Abstract
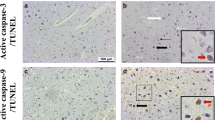
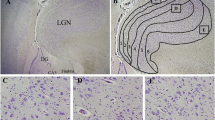
In a retrospective postmortem study, we examined the neuronal expression of active caspase-3, a specific apoptotic marker, in the brainstem of 67 infants dying from sudden infant death syndrome (SIDS), and 25 age-matched control infants (non-SIDS). Neuronal immunostaining for active caspase-3 was semi-quantitatively scored in nuclei from five brainstem levels: rostral, mid and caudal pons, and rostral and caudal medulla. Regardless of the cause of death (SIDS vs. non-SIDS), age-related differences in active caspase-3 expression were identified, predominantly in the medulla. No gender-related differences were identified. Comparing SIDS to non-SIDS cases, increased active caspase-3 expression was restricted to four nuclei in the caudal pons (abducens, facial, superior olivary, and pontine nuclei) and two nuclei in the rostral medulla (hypoglossal and dorsal motor nucleus of the vagus). We conclude that neuronal apoptosis is increased in the brainstem of SIDS compared to non-SIDS infants.

Similar content being viewed by others
References
Biondo B, Magagnin S, Bruni B, Cazzullo A, Tosi D, Matturri L (2004) Glial and neuronal alterations in the nucleus tractus solitarii of sudden infant death syndrome victims. Acta Neuropathol 108:309–318
Choi DW (1990) The role of glutamate neurotoxicity in hypoxic-ischemic neuronal death. Ann Rev Neurosci 13:171–182
David JC, Grongnet JF (2000) Effect of hypoxia on DNA fragmentation in different brain regions of the newborn piglet. Mol Reprod Dev 57:153–158
Hamill NJ, McGinn MD, Horowitz JM (1989) Auditory brainstem responses in ground squirrels arousing from hibernation. J Comp Physiol [B] 159:167–172
Harper RM, Leake B, Hoffman H, Walter DO, Hoppenbrouwers T, Hodgman J, Sterman MB (1981) Periodicity of sleep states is altered in infants at risk for the sudden infant death syndrome. Science 213:1030–1032
Horne RS, Parslow PM, Harding R (2005) Postnatal development of ventilatory and arousal responses to hypoxia in human infants. Respir Physiol Neurobiol 149:257–271
Joseph R (1999) Fetal brain and cognitive development. Dev Rev 20:81–98
Kinney HC, Burger PC, Harrell FE Jr, Hudson RP Jr (1983) ‘Reactive gliosis’ in the medulla oblongata of victims of the sudden infant death syndrome. Pediatrics 72:181–187
Kinney HC, Panigrahy A, Newburger JW, Jonas RA, Sleeper LA (2005) Hypoxic-ischemic brain injury in infants with congenital heart disease dying after cardiac surgery. Acta Neuropathol (Berl) 110:563–578
Konrat G, Halliday G, Sullivan C, Harper C (1992) Preliminary evidence suggesting delayed development in the hypoglossal and vagal nuclei of SIDS infants: a necropsy study. J Child Neurol 7:44–49
Kuida K, Zheng TS, Na S, Kuan C, Yang D, Karasuyama H, Rakic P, Flavell RA (1996) Decreased apoptosis in the brain and premature lethality in CPP32-deficient mice. Nature 384:368–372
Lamont P, Murray N, Halliday G, Hilton J, Pamphlett R (1995) Brain stem nuclei in sudden infant death syndrome (SIDS): volumes, neuronal numbers and positions. Neuropathol Appl Neurobiol 21:262–268
Lavezzi AM, Ottaviani G, Matturri L (2003) Identification of neurons responding to hypoxia in sudden infant death syndrome. Pathol Int 53:769–774
Loyola Stritch School of Medicine, http://www.meddean.luc.edu/lumen/MedEd/Neuro/frames/nlBSsL/nl40fr.htm
Machaalani R, Waters KA (2003) Increased neuronal cell death after intermittent hypercapnic hypoxia in the developing piglet brainstem. Brain Res 985:127–134
Machaalani R, Waters KA (2003) NMDA receptor 1 expression in the brainstem of human infants and its relevance to the sudden infant death syndrome (SIDS). J Neuropathol Exp Neurol 62:1076–1085
Machaalani R, Waters KA (2006) Postnatal nicotine and/or intermittent hypercapnic hypoxia effects on apoptotic markers in the developing piglet brainstem medulla. Neuroscience 142:107–117
Madhavan L, Freed WJ, Anantharam V, Kanthasamy AG (2003) 5-hydroxytryptamine 1A receptor activation protects against N-methyl-D-aspartate-induced apoptotic cell death in striatal and mesencephalic cultures. J Pharmacol Exp Ther 304:913–923
Mallard C, Tolcos M, Leditschke J, Campbell P, Rees S (1999) Reduction in choline acetyltransferase immunoreactivity but not muscarinic-m2 receptor immunoreactivity in the brainstem of SIDS infants. J Neuropathol Exp Neurol 58:255–264
Obonai T, Yasuhara M, Nakamura T, Takashima S (1998) Catecholamine neurons alteration in the brainstem of sudden infant death syndrome victims. Pediatrics 101:285–288
Oehmichen M, Theuerkauf I, Bajanowski T, Merz H, Meissner C (1998) Enhanced reactivity of Alz-50 antibody in brains of sudden infant death syndrome victims versus brains with lethal hypoxic/ischemic injury. Diagnostic significance after application of the ImmunoMax technique on routine paraffin material. Acta Neuropathol 95:280–286
Olszewski J, Baxter D (1954) Cytoarchitecture of the human brainstem. Karger, Switzerland
Ozawa Y, Okado N (2002) Alteration of serotonergic receptors in the brain stems of human patients with respiratory disorders. Neuropediatrics 33:142–149
Panigrahy A, Rosenberg PA, Assmann S, Foley EC, Kinney HC (2000) Differential expression of glutamate receptor subtypes in human brainstem sites involved in perinatal hypoxia-ischemia. J Comp Neurol 427:196–208
Paterson DS, Trachtenberg FL, Thompson EG, Belliveau RA, Beggs AH, Darnall R, Chadwick AE, Krous HF, Kinney HC (2006) Multiple serotonergic brainstem abnormalities in sudden infant death syndrome. Jama 296:2124–2132
Porzionato A, Macchi V, Morsut L, Parenti A, De Caro R (2005) Microvascular patterns in human medullary tegmentum at the level of the area postrema. J Anat 206:405–410
Quattrochi JJ, McBride PT, Yates AJ (1985) Brainstem immaturity in sudden infant death syndrome: a quantitative rapid Golgi study of dendritic spines in 95 infants. Brain Res 325:39–48
Rami A, Jansen S, Giesser I, Winckler J (2003) Post-ischemic activation of caspase-3 in the rat hippocampus: evidence of an axonal and dendritic localisation. Neurochem Int 43:211–223
Rickert CH, Zahiragic L, Nolte KW, Bajanowski T, Brinkmann B, Paulus W (2004) Cerebral c-jun expression mapping in sudden infant death syndrome. Acta Neuropathol 107:119–126
Rossiter JP, Anderson LL, Yang F, Cole GM (2002) Caspase-3 activation and caspase-like proteolytic activity in human perinatal hypoxic-ischemic brain injury. Acta Neuropathol 103:66–73
Sawaguchi T, Franco P, Kato I, Shimizu S, Kadhim H, Groswasser J, Sottiaux M, Togari H, Kobayashi M, Takashima S, Nishida H, Sawaguchi A, Kahn A (2002) From physiology to pathology: arousal deficiency theory in sudden infant death syndrome (SIDS)–with reference to apoptosis and neuronal plasticity. Forensic Sci Int 130(Suppl):S37–S43
Sherwood CC, Hof PR, Holloway RL, Semendeferi K, Gannon PJ, Frahm HD, Zilles K (2005) Evolution of the brainstem orofacial motor system in primates: a comparative study of trigeminal, facial, and hypoglossal nuclei. J Hum Evol 48:45–84
Siegel JM (1985) Ponto-medullary interactions in the generation of REM sleep. In: McGinty DJ, Drucker-Colin R, Morrison A, Parmeggiani PL (eds) Brain mechanisms of sleep. Raven Press, New York, pp 157–174
Sophou S, Dori I, Antonopoulos J, Parnavelas JG, Dinopoulos A (2006) Apoptosis in the rat basal forebrain during development and following lesions of connections. Eur J Neurosci 24:573–585
Sparks DL, Davis DG, Bigelow TM, Rasheed K, Landers TM, Liu H, Coyne CM, Hunsaker JCd (1996) Increased ALZ-50 immunoreactivity in sudden infant death syndrome. J Child Neurol 11:101–107
Srinivasan A, Roth KA, Sayers RO, Shindler KS, Wong AM, Fritz LC, Tomaselli KJ (1998) In situ immunodetection of activated caspase-3 in apoptotic neurons in the developing nervous system. Cell Death Differ 5:1004–1016
Stadelman C, Mews I, Srinivasan A, Deckwerth TL, Lassmann H, Bruck W (2001) Expression of cell death-associated proteins in neuronal apoptosis associated with pontosubicular neuron necrosis. Brain Pathol 11:273–281
Stennicke HR, Salvesen GS (1998) Properties of the caspases. Biochim Biophys Acta 1387:17–31
Thach BT (2005) The role of respiratory control disorders in SIDS. Respir Physiol Neurobiol 149:343–353
Waters KA, Gonzalez A, Jean C, Morielli A, Brouillette RT (1996) Face-straight-down and face-near-straight-down positions in healthy, prone-sleeping infants. J Pediatr 128:616–625
Waters KA, Meehan B, Huang JQ, Gravel RA, Michaud J, Cote A (1999) Neuronal apoptosis in sudden infant death syndrome. Pediatr Res 45:166–172
Willinger M, James LS, Catz C (1991) Defining the sudden infant death syndrome (SIDS): deliberations of an expert panel convened by the National Institute of Child Health and Human Development. Pediatr Pathol 11:677–684
Wong-Riley MT, Liu Q (2005) Neurochemical development of brain stem nuclei involved in the control of respiration. Respir Physiol Neurobiol 149:83–98
Yakovlev AG, Ota K, Wang G, Movsesyan V, Bao WL, Yoshihara K, Faden AI (2001) Differential expression of apoptotic protease-activating factor-1 and caspase-3 genes and susceptibility to apoptosis during brain development and after traumatic brain injury. J Neurosci 21:7439–7446
Yamanouchi H, Takashima S, Becker LE (1993) Correlation of astrogliosis and substance P immunoreactivity in the brainstem of victims of sudden infant death syndrome. Neuropediatrics 24:200–203
Acknowledgments
Research was funded by NH&MRC #302006, and The Sudden Infant Death Syndrome Foundation of South Australia. The authors would like to thank the staff at the Department of Forensic Medicine, Glebe, NSW, Australia for their co-operation in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Machaalani, R., Rodriguez, M. & Waters, K.A. Active caspase-3 in the sudden infant death syndrome (SIDS) brainstem. Acta Neuropathol 113, 577–584 (2007). https://doi.org/10.1007/s00401-007-0216-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-007-0216-7