba A, Yamasaki R, Kira JI, Sobue G, Ikewaki K (2016) IgG4 anti-neurofascin155 antibodies in chronic inflammatory demyelinating polyradiculoneuropathy: clinical significance and diagnostic utility of a conventional assay. J Neuroimmunol 301:16–22
Querol L, Rojas-Garcia R, Diaz-Manera J, Barcena J, Pardo J, Ortega-Moreno A, Sedano MJ, Sero-Ballesteros L, Carvajal A, Ortiz N, Gallardo E, Illa I (2015) Rituximab in treatment-resistant CIDP with antibodies against paranodal proteins. Neurol Neuroimmunol Neuroinflamm 2(5):e149
Briani C, Salvalaggio A, Ruiz M, Cacciavillani M, Rinaldi F, Callegari I, Gasparotti R, Franciotta D (2019) Tongue tremor in neurofascin-155 IgG4 seropositive chronic inflammatory polyradiculoneuropathy. J Neuroimmunol 330:178–180
Benedetti L, Briani C, Franciotta D, Fazio R, Paolasso I, Comi C, Luigetti M, Sabatelli M, Giannini F, Mancardi GL, Schenone A, Nobile-Orazio E, Cocito D (2011) Rituximab in patients with chronic inflammatory demyelinating polyradiculoneuropathy: a report of 13 cases and review of the literature. J Neurol Neurosurg Psychiatry 82(3):306–308
Yuki N, Watanabe H, Nakajima T, Spath PJ (2011) IVIG blocks complement deposition mediated by anti-GM1 antibodies in multifocal motor neuropathy. J Neurol Neurosurg Psychiatry 82(1):87–91
Richard A, Corvol JC, Debs R, Reach P, Tahiri K, Carpentier W, Gueguen J, Guillemot V, Labeyrie C, Adams D, Viala K, Cohen AF (2016) Transcriptome analysis of peripheral blood in chronic inflammatory demyelinating polyradiculoneuropathy patients identifies TNFR1 and TLR pathways in the IVIg response. Medicine (Baltimore) 95(19):e3370
Xu Y, Oomen R, Klein MH (1994) Residue at position 331 in the IgG1 and IgG4 CH2 domains contributes to their differential ability to bind and activate complement. J Biol Chem 269(5):3469–3474
Salles G, Barrett M, Foa R, Maurer J, O'Brien S, Valente N, Wenger M, Maloney DG (2017) Rituximab in B-cell hematologic malignancies: a review of 20 years of clinical experience. Adv Ther 34(10):2232–2273
Scheibe F, Alexander T, Pruss H, Wengert O, Harms L, Angstwurm K, Hiepe F, Arnold R, Meisel A (2016) Devastating humoral CIDP variant remitted by autologous stem cell transplantation. Eur J Neurol 23(3):e12–e14
Dorst J, Ludolph AC, Senel M, Tumani H (2018) Short-term and long-term effects of immunoadsorption in refractory chronic inflammatory demyelinating polyneuropathy: a prospective study in 17 patients. J Neurol 265(12):2906–2915
Uncini A, Vallat JM (2018) Autoimmune nodo-paranodopathies of peripheral nerve: the concept is gaining ground. J Neurol Neurosurg Psychiatry 89(6):627–635
Uncini A, Kuwabara S (2015) Nodopathies of the peripheral nerve: an emerging concept. J Neurol Neurosurg Psychiatry 86(11):1186–1195
Uncini A, Susuki K, Yuki N (2013) Nodo-paranodopathy: beyond the demyelinating and axonal classification in anti-ganglioside antibody-mediated neuropathies. Clin Neurophysiol 124(10):1928–1934
Zhang X, Zheng P, Devaux JJ, Wang Y, Liu C, Li J, Guo S, Song Y, Wang Q, Feng X, Wang Y (2019) Chronic inflammatory demyelinating polyneuropathy with anti-NF155 IgG4 in China. J Neuroimmunol 337:577074
Acknowledgements
The authors would like to thank Yi Shan for his dedication to the figure for this paper.
Author information
Authors and Affiliations
Contributions
L.T. developed the concept and design of the review, and drafted the manuscript for intellectual content. Q.H. and Z.Q. revised the manuscript for intellectual content. X.T. supervised the work and contributed to the review revision.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
The manuscript does not contain clinical studies or patient data.
Rights and permissions
About this article
Cite this article
Tang, L., Huang, Q., Qin, Z. et al. Distinguish CIDP with autoantibody from that without autoantibody: pathogenesis, histopathology, and clinical features. J Neurol 268, 2757–2768 (2021). https://doi.org/10.1007/s00415-020-09823-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-020-09823-2