Abstract
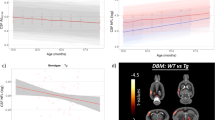
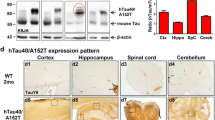
The aim of the present study was to identify the relationship between progressive neurobehavioural decline and phospho-tau levels (p-tau181) in the cerebrospinal fluid (CSF) and the brain in transgenic rats expressing human truncated tau protein. Behavioural analyses, as quantified using the NeuroScale scoring method, revealed that the transgenic rats fell into two main groups based on the baseline behavioural functioning: (1) mild neurobehavioural impairment (MNI, score 3.3–26) and (2) severe neurobehavioural impairment (SNI, score 36–44). SNI transgenic rats showed a significant increase in brain sarkosyl insoluble p-tau181 when compared to their MNI counterparts. In order to determine whether CSF phospho-tau reflects the behavioural decline and increase in sarkosyl insoluble tau in the brain, p-tau181 was measured in the CSF in a longitudinal study. The study showed a significant increase in CSF p-tau181 during the progression of the disease from MNI to SNI. Moreover, increased levels of p-tau181 in CSF correlated with an increase in the sarkosyl insoluble p-tau181 levels in the brain. The increase in the CSF level of p-tau181 during progressive behavioural decline suggests that it may represent a useful surrogate biomarker for preclinical drug development and a potential surrogate endpoint for clinical trials of disease-modifying therapy for Alzheimer’s disease and related human tauopathies.




Similar content being viewed by others
References
Andersson C, Blennow K, Almkvist O et al (2008) Increasing CSF phospho-tau levels during cognitive decline and progression to dementia. Neurobiol Aging 29:1466–1473
Andreasen N, Hesse C, Davidsson P et al (1999) Cerebrospinal fluid beta-amyloid(1–42) in Alzheimer disease: differences between early- and late-onset Alzheimer disease and stability during the course of disease. Arch Neurol 56:673–680
Andreasen N, Minthon L, Clarberg A et al (1999) Sensitivity, specificity, and stability of CSF-tau in AD in a community-based patient sample. Neurology 53:1488–1494
Andreasen N, Sjogren M, Blennow K (2003) CSF markers for Alzheimer’s disease: total tau, phospho-tau and Abeta42. World J Biol Psychiatry 4:147–155
Andreasen N, Blennow K (2005) CSF biomarkers for mild cognitive impairment and early Alzheimer’s disease. Clin Neurol Neurosurg 107:165–173
Blennow K, Wallin A, Agren H et al (1995) Tau protein in cerebrospinal fluid: a biochemical marker for axonal degeneration in Alzheimer disease? Mol Chem Neuropathol 26:231–245
Blennow K, Vanmechelen E, Hampel H (2001) CSF total tau, Abeta42 and phosphorylated tau protein as biomarkers for Alzheimer’s disease. Mol Neurobiol 24:87–97
Blennow K, Hampel H (2003) CSF markers for incipient Alzheimer’s disease. Lancet Neurol 2:605–613
Blennow K, Vanmechelen E (2003) CSF markers for pathogenic processes in Alzheimer’s disease: diagnostic implications and use in clinical neurochemistry. Brain Res Bull 61:235–242
Blennow K (2005) CSF biomarkers for Alzheimer’s disease: use in early diagnosis and evaluation of drug treatment. Expert Rev Mol Diagn 5:661–672
Blennow K, Zetterberg H, Minthon L et al (2007) Longitudinal stability of CSF biomarkers in Alzheimer’s disease. Neurosci Lett 419:18–22
Bouwman FH, van der Flier WM, Schoonenboom NS et al (2007) Longitudinal changes of CSF biomarkers in memory clinic patients. Neurology 69:1006–1011
Bouwman FH, Schoonenboom NS, Verwey NA et al (2009) CSF biomarker levels in early and late onset Alzheimer’s disease. Neurobiol Aging 30:1895–1901
Degerman Gunnarsson M, Kilander L, Basun H et al (2007) Reduction of phosphorylated tau during memantine treatment of Alzheimer’s disease. Dement Geriatr Cogn Disord 24:247–252
de Leon MJ, Segal S, Tarshish CY et al (2002) Longitudinal cerebrospinal fluid tau load increases in mild cognitive impairment. Neurosci Lett 333:183–186
de Leon MJ, DeSanti S, Zinkowski R et al (2006) Longitudinal CSF and MRI biomarkers improve the diagnosis of mild cognitive impairment. Neurobiol Aging 27:394–401
Diniz BS, Pinto JA Jr, Forlenza OV (2007) Do CSF total tau, phosphorylated tau, and beta-amyloid 42 help to predict progression of mild cognitive impairment to Alzheimer’s disease? A systematic review and meta-analysis of the literature. World J Biol Psychiatry 9:1–11
Ewers M, Buerger K, Teipel SJ et al (2007) Multicenter assessment of CSF-phosphorylated tau for the prediction of conversion of MCI. Neurology 69:2205–2212
Formichi P, Battisti C, Radi E et al (2006) Cerebrospinal fluid tau, A beta, and phosphorylated tau protein for the diagnosis of Alzheimer’s disease. J Cell Physiol 208:39–46
Frankfort SV, Tulner LR, van Campen JP et al (2008) Amyloid beta protein and tau in cerebrospinal fluid and plasma as biomarkers for dementia: a review of recent literature. Curr Clin Pharmacol 3:123–131
Greenberg SG, Davies PA (1990) Preparation of Alzheimer paired helical filaments that displays distinct tau proteins by polyacrylamide gel electrophoresis. Proc Natl Acad Sci USA 87:5827–5831
Hampel H, Buerger K, Kohnken R et al (2001) Tracking of Alzheimer’s disease progression with cerebrospinal fluid tau protein phosphorylated at threonine 231. Ann Neurol 49:545–546
Hansson O, Zetterberg H, Buchhave P et al (2006) Association between CSF biomarkers and incipient Alzheimer’s disease in patients with mild cognitive impairment: a follow-up study. Lancet Neurol 5:228–234
Herukka SK, Hallikainen M, Soininen H et al (2005) CSF Abeta42 and tau or phosphorylated tau and prediction of progressive mild cognitive impairment. Neurology 64:1294–1297
Hrnkova M, Zilka N, Minichova Z et al (2007) Neurodegeneration caused by expression of human truncated tau leads to progressive neurobehavioural impairment in transgenic rats. Brain Res 1130:206–213
Hu YY, He SS, Wang X et al (2002) Levels of nonphosphorylated and phosphorylated tau in cerebrospinal fluid of Alzheimer’s disease patients: an ultrasensitive bienzyme-substrate-recycle enzyme-linked immunosorbent assay. Am J Pathol 160:1269–1278
Hulstaert F, Blennow K, Ivanoiu A et al (1999) Improved discrimination of AD patients using beta-amyloid(1–42) and tau levels in CSF. Neurology 52:1555–1562
Iqbal K, Flory M, Khatoon S et al (2005) Subgroups of Alzheimer’s disease based on cerebrospinal fluid molecular markers. Ann Neurol 58:748–757
Ishiguro K, Ohno H, Arai H et al (1999) Phosphorylated tau in human cerebrospinal fluid is a diagnostic marker for Alzheimer’s disease. Neurosci Lett 270:91–94
Kanai M, Matsubara E, Isoe K et al (1998) Longitudinal study of cerebrospinal fluid levels of tau, A beta1–40, and A beta1–42(43) in Alzheimer’s disease: a study in Japan. Ann Neurol 44:17–26
Kluver H, Barrera E (1953) A method for the combined staining of cells and fibers in the nervous system. J Neuropathol Exp Neurol 12:400–403
Kohnken R, Buerger K, Zinkowski R et al (2000) Detection of tau phosphorylated at threonine 231 in cerebrospinal fluid of Alzheimer’s disease patients. Neurosci Lett 287:187–190
Korenova M, Zilka N, Stozicka Z et al (2009) NeuroScale, the battery of behavioral tests with novel scoring system for phenotyping of transgenic rat model of tauopathy. J Neurosci Methods 177:108–114
Koson P, Zilka N, Kovac A et al (2008) Truncated tau expression levels determine life span of a rat model of tauopathy without causing neuronal loss or correlating with terminal neurofibrillary tangle load. Eur J Neurosci 28:239–246
Mitchell AJ (2009) CSF phosphorylated tau in the diagnosis and prognosis of mild cognitive impairment and Alzheimer’s disease: a meta-analysis of 51 studies. J Neurol Neurosurg Psychiatry 80:966–975
Montine TJ, Beal MF, Cudkowicz ME et al (1999) Increased CSF F2-isoprostane concentration in probable AD. Neurology 52:562–565
Pratico D, Clark CM, Lee VM et al (2000) Increased 8, 12-iso-iPF2alpha-VI in Alzheimer’s disease: correlation of a noninvasive index of lipid peroxidation with disease severity. Ann Neurol 48:809–812
Sunderland T, Wolozin B, Galasko D et al (1999) Longitudinal stability of CSF tau levels in Alzheimer patients. Biol Psychiatry 46:750–755
Vanmechelen E, Vanderstichele H, Davidsson P et al (2000) Quantification of tau phosphorylated at threonine 181 in human cerebrospinal fluid: a sandwich ELISA with a synthetic phosphopeptide for standardization. Neurosci Lett 285:49–52
Vemuri P, Wiste HJ, Weigand SD et al (2009) MRI and CSF biomarkers in normal, MCI, and AD subjects: diagnostic discrimination and cognitive correlations. Neurology 73:287–293
Wallin AK, Blennow K, Andreasen N et al (2006) CSF biomarkers for Alzheimer’s disease: levels of beta-amyloid, tau, phosphorylated tau relate to clinical symptoms and survival. Dement Geriatr Cogn Disord 21:131–138
Zetterberg H, Pedersen M, Lind K et al (2007) Intra-individual stability of CSF biomarkers for Alzheimer’s disease over two years. J Alzheimers Dis 12:255–260
Zilka N, Filipcik P, Koson P et al (2006) Truncated tau from sporadic Alzheimer’s disease suffices to drive neurofibrillary degeneration in vivo. FEBS Lett 580:3582–3588
Acknowledgments
This work was supported by Axon Neuroscience and research grants APVV-0631-07, APVV-0621-07, VEGA 2/0144/08, VEGA 2/0067/10, LPP-0354-06, LPP-0039-09, LPP-0363-06, LPP-0326-06 and NIH grant AG028538 (K.I.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zilka, N., Korenova, M., Kovacech, B. et al. CSF phospho-tau correlates with behavioural decline and brain insoluble phospho-tau levels in a rat model of tauopathy. Acta Neuropathol 119, 679–687 (2010). https://doi.org/10.1007/s00401-010-0680-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-010-0680-3