Abstract
The zebra finch (Taeniopygia guttata) has a large Z chromosome and highly condensed W chromosome. We used the random amplified polymorphic DNA (RAPD) polymerase chain reaction (PCR) technique to isolate female-specific sequences ZBM1 and ZBM2. Southern blot hybridization to male and female zebra finch genomic DNA suggested that these sequences were located on the W chromosome, although homologous sequences appeared to be autosomal or Z-linked. Fluorescent in situ hybridization (FISH) using bacterial artificial chromosome (BAC) clones corresponding to ZBM sequences showed hybridization to the whole W chromosome, suggesting that the BACs encode sequences that are repeated across the entire W chromosome. Based on the sequencing of a ZBM repetitive sequence and Z chromosome derived BAC clones, we demonstrate a random distribution of repeat sequences that are specific to the W chromosome or encoded by both Z and W. The positions of ZW-common repeat sequences mapped to a noncoding region of a Z chromosome BAC clone containing the CHD1Z gene. The apparent lineage-specificity of W chromosome repeat sequences in passerines and galliform birds suggest that the W chromosome had not differentiated well from the Z at the time of divergence of these lineages.









Similar content being viewed by others
References
Agate RJ, Perlman WR, Arnold AP (2002) Cloning and expression of zebra finch (Taeniopygia guttata) steroidogenic factor 1: overlap with hypothalamic but not with telencephalic aromatase. Biol Reprod 66(4):1127–1133
Arnold AP (2002) Concepts of genetic and hormonal induction of vertebrate sexual differentiation in the twentieth century, with special reference to the brain. In Arnold AP, Etgen A, Fahrbach S, Rubin P, Pfaff DW (eds). San Diego: Academic Press, pp 105–135
Bachtrog D (2006) A dynamic view of sex chromosome evolution. Curr Opin Genet Dev 16(6):578–585
Charlesworth B (1991) The evolution of sex chromosomes. Science 251(4997):1030–1033
Charlesworth D, Charlesworth B, Marais G (2005) Steps in the evolution of heteromorphic sex chromosomes. Heredity 95(2):118–128
Ellegren H (1996) First gene on the avian W chromosome (CHD) provides a tag for universal sexing of non-ratite birds. Proc Biol Sci 263(1377):1635–1641
Garcia-Moreno J, Mindell DP (2000) Rooting a phylogeny with homologous genes on opposite sex chromosomes (gametologs): a case study using avian CHD. Mol Biol Evol 17(12):1826–1832
Graves JA (2003) Sex and death in birds: a model of dosage compensation that predicts lethality of sex chromosome aneuploids. Cytogenet Genome Res 101(3–4):278–282
Graves JA (2006) Sex chromosome specialization and degeneration in mammals. Cell 124(5):901–914
Griffiths R, Double MC, Orr K, Dawson RJ (1998) A DNA test to sex most birds. Mol Ecol 7(8):1071–1075
Handley LJ, Ceplitis H, Ellegren H (2004) Evolutionary strata on the chicken Z chromosome: implication for sex chromosome evolution. Genetics 167:367–376
Hori T, Asakawa S, Itoh Y, Shimizu N, Mizuno S (2000) Wpkci, encoding an altered form of PKCI, is conserved widely on the avian W chromosome and expressed in early female embryos: implication of its role in female sex determination. Mol Biol Cell 11(10):3645–3660
Itoh Y, Mizuno S (2002) Molecular and cytological characterization of SspI-family repetitive sequence on chicken W chromosome. Chromosom Res 10:499–511
Itoh Y, Arnold AP (2005) Chromosomal polymorphism and comparative painting analysis in the zebra finch. Chromosom Res 13:47–56
Itoh Y, Hori T, Saitoh H, Mizuno S (2001a) Chicken spindlin genes on W and Z chromosomes: transcriptional expression of both genes and dynamic behavior of spindlin in interphase and mitotic cells. Chromosom Res 9:283–299
Itoh Y, Suzuki M, Ogawa A, Munechika I, Murata K, Mizuno S (2001b) Identification of the sex of a wide range of Carinatae birds by PCR using primer sets selected from chicken EE0.6 and its related sequences. J Heredity 92:315–321
Itoh Y, Kampf K, Arnold AP (2006) Comparison of the chicken and zebra finch Z chromosomes shows evolutionary rearrangements. Chromosom Res 14(8):805–815
Itoh Y, Melamed E, Yang X, Kampf K, Wang S, Yehya N, Van Nas A, Replogle K, Band MR, Clayton DF, Schadt EE, Lusis AJ, Arnold AP (2007) Dosage compensation is less effective in birds than in mammals. J Biol. 6(1):2
Kodama H, Saitoh H, Tone M, Kuhara S, Sakaki Y, Mizuno S (1987) Nucleotide sequences and unusual electrophoretic behavior of the W chromosome-specific repeating DNA units of the domestic fowl, Gallus gallus domesticus. Chromosoma 96(1):18–25
Konishi M (1994) An outline of recent advances in birdsong neurobiology. Brain Behav Evol 44:279–285
Luo M, Yu Y, Kim H, Kudrna D, Itoh Y, Agate RJ, Melamed E, Goicoechea JL, Talag J, Mueller C, Wang W, Currie J, Sisneros NB, Wing RA, Arnold AP (2006) Utilization of a zebra finch BAC library to determine the structure of an avian androgen receptor genomic region. Genomics 87(1):181–190
Mank JE, Ellegren H (2007) Parallel divergence and degradation of the avian W sex chromosome. Trends Ecol Evol 22(8):389–391
Marler P (1990) Song learning: the interface between behaviour and neuroethology. Philos Trans R Soc Lond B Biol Sci 329:109–114
Matthews ME, Reed KC (1991) A DNA sequence that is present in both sexes of Artiodactyla is repeated on the Y chromosome of cattle, sheep, and goats. Cytogenet Cell Genet 56(1):40–44
Nanda I, Shan Z, Schart M, Burt DW, Koehler M, Nothwang H, Grutzner F, Paton IR, Windsor D, Dunn I, Engel W, Staeheli P, Mizuno S, Haaf T, Schmid M (1999) 300 million years of conserved synteny between chicken Z and human chromosome 9. Nature Genet 21:258–259
Nanda I, Zend-Ajusch E, Shan Z, Grutzner F, Schart M, Burt DW, Koehler M, Fowler VM, Goodwin G, Schneider WJ, Mizuno S, Dechant G, Haaf T, Schmid M (2000) Conserved syntheny between the chicken Z sex chromosome and human chromosome 9 includes the male regulatory gene DMRT1: a comparative (re)view on avian sex determination. Cytogenet Cell Genet 89:67–78
Nottebohm F (2002) Neuronal replacement in adult brain. Brain Res Bull 57:737–749
Ogawa A, Solovei I, Hutchison N, Saitoh Y, Ikeda JE, Macgregor H, Mizuno S (1997) Molecular characterization and cytological mapping of a non-repetitive DNA sequence region from the W chromosome of chicken and its use as a universal probe for sexing Carinatae birds. Chromosom Res 5:93–101
Ogawa A, Murata K, Mizuno S (1998) The location of Z- and W-linked marker genes and sequence on the homomorphic sex chromosomes of the ostrich and the emu. Proc Natl Acad Sci USA 95(8):4415–4418
O’Neill M, Binder M, Smith C, Andrews J, Reed K, Smith M, Millar C, Lambert D, Sinclair A (2000) ASW: a gene with conserved W-linkage and female specific expression in chick embryonic gonad. Dev Genes Evol 210(5):243–249
Pigozzi MI, Solari AJ (1998) Germ cell restriction and regular transmission of an accessory chromosome that mimics a sex body in the zebra finch, Taeniopygia guttata. Chromosom Res 6:105–113
Raymond CS, Kettlewell JR, Hirsch B, Bardwell VJ, Zarkower D (1999) Expression of Dmrt1 in the genital ridge of mouse and chicken embryos suggests a role in vertebrate sexual development. Dev Biol 215(2):208–220
Raymond CS, Murphy MW, O'Sullivan MG, Bardwell VJ, Zarkower D (2000) Dmrt1, a gene related to worm and fly sexual regulators, is required for mammalian testis differentiation. Genes Dev 14(20):2587–2595
Reed KJ, Sinclair AH (2002) FET-1: a novel W-limked, female specific gene up-regulated in the embryonic chicken ovary. Mech Dev 119:S87–S90
Rozen S, Skaletsky H, Marszalek JD, Minx PJ, Cordum HS, Waterston RH, Wilson RK, Page DC (2003) Abundant gene conversion between arms of palindromes in human and ape Y chromosomes. Nature 423(6942):873–876
Runciman D, Zann RA, Murray ND (1999) A W-chromosome linked marker for gender identification in the zebra finch. Anim Genet 30(1):66–67
Saitoh Y, Mizuno S (1992) Distribution of XhoI and EcoRI family repetitive DNA sequences into separate domains in the chicken W chromosome. Chromosoma 101:474–477
Schmid M, Nanda I, Hoehn H, Schartl M, Haaf T, Buerstedde JM, Arakawa H, Caldwell RB, Weigend S, Burt DW, Smith J, Griffin DK, Masabanda JS, Groenen MA, Crooijmans RP, Vignal A, Fillon V, Morisson M, Pitel F, Vignoles M, Garrigues A, Gellin J, Rodionov AV, Galkina SA, Lukina NA, Ben-Ari G, Blum S, Hillel J, Twito T, Lavi U, David L, Feldman MW, Delany ME, Conley CA, Fowler VM, Hedges SB, Godbout R, Katyal S, Smith C, Hudson Q, Sinclair A, Mizuno S (2005) Second report on chicken genes and chromosomes 2005. Cytogenet Genome Res 109(4):415–479
Schmidtmer J, Engelkamp D (2004) Isolation and expression pattern of three mouse homologues of chick Rgm. Gene Expr Patterns 4(1):105–110
Shetty S, Griffin DK, Graves JA (1999) Comparative painting reveals strong chromosome homology over 80 million years of bird evolution. Chromosom Res 7:289–295
Shetty S, Kirby P, Zarkower D, Graves JA (2002) DMRT1 in a ratite bird: evidence for a role in sex determination and discovery of a putative regulatory element. Cytogenet Genome Res 99(1–4):245–251
Soderstrom K, Qin W, Leggett MH (2007) A minimally invasive procedure for sexing young zebra finches. J Neurosci Methods 164(1):116–119
Steinemann S, Steinemann M (2005) Y chromosomes: born to be destroyed. BioEssays 27(10):1076–1083
Suka N, Shinohara Y, Saitoh Y, Saitoh H, Ohtomo K, Harata M, Shpigelman E, Mizuno S (1993) W-heterochromatin of chicken; its unusual DNA components, late replication, and chromatin structure. Genetica 88(2–3):93–105
Tsuda Y, Nishida-Umehara C, Ishijima J, Yamada K, Matsuda Y (2007) Comparison of the Z and W sex chromosomal architectures in elegant crested tinamou (Eudromia elegans) and ostrich (Struthio camelus) and the process of sex chromosome differentiation in palaeognathous birds. Chromosoma 116(2):159–173
Waters PD, Wallis MC, Graves JA (2007) Mammalian sex—origin and evolution of the Y chromosome and SRY. Semin Cell Dev Biol 18(3):389–400
Yamada K, Nishida-Umehara C, Ishijima J, Murakami T, Shibusawa M, Tsuchiya K, Tsudzuki M, Matsuda Y (2006) A novel family of repetitive DNA sequences amplified site-specifically on the W chromosomes in Neognathous birds. Chromosom Res 14(6):613–627
Acknowledgements
This study was supported by NIH grant DC00217 to A.P. Arnold and a Yamada Science Foundation grant to Y. Itoh. BAC clones were sequenced by the Washington University Genome Center. Thanks to Esther Melamed, Jun Kato, and Wes Warren for assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Henikoff
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
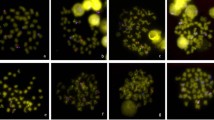
Comparison of FISH signal enhancement using BACs containing repeats or no repeats. a, b ZBM BAC 201O20 hybridization to male chromosome spreads and c, d androgen receptor BAC (319A15, Luo et al 2006) hybridization to female chromosome spreads as a control experiment. The signal (red) was enhanced identically in the two experiments, as described in Fig. 4c,d. b The amplification of ZBM signal in male metaphase spread allowed visualization of the distribution of ZBM sequence on numerous chromosomes including the Z chromosome. d In contrast, hybridization of the androgen receptor BAC showed specific labeling of a microchromosome. a, c DAPI staining. Scale bar = 10 μm. (JPG 27.5 kb)
Rights and permissions
About this article
Cite this article
Itoh, Y., Kampf, K. & Arnold, A.P. Molecular cloning of zebra finch W chromosome repetitive sequences: evolution of the avian W chromosome. Chromosoma 117, 111–121 (2008). https://doi.org/10.1007/s00412-007-0130-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-007-0130-8