Abstract
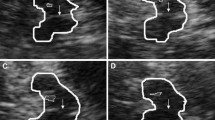
Transcranial sonography (TCS) appeared to be a promising marker associated with depression: hypo/anechogenicity of the brainstem raphe (BR) was found in 50–70 % of patients with unipolar depression, in 40–60 % of depressed patients with Parkinson’s disease (PD), but also in 8–28 % of healthy controls. Our study included 120 consecutive PD outpatients. Abnormal BR echogenicity was found in 51 (43 %), while normal findings were present in 67 PD patients (57 %). Patients with abnormal BR echogenicity had higher scores on the Montgomery–Asberg Depression Rating Scale (MADRS) items of apparent sadness (p = 0.03), reported sadness (p = 0.01), and pessimistic thoughts (p = 0.049), when compared to those with normal BR. In the second part of the study, previously suggested cut-off value at 14/15 on the MADRS was used to dichotomize patients into depressed (dPD) (46 patients; 39 %) and non-depressed PD patients (ndPD) (72 patients; 61 %). Abnormal TCS BR findings were obtained in 27 dPD (58.7 %) and in only 24 ndPD patients (33.3 %) (p = 0.007): the risk that PD patients with the TCS BR abnormality would display depressive symptoms was about 3.5 times higher when compared to PD patients with intact BR, controlling for the effect of motor difficulties (cross-odds ratio; OR = 3.48). Therefore, at least in a subgroup of dPD patients, TCS of the midbrain midline structure may potentially be a useful tool for depressive symptoms prediction.

Similar content being viewed by others

References
Reijnders JS, Ehrt U, Weber WE, Aarsland D, Leentjens AF (2008) A systematic review of prevalence studies of depression in Parkinson’s disease. Mov Disord 23:183–189
Schrag A (2006) Quality of life and depression in Parkinson’s disease. J Neurol Sci 248:151–157
Lieberman A (2006) Depression in Parkinson’s disease—a review. Acta Neurol Scand 113:1–8
Cummings JL (1992) Depression and Parkinson’s disease: a review. Am J Psychiatry 149:443–454
Brooks DJ, Piccini P (2006) Imaging of Parkinson’s disease: the role of monoamines in behaviour. Biol Psychiatry 59:908–918
Becker G, Seufert J, Bogdahn U, Reichmann H, Reiners K (1995) Degeneration of substantia nigra in chronic Parkinson’s disease visualized by transcranial color-coded real-time sonography. Neurology 45:182–184
Berg D, Siefker C, Becker G (2001) Echogenicity of the substantia nigra in Parkinson’s disease and its relation to clinical findings. J Neurol 248:684–689
Walter U, Wittstock M, Benecke R, Dressler D (2002) Substantia nigra echogenicity is normal in non-extrapyramidal cerebral disorders but increased in Parkinson’s disease. J Neural Transm 109:191–196
Berg D, Godau J, Walter U (2008) Transcranial sonography in movement disorders. Lancet Neurol 7:1044–1055
Becker G, Struck M, Bogdahn U, Becker T (1994) Echogenicity of the brainstem raphe in patients with major depression. Psychiatry Res 55:75–84
Walter U, Hoeppner J, Prudente-Morrissey L, Horowski S, Herpertz SC, Benecke R (2007) Parkinson’s disease-like midbrain sonography abnormalities are frequent in depressive disorders. Brain 130:1799–1807
Mijajlovic MD (2010) Transcranial sonography in depression. Int Rev Neurobiol 90:259–272
Becker T, Becker G, Seufert J, Hofmann E, Lange KW, Naumann M, Lindner A, Reichmann H, Riederer P, Beckmann H, Reiners K (1997) Parkinson’s disease and depression: evidence for an alteration of the basal limbic system detected by transcranial sonography. J Neurol Neurosurg Psychiatry 63:590–596
Berg D, Supprian T, Hoffmann E, Zeiler B, Jäger A, Lange KW, Reiners K, Becker T, Becker G (1999) Depression in Parkinson’s disease: brainstem midline alteration on transcranial sonography and magnetic resonance imaging. J Neurol 246:1186–1193
Becker G, Becker T, Struck M, Lindner A, Burzer K, Retz W, Bogdahn U, Beckmann H (1995) Reduced echogenicity of brainstem raphe specific to unipolar depression: a transcranial color-coded real-time sonography study. Biol Psychiatry 38:180–184
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease. A clinico-pathological study of 100 cases. JNNP 55:181–184
Dubois B, Burn D, Goetz C, Aarsland D, Brown RG, Broe GA, Dickson D, Duyckaerts C, Cummings J, Gauthier S, Korczyn A, Lees A, Levy R, Litvan I, Mizuno Y, McKeith IG, Olanow CW, Poewe W, Sampaio C, Tolosa E, Emre M (2007) Diagnostic procedures for Parkinson’s disease dementia: recommendations from the Movement Disorder Society Task Force. Mov Disord 22:2314–2324
Hoehn M, Yahr M (1967) Parkinsonism: onset, progression and mortality. Neurology 17:427–442
Goetz CG, Tilley BC, Shaftman SR, Stebbins GT, Fahn S, Martinez-Martin P, Poewe W, Sampaio C, Stern MB, Dodel R, Dubois B, Holloway R, Jankovic J, Kulisevsky J, Lang AE, Lees A, Leurgans S, LeWitt PA, Nyenhuis D, Olanow CW, Rascol O, Schrag A, Teresi JA, van Hilten JJ, LaPelle N, Movement Disorder Society UPDRS Revision Task Force (2008) Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord 23:2129–2170
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Davidson J, Turnbull CD, Strickland R, Miller R, Graves K (1986) The Montgomery Asberg Depression Scale: reliability and validity. Acta Psychiatr Scand 73:544–548
Hamilton M (1959) The assessment of anxiety states by rating. Br J Med Psychol 32:50–55
Starkstein SE, Mayberg HS, Preziosi TJ, Andrezejewski P, Leiguarda R, Robinson RG (1992) Reliability, validity, and clinical correlates of apathy in Parkinson’s disease. J Neuropsychiatry Clin Neurosci 4:134–139
Schrag A, Barone P, Brown RG, Leentjens AF, McDonald WM, Starkstein S, Weintraub D, Poewe W, Rascol O, Sampaio C, Stebbins GT, Goetz CG (2007) Depression rating scales in Parkinson’s disease: critique and recommendations. Mov Disord 22:1077–1092
Leentjens A, Dujardin K, Marsh L, Richard I, Starkstein S, Martinez-Martin P (2011) Anxiety rating scales in Parkinson’s disease: a validation study of the Hamilton anxiety rating scale, the Beck anxiety inventory, and the hospital anxiety and depression scale. Mov Disord 26:407–415
Berg D, Siefker C, Ruprecht-Dorfler P, Becker G (2001) Relationship of substantia nigra echogenicity and motor function in elderly subjects. Neurology 56:13–179
Mahlknecht P, Seppi K, Stockner H, Nocker M, Scherfler C, Kiechl S, Willeit J, Schmidauer C, Gasperi A, Rungger G, Poewe W (2013) Substantia nigra hyperechogenicity as a marker for Parkinson’s disease: a population based study. Neurodegener Dis 12:212–218
Krogias C, Strassburger K, Eyding J, Gold R, Norra C, Juckel G, Saft C, Ninphius D (2011) Depression in patients with Huntington disease correlates with alterations of the brain stem raphe depicted by transcranial sonography. J Psychiatry Neurosci 36:187–194
Cho JW, Baik JS, Lee MS (2011) Mesencephalic midline change on transcranial sonography in early Parkinson’s disease patients with depression. J Neurol Sci 310:50–52
Walter U, Skoloudík D, Berg D (2010) Transcranial sonography findings related to non-motor features of Parkinson’s disease. J Neurol Sci 289:123–127
Naumann M, Becker G, Toyka KV, Supprian T, Reiners K (1996) Lenticular nucleus lesion in idiopathic dystonia detected by transcranial sonography. Neurology 47:1284–1290
Walter U, Krolikowski K, Tarnacka B, Benecke R, Czlonkowska A, Dressler D (2005) Sonographic detection of basal ganglia lesions in asymptomatic and symptomatic Wilson disease. Neurology 64:1726–1732
Krogias C, Hoffmann K, Eyding J, Scheele D, Norra C, Gold R, Juckel G, Assion HJ (2011) Evaluation of basal ganglia, brainstem raphe and ventricles in bipolar disorder by transcranial sonography. Psychiatry Res 194:190–197
Walter U, Prudente-Morrissey L, Herpertz SC, Benecke R, Hoeppner J (2007) Relationship of brainstem raphe echogenicity and clinical findings in depressive states. Psychiatry Res 155:67–73
Becker G, Berg D, Lesch KP, Becker T (2001) Basal limbic system alteration in major depression: a hypothesis supported by transcranial sonography and MRI findings. Int J Neuropsychopharmacol 4:21–31
Paulus W, Jellinger K (1991) The neuropathologic basis of different clinical subgroups of Parkinson’s disease. J Neuropathol Exp Neurol 50:743–755
Boileau I, Warsh JJ, Guttman M, Saint-Cyr JA, McCluskey T, Rusjan P, Houle S, Wilson AA, Meyer JH, Kish SJ (2008) Elevated serotonin transporter binding in depressed patients with Parkinson’s disease: a preliminary PET study with [11C]DASB. Mov Disord 23:1776–1780
Politis M, Wu K, Loane C, Kiferle L, Molloy S, Brooks DJ, Piccini P (2010) Staging of serotonergic dysfunction in Parkinson’s disease: an in vivo 11C-DASB PET study. Neurobiol Dis 40:216–221
Prediger RD, Matheus FC, Schwarzbold ML, Lima MM, Vital MA (2012) Anxiety in Parkinson’s disease: a critical review of experimental and clinical studies. Neuropharmacology 62:115–124
Mavrogiorgou P, Nalato F, Meves S, Luksnat S, Norra C, Gold R, Juckel G, Krogias C (2013) Transcranial sonography in obsessive–compulsive disorder. J Psych Res 47:1642–1648
Lowry CA, Hale MW, Evans AK, Heerkens J, Staub DR, Gasser PJ, Shekhar A (2008) Serotonergic systems, anxiety, and affective disorder: focus on the dorsomedial part of the dorsal raphe nucleus. Ann NY Acad Sci 1148:86–94
Huot P, Fox SH (2013) The serotonergic system in motor and non-motor manifestations of Parkinson’s disease. Exp Brain Res 230:463–476
Even C, Weintraub D (2012) Is depression in Parkinson’s disease (PD) a specific entity? J Affect Disord 139:103–112
Gallagher DA, Schrag A (2012) Psychosis, apathy, depression and anxiety in Parkinson’s disease. Neurobiol Dis 46:581–589
Acknowledgments
This work has been supported by the Ministry of Education, Science, and Technological Development, Republic of Serbia (Project No. 175090).
Conflicts of interest
IS, LjZ, MM and AP declare no conflict of interest. ES receives speaker’s honoraria from Novartis, Boehringer Ingelheim, Pfizer and Lundbeck. VSK receives honoraria from Novartis, Boehringer Ingelheim, Pfizer, Lundbeck, Glaxo-Smith-Kline, Swiss-Pharm and Eurolek and a grant form SANU.
Ethical standard
The research presented in this study was approved by the Ethical Committee of the School of Medicine, University of Belgrade (Serbia), and written informed consent was obtained from each patient prior to their inclusion in this study. The authors declare that they acted in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stanković, I., Stefanova, E., Žiropadja, L. et al. Transcranial midbrain sonography and depressive symptoms in patients with Parkinson’s disease. J Neurol 262, 689–695 (2015). https://doi.org/10.1007/s00415-014-7624-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-014-7624-0