Abstract
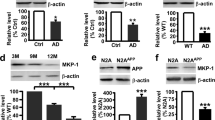
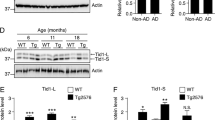
Altered proteostasis is a salient feature of Alzheimer’s disease (AD), highlighting the occurrence of endoplasmic reticulum (ER) stress and abnormal protein aggregation. ER stress triggers the activation of the unfolded protein response (UPR), a signaling pathway that enforces adaptive programs to sustain proteostasis or eliminate terminally damaged cells. IRE1 is an ER-located kinase and endoribonuclease that operates as a major stress transducer, mediating both adaptive and proapoptotic programs under ER stress. IRE1 signaling controls the expression of the transcription factor XBP1, in addition to degrade several RNAs. Importantly, a polymorphism in the XBP1 promoter was suggested as a risk factor to develop AD. Here, we demonstrate a positive correlation between the progression of AD histopathology and the activation of IRE1 in human brain tissue. To define the significance of the UPR to AD, we targeted IRE1 expression in a transgenic mouse model of AD. Despite initial expectations that IRE1 signaling may protect against AD, genetic ablation of the RNase domain of IRE1 in the nervous system significantly reduced amyloid deposition, the content of amyloid β oligomers, and astrocyte activation. IRE1 deficiency fully restored the learning and memory capacity of AD mice, associated with improved synaptic function and improved long-term potentiation (LTP). At the molecular level, IRE1 deletion reduced the expression of amyloid precursor protein (APP) in cortical and hippocampal areas of AD mice. In vitro experiments demonstrated that inhibition of IRE1 downstream signaling reduces APP steady-state levels, associated with its retention at the ER followed by proteasome-mediated degradation. Our findings uncovered an unanticipated role of IRE1 in the pathogenesis of AD, offering a novel target for disease intervention.







Similar content being viewed by others
References
Abisambra JF, Jinwal UK, Blair LJ et al (2013) Tau accumulation activates the unfolded protein response by impairing endoplasmic reticulum-associated degradation. J Neurosci 33:9498–9507. doi:10.1523/JNEUROSCI.5397-12.2013
Acosta-Alvear D, Zhou Y, Blais A et al (2007) XBP1 controls diverse cell type- and condition-specific transcriptional regulatory networks. Mol Cell 27:53–66. doi:10.1016/j.molcel.2007.06.011
Alafuzoff I, Pikkarainen M, Neumann M et al (2015) Neuropathological assessments of the pathology in frontotemporal lobar degeneration with TDP43-positive inclusions: an inter-laboratory study by the BrainNet Europe consortium. J Neural Transm (Vienna) 122:957–972. doi:10.1007/s00702-014-1304-1
Ballard C, Gauthier S, Corbett A et al (2011) Alzheimer’s disease. Lancet 377:1019–1031. doi:10.1016/S0140-6736(10)61349-9
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82:239–259
Burgos PV, Mardones GA, Rojas AL et al (2010) Sorting of the Alzheimer’s disease amyloid precursor protein mediated by the AP-4 complex. Dev Cell 18:425–436. doi:10.1016/j.devcel.2010.01.015
Calfon M, Zeng H, Urano F et al (2002) IRE1 couples endoplasmic reticulum load to secretory capacity by processing the XBP-1 mRNA. Nature 415:92–96. doi:10.1038/415092a
Casas-Tinto S, Zhang Y, Sanchez-Garcia J et al (2011) The ER stress factor XBP1 s prevents amyloid-beta neurotoxicity. Hum Mol Genet 20:2144–2160. doi:10.1093/hmg/ddr100
Cescon M, Chen P, Castagnaro S, Gregorio I, Bonaldo P (2016) Lack of collagen VI promotes neurodegeneration by impairing autophagy and inducing apoptosis during aging. Aging (Albany NY) 8:1083–1101. doi:10.18632/aging.100924
Chen X, Iliopoulos D, Zhang Q et al (2014) XBP1 promotes triple-negative breast cancer by controlling the HIF1alpha pathway. Nature 508:103–107. doi:10.1038/nature13119
Cheng JS, Dubal DB, Kim DH et al (2009) Collagen VI protects neurons against Abeta toxicity. Nat Neurosci 12:119–121. doi:10.1038/nn.2240
Clayton BL, Popko B (2016) Endoplasmic reticulum stress and the unfolded protein response in disorders of myelinating glia. Brain Res 1:594–602. doi:10.1016/j.brainres.2016.03.046
Cornejo VH, Hetz C (2013) The unfolded protein response in Alzheimer’s disease. Semin Immunopathol 35:277–292. doi:10.1007/s00281-013-0373-9
Cornejo VH, Pihan P, Vidal RL, Hetz C (2013) Role of the unfolded protein response in organ physiology: lessons from mouse models. IUBMB Life 65:962–975. doi:10.1002/iub.1224
Costa-Mattioli M, Gobert D, Harding H et al (2005) Translational control of hippocampal synaptic plasticity and memory by the eIF2alpha kinase GCN2. Nature 436:1166–1173. doi:10.1038/nature03897
Costa-Mattioli M, Sossin WS, Klann E, Sonenberg N (2009) Translational control of long-lasting synaptic plasticity and memory. Neuron 61:10–26. doi:10.1016/j.neuron.2008.10.055
Cuanalo-Contreras K, Mukherjee A, Soto C (2013) Role of protein misfolding and proteostasis deficiency in protein misfolding diseases and aging. Int J Cell Biol 2013:638083. doi:10.1155/2013/638083
Devi L, Ohno M (2013) Deletion of the eIF2alpha Kinase GCN2 fails to rescue the memory decline associated with Alzheimer’s disease. PLoS OnE 8:e77335. doi:10.1371/journal.pone.0077335
Devi L, Ohno M (2014) PERK mediates eIF2alpha phosphorylation responsible for BACE1 elevation, CREB dysfunction and neurodegeneration in a mouse model of Alzheimer’s disease. Neurobiol Aging 35:2272–2281. doi:10.1016/j.neurobiolaging.2014.04.031
Douglas PM, Dillin A (2010) Protein homeostasis and aging in neurodegeneration. J Cell Biol 190:719–729. doi:10.1083/jcb.201005144
Du J, Duan S, Wang H et al (2008) Comprehensive analysis of polymorphisms throughout GAD1 gene: a family-based association study in schizophrenia. J Neural Transm 115:513–519. doi:10.1007/s00702-007-0844-z
Duran-Aniotz C, Martinez G, Hetz C (2014) Memory loss in Alzheimer’s disease: are the alterations in the UPR network involved in the cognitive impairment? Front Aging Neurosci 6:8. doi:10.3389/fnagi.2014.00008
Duran-Aniotz C, Morales R, Moreno-Gonzalez I et al (2014) Aggregate-depleted brain fails to induce Abeta deposition in a mouse model of Alzheimer’s disease. PLoS One 9:e89014. doi:10.1371/journal.pone.0089014
Duran-Aniotz C, Morales R, Moreno-Gonzalez I, Hu PP, Soto C (2013) Brains from non-Alzheimer’s individuals containing amyloid deposits accelerate Abeta deposition in vivo. Acta Neuropathol Commun 1:76. doi:10.1186/2051-5960-1-76
Eimer WA, Vassar R (2013) Neuron loss in the 5XFAD mouse model of Alzheimer’s disease correlates with intraneuronal Abeta42 accumulation and Caspase-3 activation. Mol Neurodegener 8:2. doi:10.1186/1750-1326-8-2
Freeman OJ, Mallucci GR (2016) The UPR and synaptic dysfunction in neurodegeneration. Brain Res 1:530–537. doi:10.1016/j.brainres.2016.03.029
Gambella M, Rocci A, Passera R et al (2014) High XBP1 expression is a marker of better outcome in multiple myeloma patients treated with bortezomib. Haematologica 99:e14–16. doi:10.3324/haematol.2013.090142
Ghosh R, Wang L, Wang ES et al (2014) Allosteric inhibition of the IRE1alpha RNase preserves cell viability and function during endoplasmic reticulum stress. Cell 158:534–548. doi:10.1016/j.cell.2014.07.002
Granger AJ, Nicoll RA (2014) Expression mechanisms underlying long-term potentiation: a postsynaptic view, 10 years on. Philos Trans R Soc Lond B Biol Sci 369:20130136. doi:10.1098/rstb.2013.0136
Han D, Lerner AG, Vande Walle L et al (2009) IRE1alpha kinase activation modes control alternate endoribonuclease outputs to determine divergent cell fates. Cell 138:562–575. doi:10.1016/j.cell.2009.07.017
Hetz C, Chevet E, Harding HP (2013) Targeting the unfolded protein response in disease. Nat Rev Drug Discov 12:703–719. doi:10.1038/nrd3976
Hetz C, Chevet E, Oakes SA (2015) Proteostasis control by the unfolded protein response. Nat Cell Biol 17:829–838. doi:10.1038/ncb3184
Hetz C, Lee AH, Gonzalez-Romero D et al (2008) Unfolded protein response transcription factor XBP-1 does not influence prion replication or pathogenesis. Proc Natl Acad Sci USA 105:757–762. doi:10.1073/pnas.0711094105
Hetz C, Mollereau B (2014) Disturbance of endoplasmic reticulum proteostasis in neurodegenerative diseases. Nat Rev Neurosci 15:233–249. doi:10.1038/nrn3689
Hetz C, Thielen P, Matus S et al (2009) XBP-1 deficiency in the nervous system protects against amyotrophic lateral sclerosis by increasing autophagy. Genes Dev 23:2294–2306. doi:10.1101/gad.1830709
Hollien J, Lin JH, Li H et al (2009) Regulated Ire1-dependent decay of messenger RNAs in mammalian cells. J Cell Biol 186:323–331. doi:10.1083/jcb.200903014
Hoozemans JJ, van Haastert ES, Nijholt DA et al (2009) The unfolded protein response is activated in pretangle neurons in Alzheimer’s disease hippocampus. Am J Pathol 174:1241–1251. doi:10.2353/ajpath.2009.080814
Hu Y, Park KK, Yang L et al (2012) Differential effects of unfolded protein response pathways on axon injury-induced death of retinal ganglion cells. Neuron 73:445–452. doi:10.1016/j.neuron.2011.11.026
Iwawaki T, Akai R, Kohno K (2010) IRE1alpha disruption causes histological abnormality of exocrine tissues, increase of blood glucose level, and decrease of serum immunoglobulin level. PLoS One 5:e13052. doi:10.1371/journal.pone.0013052
Iwawaki T, Akai R, Yamanaka S, Kohno K (2009) Function of IRE1 alpha in the placenta is essential for placental development and embryonic viability. Proc Natl Acad Sci USA 106:16657–16662. doi:10.1073/pnas.0903775106
Jiang Z, Belforte JE, Lu Y et al (2010) eIF2alpha Phosphorylation-dependent translation in CA1 pyramidal cells impairs hippocampal memory consolidation without affecting general translation. J Neurosci 30:2582–2594. doi:10.1523/JNEUROSCI.3971-09.2010
Kakiuchi C, Iwamoto K, Ishiwata M et al (2003) Impaired feedback regulation of XBP1 as a genetic risk factor for bipolar disorder. Nat Genet 35:171–175. doi:10.1038/ng1235
Kaushik S, Cuervo AM (2015) Proteostasis and aging. Nat Med 21:1406–1415. doi:10.1038/nm.4001
Kennedy BK, Berger SL, Brunet A et al (2014) Geroscience: linking aging to chronic disease. Cell 159:709–713. doi:10.1016/j.cell.2014.10.039
Kim B, Kim CY, Lee MJ, Joo YH (2009) Preliminary evidence on the association between XBP1-116C/G polymorphism and response to prophylactic treatment with valproate in bipolar disorders. Psychiatry Res 168:209–212. doi:10.1016/j.psychres.2008.05.010
Kondo T, Asai M, Tsukita K et al (2013) Modeling Alzheimer’s disease with iPSCs reveals stress phenotypes associated with intracellular abeta and differential drug responsiveness. Cell Stem Cell 4:487–496. doi:10.1016/j.stem.2013.01.009
Labbadia J, Morimoto RI (2015) The biology of proteostasis in aging and disease. Annu Rev Biochem 84:435–464. doi:10.1146/annurev-biochem-060614-033955
Lee AH, Iwakoshi NN, Glimcher LH (2003) XBP-1 regulates a subset of endoplasmic reticulum resident chaperone genes in the unfolded protein response. Mol Cell Biol 23:7448–7459
Lee JH, Won SM, Suh J et al (2010) Induction of the unfolded protein response and cell death pathway in Alzheimer’s disease, but not in aged Tg2576 mice. Exp Mol Med 42:386–394
Lee JH, Yu WH, Kumar A et al (2010) Lysosomal proteolysis and autophagy require presenilin 1 and are disrupted by Alzheimer-related PS1 mutations. Cell 141:1146–1158. doi:10.1016/j.cell.2010.05.008
Lee K, Tirasophon W, Shen X et al (2002) IRE1-mediated unconventional mRNA splicing and S2P-mediated ATF6 cleavage merge to regulate XBP1 in signaling the unfolded protein response. Genes Dev 16:452–466. doi:10.1101/gad.964702
Lerner AG, Upton JP, Praveen PV et al (2012) IRE1alpha induces thioredoxin-interacting protein to activate the NLRP3 inflammasome and promote programmed cell death under irremediable ER stress. Cell Metab 16:250–264. doi:10.1016/j.cmet.2012.07.007
Loewen CA, Feany MB (2010) The unfolded protein response protects from tau neurotoxicity in vivo. PLoS One 5:e13084. doi:10.1371/journal.pone.0013084
Lopez-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G (2013) The hallmarks of aging. Cell 153:1194–1217. doi:10.1016/j.cell.2013.05.039
Lourenco MV, Clarke JR, Frozza RL et al (2013) TNF-alpha mediates PKR-dependent memory impairment and brain IRS-1 inhibition induced by Alzheimer’s beta-amyloid oligomers in mice and monkeys. Cell Metab 18:831–843. doi:10.1016/j.cmet.2013.11.002
Ma T, Trinh MA, Wexler AJ et al (2013) Suppression of eIF2alpha kinases alleviates Alzheimer’s disease-related plasticity and memory deficits. Nat Neurosci 16:1299–1305. doi:10.1038/nn.3486
Maly DJ, Papa FR (2014) Druggable sensors of the unfolded protein response. Nat Chem Biol 10:892–901. doi:10.1038/nchembio.1664
Martinez G, Vidal RL, Mardones P et al (2016) Regulation of Memory Formation by the Transcription Factor XBP1. Cell Rep 16:1382–1394. doi:10.1016/j.celrep.2016.01.028
Maurel M, Chevet E, Tavernier J, Gerlo S (2014) Getting RIDD of RNA: IRE1 in cell fate regulation. Trends Biochem Sci 39:245–254. doi:10.1016/j.tibs.2014.02.008
Medinas DB, Hetz C (2013) Proteostasis impairment: at the intersection between Alzheimer’s disease and diabetes. Cell Metab 18:771–772. doi:10.1016/j.cmet.2013.11.009
Moreno JA, Halliday M, Molloy C et al (2013) Oral treatment targeting the unfolded protein response prevents neurodegeneration and clinical disease in prion-infected mice. Sci Transl Med 5:206ra138. doi:10.1126/scitranslmed.3006767
Moreno JA, Radford H, Peretti D et al (2012) Sustained translational repression by eIF2alpha-P mediates prion neurodegeneration. Nature 485:507–511. doi:10.1038/nature11058
Nijholt DA, van Haastert ES, Rozemuller AJ, Scheper W, Hoozemans JJ (2012) The unfolded protein response is associated with early tau pathology in the hippocampus of tauopathies. J Pathol 226:693–702. doi:10.1002/path.3969
O’Connor T, Sadleir KR, Maus E et al (2008) Phosphorylation of the translation initiation factor eIF2alpha increases BACE1 levels and promotes amyloidogenesis. Neuron 60:988–1009. doi:10.1016/j.neuron.2008.10.047
Oakes SA, Papa FR (2015) The role of endoplasmic reticulum stress in human pathology. Annu Rev Pathol 10:173–194. doi:10.1146/annurev-pathol-012513-104649
Oakley H, Cole SL, Logan S et al (2006) Intraneuronal beta-amyloid aggregates, neurodegeneration, and neuron loss in transgenic mice with five familial Alzheimer’s disease mutations: potential factors in amyloid plaque formation. J Neurosci 26:10129–10140. doi:10.1523/JNEUROSCI.1202-06.2006
Ohno M, Chang L, Tseng W et al (2006) Temporal memory deficits in Alzheimer’s mouse models: rescue by genetic deletion of BACE1. Eur J Neurosci 23:251–260. doi:10.1111/j.1460-9568.2005.04551.x
Ohno M, Cole SL, Yasvoina M et al (2007) BACE1 gene deletion prevents neuron loss and memory deficits in 5XFAD APP/PS1 transgenic mice. Neurobiol Dis 26:134–145. doi:10.1016/j.nbd.2006.12.008
Onate M, Catenaccio A, Martinez G et al (2016) Activation of the unfolded protein response promotes axonal regeneration after peripheral nerve injury. Sci Rep 6:21709. doi:10.1038/srep21709
Peng Y, Kim MJ, Hullinger R et al (2016) Improved proteostasis in the secretory pathway rescues Alzheimer’s disease in the mouse. Brain 139:937–952. doi:10.1093/brain/awv385
Placido AI, Pereira CM, Duarte AI et al (2014) The role of endoplasmic reticulum in amyloid precursor protein processing and trafficking: implications for Alzheimer’s disease. Biochim Biophys Acta 1842:1444–1453. doi:10.1016/j.bbadis.2014.05.003
Pluquet O, Dejeans N, Bouchecareilh M et al (2013) Posttranscriptional regulation of PER1 underlies the oncogenic function of IREalpha. Cancer Res 73:4732–4743. doi:10.1158/0008-5472.CAN-12-3989
Ramirez O, Garcia A, Rojas R, Couve A, Hartel S (2010) Confined displacement algorithm determines true and random colocalization in fluorescence microscopy. J Microsc 239:173–183. doi:10.1111/j.1365-2818.2010.03369.x
Reimold AM, Etkin A, Clauss I et al (2000) An essential role in liver development for transcription factor XBP-1. Genes Dev 14:152–157
Reinhardt S, Schuck F, Grosgen S et al (2014) Unfolded protein response signaling by transcription factor XBP-1 regulates ADAM10 and is affected in Alzheimer’s disease. FASEB J 28:978–997. doi:10.1096/fj.13-234864
Sado M, Yamasaki Y, Iwanaga T et al (2009) Protective effect against Parkinson’s disease-related insults through the activation of XBP1. Brain Res 1257:16–24. doi:10.1016/j.brainres.2008.11.104
Scheper W, Hoozemans JJ (2015) The unfolded protein response in neurodegenerative diseases: a neuropathological perspective. Acta Neuropathol 130:315–331. doi:10.1007/s00401-015-1462-8
Selkoe DJ (2008) Soluble oligomers of the amyloid beta-protein impair synaptic plasticity and behavior. Behav Brain Res 192:106–113. doi:10.1016/j.bbr.2008.02.016
Shankar GM, Li S, Mehta TH et al (2008) Amyloid-beta protein dimers isolated directly from Alzheimer’s brains impair synaptic plasticity and memory. Nat Med 14:837–842. doi:10.1038/nm1782
Smith HL, Mallucci GR (2016) The unfolded protein response: mechanisms and therapy of neurodegeneration. Brain 139:2113–2121. doi:10.1093/brain/aww101
Song Y, Sretavan D, Salegio EA et al (2015) Regulation of axon regeneration by the RNA repair and splicing pathway. Nat Neurosci 18:817–825. doi:10.1038/nn.4019
Stutzbach LD, Xie SX, Naj AC et al (2013) The unfolded protein response is activated in disease-affected brain regions in progressive supranuclear palsy and Alzheimer’s disease. Acta Neuropathol Commun 1:31. doi:10.1186/2051-5960-1-31
Thal DR, Rub U, Orantes M, Braak H (2002) Phases of A beta-deposition in the human brain and its relevance for the development of AD. Neurology 58:1791–1800
Trinh MA, Kaphzan H, Wek RC et al (2012) Brain-specific disruption of the eIF2alpha kinase PERK decreases ATF4 expression and impairs behavioral flexibility. Cell Rep 1:676–688. doi:10.1016/j.celrep.2012.04.010
Trinh MA, Ma T, Kaphzan H et al (2014) The eIF2alpha kinase PERK limits the expression of hippocampal metabotropic glutamate receptor-dependent long-term depression. Learn Mem 21:298–304. doi:10.1101/lm.032219.113
Uchihara T (2007) Silver diagnosis in neuropathology: principles, practice and revised interpretation. Acta Neuropathol 113:483–499. doi:10.1007/s00401-007-0200-2
Unterberger U, Hoftberger R, Gelpi E et al (2006) Endoplasmic reticulum stress features are prominent in Alzheimer disease but not in prion diseases in vivo. J Neuropathol Exp Neurol 65:348–357. doi:10.1097/01.jnen.0000218445.30535.6f
Upton JP, Wang L, Han D et al (2012) IRE1alpha cleaves select microRNAs during ER stress to derepress translation of proapoptotic Caspase-2. Science 338:818–822. doi:10.1126/science.1226191
Urra H, Dufey E, Lisbona F, Rojas-Rivera D, Hetz C (2013) When ER stress reaches a dead end. Biochim Biophys Acta 1833:3507–3517. doi:10.1016/j.bbamcr.2013.07.024
Valdes P, Mercado G, Vidal RL et al (2014) Control of dopaminergic neuron survival by the unfolded protein response transcription factor XBP1. Proc Natl Acad Sci USA 111:6804–6809. doi:10.1073/pnas.1321845111
Valenzuela V, Collyer E, Armentano D et al (2012) Activation of the unfolded protein response enhances motor recovery after spinal cord injury. Cell Death Dis 3:e272. doi:10.1038/cddis.2012.8
Verwey NA, Hoozemans JJ, Korth C et al (2013) Immunohistochemical characterization of novel monoclonal antibodies against the N-terminus of amyloid beta-peptide. Amyloid 20:179–187. doi:10.3109/13506129.2013.797389
Viana RJ, Nunes AF, Rodrigues CM (2012) Endoplasmic reticulum enrollment in Alzheimer’s Disease. Mol Neurobiol 46:522–534. doi:10.1007/s12035-012-8301-x
Vidal RL, Figueroa A, Court FA et al (2012) Targeting the UPR transcription factor XBP1 protects against Huntington’s disease through the regulation of FoxO1 and autophagy. Hum Mol Genet 21:2245–2262. doi:10.1093/hmg/dds040
Vorhees CV, Williams MT (2006) Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc 1:848–858. doi:10.1038/nprot.2006.116
Walter P, Ron D (2011) The unfolded protein response: from stress pathway to homeostatic regulation. Science 334:1081–1086. doi:10.1126/science.1209038
Wang M, Kaufman RJ (2016) Protein misfolding in the endoplasmic reticulum as a conduit to human disease. Nature 529:326–335. doi:10.1038/nature17041
Xu T, Yang L, Yan C et al (2014) The IRE1alpha-XBP1 pathway regulates metabolic stress-induced compensatory proliferation of pancreatic beta-cells. Cell Res 24:1137–1140. doi:10.1038/cr.2014.55
Yang DS, Stavrides P, Mohan PS et al (2011) Reversal of autophagy dysfunction in the TgCRND8 mouse model of Alzheimer’s disease ameliorates amyloid pathologies and memory deficits. Brain 134:258–277. doi:10.1093/brain/awq341
Yang DS, Stavrides P, Saito M et al (2014) Defective macroautophagic turnover of brain lipids in the TgCRND8 Alzheimer mouse model: prevention by correcting lysosomal proteolytic deficits. Brain 137:3300–3318. doi:10.1093/brain/awu278
Yang J, Cheng D, Zhou S et al (2015) Overexpression of X-Box binding protein 1 (XBP1) correlates to poor prognosis and Up-regulation of PI3 K/mTOR in human osteosarcoma. Int J Mol Sci 16:28635–28646. doi:10.3390/ijms161226123
Yang W, Zhou X, Zimmermann HR et al (2016) Repression of the eIF2alpha kinase PERK alleviates mGluR-LTD impairments in a mouse model of Alzheimer’s disease. Neurobiol Aging 41:19–24. doi:10.1016/j.neurobiolaging.2016.02.005
Yoshida H, Matsui T, Yamamoto A, Okada T, Mori K (2001) XBP1 mRNA is induced by ATF6 and spliced by IRE1 in response to ER stress to produce a highly active transcription factor. Cell 107:881–891
Acknowledgements
We thank Javiera Ponce for technical support in animal care supervision. We also thank Dr. Alejandra Alvarez from The Pontifical Catholic University of Chile for the use of behavioral facilities and Dr. Patricia Burgos from University Austral of Chile for providing APP-GFP constructs. Additionally, we would like to thank Dr. Rodrigo Morales, Dr. Ines Moreno-Gonzalez, and Dr. Mohammad Shahnawaz of University of Texas Houston Medical School at Houston and Dr. Rene Vidal and Dr. Gabriela Martinez Bravo of University of Chile, for providing helpful support. We also thank the Netherlands Brain Bank for supplying the human brain tissue, Wouter Gerritsen, Tjado Morrema, Kimberley Ummenthum, and Fabian Bangel for technical assistance in human studies. This work was directly funded by FONDAP program 15150012, CONICYT-Brazil cooperation Grant 441921/2016-7, Office of Naval Research Global (ONR-G) N62909-16-1-2003, Millennium Institute P09-015-F, FONDEF ID16I10223 (CH) and FONDECYT no 11160760 (CDA). We also thank the support Muscular Dystrophy Association 382453, FONDECYT No. 1140549, and ALSRP Therapeutic Idea Award AL150111, U.S. Air Force Office of Scientific Research FA9550-16-1-0384, European Commission R&D, MSCA-RISE #734749 (CH), FONDECYT Grant No. 3160725 (VHC), FONDECYT Grant No. 11150776 (AOA), and FONDECYT Grant No. 11150579 (DBM) and Rotary International Global Grant for Disease Treatment and Prevention (AF) and Millennium Institute ICM-P09-022-F (AP).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
C. Duran-Aniotz and V. H. Cornejo contributed equally to this work
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Duran-Aniotz, C., Cornejo, V.H., Espinoza, S. et al. IRE1 signaling exacerbates Alzheimer’s disease pathogenesis. Acta Neuropathol 134, 489–506 (2017). https://doi.org/10.1007/s00401-017-1694-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-017-1694-x