Abstract
Background
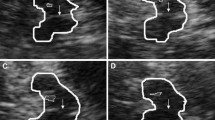
Substantia nigra hyperechogenicity (SNH) is a characteristic transcranial sonography (TCS) finding in Parkinson’s disease (PD). SNH, found also in about 10% of healthy adults, was related to a subclinical malfunction of the nigrostriatal dopaminergic system on positron emission tomography studies. Both, liability for developing PD and frequency of SNH were found to be increased in depressed subjects. Here, we investigated whether SNH in depression is related to motor or cognitive abnormalities resembling early PD.
Methods
Fourty-one patients with major depressive disorder and 15 with adjustment disorder with depressed mood were studied clinically and with TCS.
Results
Frequency of SNH was similar in both groups (39, 33%; Chi-square test, P = 0.70). Larger SN echogenic size correlated with larger right-to-left asymmetry of finger tapping (Spearman test, r = 0.37, P = 0.009) and lower verbal fluency (r = −0.35, P = 0.038). These correlations were stronger in patients at ages ≥ 50 years (r = 0.52, P = 0.007; r = −0.50, P = 0.020), and, independently from age, in patients with reduced echogenicity of brainstem raphe suggested to reflect alteration of the serotonergic system (r = 0.40, P = 0.045; r = −0.51, P = 0.044). Whereas bilateral sum score of finger tapping was negatively correlated with severity of depression on the beck depression inventory (r = −0.50, P = 0.001) and the Hamilton depression rating scale (r = −0.34, P = 0.019), no correlation was found between depression severity and tapping asymmetry, or between depression severity and verbal fluency.
Conclusion
Data suggest that TCS detects a subgroup of patients with depression characterized by symptoms of early parkinsonism who are possibly at an elevated risk of later developing definite PD.



Similar content being viewed by others

References
Alex KD, Yavanian GJ, McFarlane HG, Pluto CP, Pehek EA (2005) Modulation of dopamine release by striatal 5-HT2C receptors. Synapse 55:242–251
American Psychiatric Association (1994) Diagnostic and statistical manual of mental disorders. American Psychiatric Press, Washington DC
Beck AT, Ward CH, Mendelson M, Mock J, Erbaugh J (1961) An inventory for measuring depression. Arch Gen Psychiatry 4:561–571
Becker G, Seufert J, Bogdahn U, Reichmann H, Reiners K (1995) Degeneration of substantia nigra in chronic Parkinson’s disease visualized by transcranial color-coded real-time sonography. Neurology 45:182–184
Becker G, Becker T, Struck M, Lindner A, Burzer K, Retz W, Bogdahn U, Beckmann H (1995) Reduced echogenicity of brainstem raphe specific to unipolar depression: a transcranial color-coded real-time sonography study. Biol Psychiatry 38:180–184
Becker G, Berg D, Lesch KP, Becker T (2001) Basal limbic system alteration in major depression: a hypothesis supported by transcranial sonography and MRI findings. Int J Neuropsychopharmacol 4:21–31
Berg D, Becker G, Zeiler B, Tucha O, Hofmann E, Preier M, Benz P, Jost W, Reiners K, Lange KW (1999) Vulnerability of the nigrostriatal system as detected by transcranial ultrasound. Neurology 53:1026–1031
Berg D, Jabs B, Merschdorf U, Beckmann H, Becker G (2001) Echogenicity of substantia nigra determined by transcranial ultrasound correlates with severity of parkinsonian symptoms induced by neuroleptic therapy. Biol Psychiatry 50:463–467
Berg D, Siefker C, Ruprecht-Dörfler P, Becker G (2001) Relationship of substantia nigra echogenicity and motor function in elderly subjects. Neurology 56:13–17
Berg D, Roggendorf W, Schröder U, Klein R, Tatschner T, Benz P, Tucha O, Preier M, Lange KW, Reiners K, Gerlach M, Becker G (2002) Echogenicity of the substantia nigra: association with increased iron content and marker for susceptibility to nigrostriatal injury. Arch Neurol 56:999–1005
Berg D, Merz B, Reiners K, Naumann M, Becker G (2005) Five-year follow-up study of hyperechogenicity of the substantia nigra in Parkinson’s disease. Mov Disord 20:383–385
Brandt-Christensen M, Kvist K, Nilsson FM, Andersen PK, Kessing LV (2006) Treatment with antidepressants and lithium is associated with increased risk of treatment with antiparkinson drugs: a pharmacoepidemiological study. J Neurol Neurosurg Psychiatry 77:781–783
Brooks DJ, Doder M (2001) Depression in Parkinson’s disease. Curr Opin Neurol 14:465–470
Camicioli R, Grossmann SJ, Spencer PS, Hudnell K, Anger WK (2001) Discriminating mild parkinsonism: methods for epidemiological research. Mov Disord 16:33–40
Dujardin K, Duhamel A, Becquet E, Grunberg C, Defebvre L, Destee A (1999) Neuropsychological abnormalities in first degree relatives of patients with familial Parkinson’s disease. J Neurol Neurosurg Psychiatry 67:323–328
Fahn S, Elton RL, Members of the UPDRS Development Committee (1987) Unified Parkinson’s disease rating scale. In: Fahn S, Marsden CD, Goldstein M, Calne DB (eds) Recent developments in Parkinsońs disease II. Macmillan, New York, pp 153–163
First MB, Spitzer RL, Gibbon M, Williams JBW (1996) Structured clinical interview for DSM-IV axis I disorders, clinician version (SCID-CV). American Psychiatric Press, Washington DC
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Hamilton M (1967) Development of a rating scale for primary depressive illness. Br J Soc Clin Psychol 6:278–296
Jahanshahi M, Ardouin CM, Brown RG, Rothwell JC, Obeso J, Albanese A, Rodriguez-Oroz MC, Moro E, Benabid AL, Pollak P, Limousin-Dowsey P (2000) The impact of deep brain stimulation on executive function in Parkinson’s disease. Brain 123:1142–1154
Leentjens AF, Van den Akker M, Metsemakers JF, Lousberg R, Verhey FR (2003) Higher incidence of depression preceding the onset of Parkinson’s disease: a register study. Mov Disord 18:414–418
Lees AJ, Smith E (1983) Cognitive deficits in the early stages of Parkinson’s disease. Brain 106:257–270
Lees AJ (1992) When did Ray Kennedy’s Parkinson’s disease begin? Mov Disord 7:110–116
Lemke MR, Koethe NH, Schleidt M (2000) Segmentation of behavior and time structure of movements in depressed patients. Psychopathology 33:131–136
Lezak MD (1995) Neuropsychological assessment. Oxford University Press, New York
Nilsson FM, Kessing LV, Bolwig TG (2001) Increased risk of developing Parkinson’s disease for patients with major affective disorder: a register study. Acta Psychiatr Scand 104:380–386
Reitan RM (1985) Validity of the trail making test as an indication of organic brain damage. Percept Mot Skills 8:271–276
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9:97–113
Ruprecht-Dorfler P, Klotz P, Becker G, Berg D (2007) Substantia nigra hyperechogenicity correlates with subtle motor dysfunction in tap dancers. Parkinsonism Relat Disord 13:362–364
Schuurman AG, van den Akker M, Ensinck KT, Metsemakers JF, Knottnerus JA, Leentjens AF, Buntinx F (2002) Increased risk of Parkinson’s disease after depression: a retrospective cohort study. Neurology 58:1501–1504
Shiba M, Bower JH, Maraganore DM, McDonnell SK, Peterson BJ, Ahlskog JE, Schaid DJ, Rocca WA (2000) Anxiety disorders and depressive disorders preceding Parkinson’s disease: a case-control study. Mov Disord 15:669–677
Sobin C, Mayer L, Endicott J (1998) The motor agitation and retardation scale: a scale for the assessment of motor abnormalities in depressed patients. J Neuropsychiatry Clin Neurosci 10:85–92
Sommer U, Hummel T, Cormann K, Mueller A, Frasnelli J, Kropp J, Reichmann H (2004) Detection of presymptomatic Parkinson’s disease: combining smell tests, transcranial sonography, and SPECT. Mov Disord 19:1196–1202
Uc EY, Rizzo M, Anderson SW, Qian S, Rodnitzky RL, Dawson JD (2005) Visual dysfunction in Parkinson disease without dementia. Neurology 65:1907–1913
Van Gerpen JA (2002) Drug-induced parkinsonism. Neurologist 8:363–370
Walter U, Niehaus L, Probst T, Benecke R, Meyer BU, Dressler D (2003) Brain parenchyma sonography discriminates Parkinson’s disease and atypical parkinsonian syndromes. Neurology 60:74–77
Walter U, Klein C, Hilker R, Benecke R, Pramstaller PP, Dressler D (2004) Brain parenchyma sonography detects preclinical parkinsonism. Mov Disord 19:1445–1449
Walter U, Dressler D, Wolters A, Wittstock M, Benecke R (2007) Transcranial brain sonography findings in clinical subgroups of idiopathic Parkinson’s disease. Mov Disord 22:48–54
Walter U, Hoeppner J, Prudente-Morrissey L, Horowski S, Herpertz SC, Benecke R (2007) Parkinson’s disease-like midbrain sonography abnormalities are frequent in depressive disorders. Brain 130:1799–1807
Walter U, Prudente-Morrissey L, Herpertz SC, Benecke R, Hoeppner J (2007) Relationship of brainstem raphe echogenicity and clinical findings in depressive states. Psychiatry Res 155:67–73
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hoeppner, J., Prudente-Morrissey, L., Herpertz, S.C. et al. Substantia nigra hyperechogenicity in depressive subjects relates to motor asymmetry and impaired word fluency. Eur Arch Psychiatry Clin Neurosci 259, 92–97 (2009). https://doi.org/10.1007/s00406-008-0840-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00406-008-0840-9