Abstract
Background
One of the most characteristic changes in Alzheimer’s disease (AD) is a deficit in cortical cholinergic neurotransmission and associated receptor changes.
Objective
To investigate differences in the distribution of M1/M4 receptors using (R, R) 123I-iodo-quinuclidinyl-benzilate (QNB) and single photon emission computed tomography (SPECT) in patients with mild/moderate AD and agematched controls. Also, to compare 123I-QNB uptake to the corresponding changes in regional cerebral blood flow (rCBF) in the same subjects.
Methods
Forty two subjects (18 AD and 24 healthy elderly controls) underwent 123IQNB and perfusion 99mTc-exametazime SPECT scanning. Image analysis was performed using statistical parametric mapping (SPM99) following intensity normalisation of each image to its corresponding mean whole brain uptake. Group differences and correlations were assessed using two sample t-tests and linear regression respectively.
Results
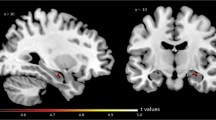
Significant reductions in 123I-QNB uptake were observed in regions of the frontal rectal gyrus, right parahippocampal gyrus, left hippocampus and areas of the left temporal lobe in AD compared to controls (height threshold of p ≤ 0.001 uncorrected). Such regions were also associated with marked deficits in rCBF. No significant correlations were identified between imaging data and clinical variables.
Conclusion
Functional impairment as measured by rCBF is more widespread than changes in M1/M4 receptor density in mild/moderate AD, where there was little or no selective loss of M1/M4 receptors in these patients that was greater than the general functional deficits shown on rCBF scans.
Similar content being viewed by others
References
Araujo DM, Lapchak PA, Robitaille Y, Gauthier S, Quirion R (1988) Differential alteration of various cholinergic markers in cortical and subcortical regions of human brain in Alzheimer’s disease. J Neurochem 50:1914–1923
Bierer LM, Haroutunian V, Gabriel S, Knott PJ, Carlin LS, Purohit DP, Perl DP, Schmeidler J, Kanof P, Davis KL (1995) Neurochemical correlates of dementia severity in Alzheimer’s disease: relative importance of the cholinergic deficits. J Neurochem 64:749–760
Davies P, Maloney AJ (1976) Selective loss of central cholinergic neurons in Alzheimer’s disease. Lancet 2:1403
Samuel W, Terry RD, DeTeresa R, Butters N, Masliah E (1994) Clinical correlates of cortical and nucleus basalis pathology in Alzheimer dementia. Arch Neurol 51:772–778
White P, Hiley CR, Goodhardt MJ, Carrasco LH, Keet JP, Williams IE, Bowen DM (1977) Neocortical cholinergic neurons in elderly people. Lancet 1:668–671
Geula C, Mesulam MM (1996) Systematic regional variations in the loss of cortical cholinergic fibers in Alzheimer’s disease. Cereb Cortex 6:165–177
Perry E, Court J, Goodchild R, Griffiths M, Jaros E, Johnson M, Lloyd S, Piggott M, Spurden D, Ballard C, McKeith I, Perry R (1998) Clinical neurochemistry: developments in dementia research based on brain bank material. J Neural Transm 105:915–933
Geula C, Mesulam MM, Saroff DM, Wu CK (1998) Relationship between plaques, tangles, and loss of cortical cholinergic fibers in Alzheimer disease. J Neuropathol Exp Neurol 57:63–65
Perry EK, Tomlinson BE, Blessed G, Bergmann K, Gibson PH, Perry RH (1978) Correlation of cholinergic abnormalities with senile plaques and mental test scores in senile dementia. Br Med J 2:1457–1459
Rinne JO, Laakso K, Lonnberg P, Molsa P, Paljarvi L, Rinne JK, Sako E, Rinne UK (1985) Brain muscarinic receptors in senile dementia. Brain Res 336:19–25
Shimohama S, Taniguchi T, Fujiwara M, Kameyama M (1986) Changes in nicotinic and muscarinic cholinergic receptors in Alzheimer-type dementia. J Neurochem 46:288–293
Shiozaki K, Iseki E, Uchiyama H, Watanabe Y, Haga T, Kameyama K, Ikeda T, Yamamoto T, Kosaka K (1999) Alterations of muscarinic acetylcholine receptor subtypes in diffuse lewy body disease: relation to Alzheimer’s disease [see comments]. J Neurol Neurosurg Psychiatry 67:209–213
Nordberg A, Winblad B (1986) Reduced number of [3H]nicotine and [3H]acetylcholine binding sites in the frontal cortex of Alzheimer brains. Neurosci Lett 72:115–119
Aubert I, Araujo DM, Cecyre D, Robitaille Y, Gauthier S, Quirion R (1992) Comparative alterations of nicotinic and muscarinic binding sites in Alzheimer’s and Parkinson’s diseases. J Neurochem 58:529–541
Piggott MA, Owens J, O’Brien J, Colloby S, Fenwick J, Wyper D, Jaros E, Johnson M, Perry RH, Perry EK (2003) Muscarinic receptors in basal ganglia in dementia with Lewy bodies, Parkinson’s disease and Alzheimer’s disease. J Chem Neuroanat 25:161–173
Piggott M, Owens J, O’Brien J, Paling S, Wyper D, Fenwick J, Johnson M, Perry R, Perry E (2002) Comparative distribution of binding of the muscarinic receptor ligands pirenzepine, AF-DX 384, (R,R)-I-QNB and (R,S)-I-QNB to human brain. J Chem Neuroanat 24:211–223
Folstein MF, Folstein SE, McHugh PR (1975) "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician. J Psychiatry Res 12:189–198
Roth M, Tym E, Mountjoy CQ, Huppert FA, Hendrie H, Verma S, Goddard R (1986) CAMDEX A Standardised Instrument for the Diagnosis of Mental Disorder in the Elderly with Special Reference to the Early Detection of Dementia. Br J Psychiatry 149:698–709
Cummings JL, Mega M, Gray K, Rosenberg-Thompson S, Carusi DA, Gornbein J (1994) The Neuropsychiatric Inventory: comprehensive assessment of psychopathology in dementia. Neurology 44:2308–2314
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM (1984) Clinical diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34:939–944
Lee KS, He XS, Jones DW, Coppola R, Gorey JG, Knable MB, deCosta BR, Rice KC, Weinberger DR (1996) An improved method for rapid and efficient radioiodination of iodine-123-IQNB. J Nucl Med 37:2021–2024
Brown D, Chisholm JA, Owens J, Pimlott S, Patterson J, Wyper D (2003) Acetylcholine muscarinic receptors and response to anti-cholinesterase therapy in patients with Alzheimer’s disease. Eur J Nucl Med Mol Imag 30:296–200
Talairach J, Tournoux P (1988) Coplanar stereotactic atlas of the human brain. Thieme, Stuttgart New York
Acton PD, Friston KJ (1998) Statistical parametric mapping in functional neuroimaging: beyond PET and fMRI activation studies. Eur J Nucl Med 25:663–667
Chatfield C, Collins AJ (1980) Introduction to multivariate analysis.189–210
Frackowiak RS, Friston KJ, Frith CD, Dolan RJ, Mazziotta JC (1997) Human brain function. In: Academic Press, p 67–70
Friston KJ, Worsley KJ, Frackowiak RSJ, Mazziotta JC, Evans AC (1994) Assessing the significance of focal activations using their spatial extent. Hum Brain Map 1:214–220
Brett M (1999) The MNI brain and the Talairach Atlas. In:MRC Cognition and Brain Sciences Unit. http://www.mrccbu. cam.ac.uk/Imaging/contents.html
Bartenstein P, Minoshima S, Hirsch C, Buch K, Willoch F, Mosch D, Schad D, Schwaiger M, Kurz A (1997) Quantitative assessment of cerebral blood flow in patients with Alzheimer’s disease by SPECT. J Nucl Med 38:1095–1101
Grossman M, Payer F, Onishi K, D’Esposito M, Morrison D, Sadek A, Alavi A (1998) Language comprehension and regional cerebral defects in frontotemporal degeneration and Alzheimer’s disease. Neurology 50:157–163
Osimani A, Ichise M, Chung DG, Pogue JM, Freedman M (1994) SPECT for differential diagnosis of dementia and correlation of rCBF with cognitive impairment. Can J Neurol Sci 21:104–111
Holman BL, Gibson RE, Hill TC, Eckelman WC, Albert M, Reba RC (1985) Muscarinic acetylcholine receptors in Alzheimer’s disease. In vivo imaging with iodine 123-labeled 3-quinuclidinyl- 4-iodobenzilate and emission tomography. Jama 254:3063–3066
Weinberger DR, Gibson R, Coppola R, Jones DW, Molchan S, Sunderland T, Berman KF, Reba RC (1991) The distribution of cerebral muscarinic acetylcholine receptors in vivo in patients with dementia. A controlled study with 123IQNB and single photon emission computed tomography. Arch Neurol 48:169–176
Kemp PM, Holmes C, Hoffmann S, Wilkinson S, Zivanovic M, Thom J, Bolt L, Fleming J, Wilkinson DG (2003) A randomised placebo controlled study to assess the effects of cholinergic treatment on muscarinic receptors in Alzheimer’s disease. J Neurol Neurosurg Psychiatry 74:1567–1570
Wyper DJ, Brown D, Patterson J, Owens J, Hunter R, Teasdale E, McCulloch J (1993) Deficits in iodinelabelled 3-quinuclidinyl benzilate binding in relation to cerebral blood flow in patients with Alzheimer’s disease. Eur J Nucl Med 20:379–386
Claus JJ, Dubois EA, Booij J, Habraken J, de Munck JC, van Herk M, Verbeeten B Jr., van Royen EA (1997) Demonstration of a reduction in muscarinic receptor binding in early Alzheimer’s disease using iodine-123 dexetimide single-photon emission tomography. Eur J Nucl Med 24:602–608
Boundy KL, Barnden LR, Katsifis AG, Rowe CC (2005) Reduced posterior cingulate binding of I-123 iodo-dexetimide to muscarinic receptors in mild Alzheimer’s disease. J Clin Neurosci 12:421–425
Yoshida T, Kuwabara Y, Ichiya Y, Sasaki M, Fukumura T, Ichimiya A, Takita M, Ogomori K, Masuda K (1998) Cerebral muscarinic acetylcholinergic receptor measurement in Alzheimer’s disease patients on 11C-N-methyl-4- piperidyl benzilate–comparison with cerebral blood flow and cerebral glucose metabolism. Ann Nucl Med 12:35–42
Zubieta JK, Koeppe RA, Frey KA, Kilbourn MR, Mangner TJ, Foster NL, Kuhl DE (2001) Assessment of muscarinic receptor concentrations in aging and Alzheimer disease with [11C]NMPB and PET. Synapse 39:275–287
Ogawa M, Iida Y, Nakagawa M, Kuge Y, Kawashima H, Tominaga A, Ueda M, Magata Y, Saji H (2006) Change of central cholinergic receptors following lesions of nucleus basalis magnocellularis in rats: search for an imaging index suitable for the early detection of Alzheimer’s disease. Nucl Med Biol 33:249–254
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pakrasi, S., Colloby, S.J., Firbank, M.J. et al. Muscarinic acetylcholine receptor status in Alzheimer’s disease assessed using (R, R) 123I-QNB SPECT. J Neurol 254, 907–913 (2007). https://doi.org/10.1007/s00415-006-0473-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-006-0473-8