Abstract
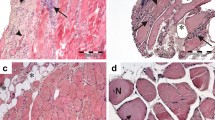
Myostatin is a negative regulator of muscle mass and strength. Sporadic inclusion-body myositis (s-IBM) is the most common degenerative muscle disease of older persons and is characterized by pronounced muscle wasting. s-IBM is of unknown etiology and pathogenesis, and it lacks definitive treatment. We have now demonstrated in samples from 12 s-IBM biopsies that: (1) by light and electron microscopic immunocytochemistry, myostatin/myostatin precursor is accumulated within muscle fibers and co-localized with amyloid-β (Aβ); (2) by immunoblots, both myostatin and myostatin precursor are increased; and (3) by immunoprecipitation, myostatin precursor complexes with Aβ. Our study suggests that myostatin/myostatin precursor, either alone, or bound to Aβ, may play a novel role in the pathogenesis of s-IBM.



Similar content being viewed by others
References
Askanas V, Engel WK (2003) Proposed pathogenetic cascade of inclusion-body myositis: importance of amyloid-beta, misfolded proteins, predisposing genes, and aging. Curr Opin Rheumatol 15:737–744
Askanas V, McFerrin J, Alvarez RB, Baque S, Engel WK (1997) Beta APP gene transfer into cultured human muscle induces inclusion-body myositis aspects. Neuroreport 8:2155–2158
Askanas V, Engel WK, Alvarez RB, McFerrin J, Broccolini A (2000) Novel immunolocalization of alpha-synuclein in human muscle of inclusion-body myositis, regenerating and necrotic muscle fibers, and at neuromuscular junctions. J Neuropathol Exp Neurol 59:592–598
Gonzalez-Cadavid NF, Bhasin S (2004) Role of myostatin in metabolism. Curr Opin Clin Nutr Metab Care 7:451–457
Gonzalez-Cadavid NF, Taylor WE, Yarasheski K, Sinha-Hikim I, Ma K, Ezzat S, Shen R, Lalani R, Asa S, Mamita M, Nair G, Arver S, Bhasin S (1998) Organization of the human myostatin gene and expression in healthy men and HIV-infected men with muscle wasting. Proc Natl Acad Sci USA 95:14938–14943
Jin HJ, Dunn MA, Borthakur D, Kim YS ( 2004) Refolding and purification of unprocessed porcine myostatin expressed in Escherichia coli. Protein Expr Purif 35:1–10
Kim KS, Wen GY, Bancher C, Chen J, Sapienza V, Hong H, Wisniewski HM (1990) Detection and quantitation of amyloid B-peptide with 2 monoclonal antibodies. Neurosci Res Commun 7:113–122
Liu W, Thomas SG, Asa SL, Gonzalez-Cadavid N, Bhasin S, Ezzat S (2003) Myostatin is a skeletal muscle target of growth hormone anabolic action. J Clin Endocrinol Metab 88:5490–5496
McFerrin J, Engel WK, Askanas V (1998) Impaired innervation of cultured human muscle overexpressing βAPP experimentally and genetically: relevance to inclusion-body myopathies. Neuroreport 9:3201–3205
McNally EM ( 2004) Powerful genes—myostatin regulation of human muscle mass. N Engl J Med 350:2642–2644
McPherron AC, Lee SJ (1997) Double muscling in cattle due to mutations in the myostatin gene. Proc Natl Acad Sci USA 94:12457–12461
Mirabella M, Alvarez RB, Bilak M, Engel WK, Askanas V (1996) Difference in expression of phosphorylated tau epitopes between sporadic inclusion-body myositis and hereditary inclusion-body myositis. J Neuropathol Exp Neurol 55:774–786
Reardon KA, Davis J, Kapsa RM, Choong P, Byrne E (2001) Myostatin, insulin-like growth factor-1, and leukemia inhibitory factor mRNAs are upregulated in chronic human disuse muscle atrophy. Muscle Nerve 24:893–899
Reisz-Porszasz S, Bhasin S, Artaza JN, Shen R, Sinha-Hikim I, Hogue A, Fielder TJ, Gonzalez-Cadavid NF (2003) Lower skeletal muscle mass in male transgenic mice with muscle-specific overexpression of myostatin. Am J Physiol Endocrinol Metab 285:876–888
Rios R, Fernandez-Nocelos S, Carneiro I, Arce VM, Devesa J (2004) Differential response to exogenous and endogenous myostatin in myoblasts suggests that myostatin acts as an autocrine factor in vivo. Endocrinology 145:2795–2803
Schuelke M, Wagner KR, Stolz LE, Hubner C, Riebel T, Komen W, Braun T, Tobin JF, Lee SJ (2004) Myostatin mutation associated with gross muscle hypertrophy in a child. N Engl J Med 350:2682–2688
Sharma M, Kambadur R, Matthews KG, Somers WG, Devlin GP, Conglen JV, Fowke PJ, Bass JJ (1999) Myostatin, transforming growth factor-β superfamily member, is expressed in heart muscle and is upregulated in cardiomyocytes after infarct. J Cell Physiol 180:1–9
Vattemi G, Engel WK, McFerrin J, Askanas V (2003) Cystatin C colocalizes with amyloid-beta and coimmunoprecipitates with amyloid-beta precursor protein in sporadic inclusion-body myositis muscles. J Neurochem 85:1539–1546
Vattemi G, Engel WK, McFerrin J, Askanas V (2004) Endoplasmic reticulum stress and unfolded protein response in inclusion body myositis muscle. Am J Pathol 164:1–7
Acknowledgments
This study was supported by grants (to V.A.) from the National Institutes of Health (AG16768 Merit Award), the Myositis Association, the Muscular Dystrophy Association, and the Helen Lewis Research Fund. S.W. is on leave from the Department of Anatomy and Neurobiology, Medical University of Gdansk, Gdansk, Poland. Maggie Baburyan provided excellent technical assistance in electron microscopy.
Conflict of interest:
No information supplied
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wójcik, S., Engel, W.K., McFerrin, J. et al. Myostatin is increased and complexes with amyloid-β within sporadic inclusion-body myositis muscle fibers. Acta Neuropathol 110, 173–177 (2005). https://doi.org/10.1007/s00401-005-1035-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-005-1035-3