Abstract
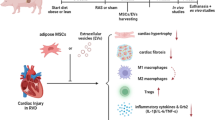
Extracellular vesicles (EVs) deliver genes and proteins to recipient cells, and mediate paracrine actions of their parent cells. Intrarenal delivery of mesenchymal stem cell (MSC)-derived EVs preserves stenotic-kidney function and reduces release of pro-inflammatory cytokines in a swine model of coexisting metabolic syndrome (MetS) and renal artery stenosis (RAS). We hypothesized that this approach is also capable of blunting cardiac injury and dysfunction. Five groups of pigs were studied after 16 weeks of diet-induced MetS and RAS (MetS + RAS), MetS and MetS + RAS treated 4 weeks earlier with a single intrarenal delivery of EVs-rich fraction harvested from autologous adipose tissue-derived MSCs, and lean and MetS Shams. Cardiac structure, function, and myocardial oxygenation were assessed in vivo using imaging, and cardiac inflammation, senescence, and fibrosis ex vivo. Inflammatory cytokine levels were measured in circulating and renal vein blood. Intrarenal EV delivery improved stenotic-kidney glomerular filtration rate and renal blood flow, and decreased renal release of monocyte-chemoattractant protein-1 and interleukin-6. Furthermore, despite unchanged systemic hemodynamics, intrarenal EV delivery in MetS + RAS normalized cardiac diastolic function, attenuated left ventricular remodeling, cellular senescence and inflammation, and improved myocardial oxygenation and capillary density in MetS + RAS. Intrarenal delivery of MSC-derived EVs blunts myocardial injury in experimental MetS + RAS, possibly related to improvement in renal function and systemic inflammatory profile. These observations underscore the central role of inflammation in the crosstalk between the kidney and heart, and the important contribution of renal function to cardiac structural and functional integrity in coexisting MetS and RAS.





Similar content being viewed by others
References
Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, Fruchart JC, James WP, Loria CM, Smith SC Jr, International Diabetes Federation Task Force on E, Prevention, National Heart L, Blood I, American Heart A, World Heart F, International Atherosclerosis S, International Association for the Study of O (2009) Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 120:1640–1645. https://doi.org/10.1161/CIRCULATIONAHA.109.192644
Bagno L, Hatzistergos KE, Balkan W, Hare JM (2018) Mesenchymal stem cell-based therapy for cardiovascular disease: progress and challenges. Mol Ther 26:1610–1623. https://doi.org/10.1016/j.ymthe.2018.05.009
Baranyai T, Herczeg K, Onodi Z, Voszka I, Modos K, Marton N, Nagy G, Mager I, Wood MJ, El Andaloussi S, Palinkas Z, Kumar V, Nagy P, Kittel A, Buzas EI, Ferdinandy P, Giricz Z (2015) Isolation of exosomes from blood plasma: qualitative and quantitative comparison of ultracentrifugation and size exclusion chromatography methods. PLoS ONE 10:e0145686. https://doi.org/10.1371/journal.pone.0145686
Chrysochou C, Kalra PA (2009) Epidemiology and natural history of atherosclerotic renovascular disease. Prog Cardiovasc Dis 52:184–195. https://doi.org/10.1016/j.pcad.2009.09.001
Davidson SM, Andreadou I, Barile L, Birnbaum Y, Cabrera-Fuentes HA, Cohen MV, Downey JM, Girao H, Pagliaro P, Penna C, Pernow J, Preissner KT, Ferdinandy P (2019) Circulating blood cells and extracellular vesicles in acute cardioprotection. Cardiovasc Res 115:1156–1166. https://doi.org/10.1093/cvr/cvy314
Davies MG, Saad WE, Bismuth J, Naoum JJ, Peden EK, Lumsden AB (2010) Impact of metabolic syndrome on the outcomes of percutaneous renal angioplasty and stenting. J Vasc Surg 51:926–932. https://doi.org/10.1016/j.jvs.2009.09.042
Eckel RH, Krauss RM (1998) American Heart Association call to action: obesity as a major risk factor for coronary heart disease. AHA Nutrition Committee. Circulation 97:2099–2100
Eirin A, Ebrahimi B, Zhang X, Zhu XY, Tang H, Crane JA, Lerman A, Textor SC, Lerman LO (2012) Changes in glomerular filtration rate after renal revascularization correlate with microvascular hemodynamics and inflammation in Swine renal artery stenosis. Circ Cardiovasc Interv 5:720–728. https://doi.org/10.1161/CIRCINTERVENTIONS.112.972596
Eirin A, Gloviczki ML, Tang H, Gossl M, Jordan KL, Woollard JR, Lerman A, Grande JP, Textor SC, Lerman LO (2013) Inflammatory and injury signals released from the post-stenotic human kidney. Eur Heart J 34:540–548a. https://doi.org/10.1093/eurheartj/ehs197
Eirin A, Williams BJ, Ebrahimi B, Zhang X, Crane JA, Lerman A, Textor SC, Lerman LO (2014) Mitochondrial targeted peptides attenuate residual myocardial damage after reversal of experimental renovascular hypertension. J Hypertens 32:154–165. https://doi.org/10.1097/HJH.0b013e3283658a53
Eirin A, Zhang X, Zhu XY, Tang H, Jordan KL, Grande JP, Dietz AB, Lerman A, Textor SC, Lerman LO (2014) Renal vein cytokine release as an index of renal parenchymal inflammation in chronic experimental renal artery stenosis. Nephrol Dial Transplant 29:274–282. https://doi.org/10.1093/ndt/gft305
Eirin A, Zhu XY, Ebrahimi B, Krier JD, Riester SM, van Wijnen AJ, Lerman A, Lerman LO (2015) Intrarenal delivery of mesenchymal stem cells and endothelial progenitor cells attenuates hypertensive cardiomyopathy in experimental renovascular hypertension. Cell Transplant 24:2041–2053. https://doi.org/10.3727/096368914X685582
Eirin A, Zhu XY, Ferguson CM, Riester SM, van Wijnen AJ, Lerman A, Lerman LO (2015) Intra-renal delivery of mesenchymal stem cells attenuates myocardial injury after reversal of hypertension in porcine renovascular disease. Stem Cell Res Ther 6:7. https://doi.org/10.1186/scrt541
Eirin A, Zhu XY, Jonnada S, Lerman A, van Wijnen AJ, Lerman LO (2018) Mesenchymal stem cell-derived extracellular vesicles improve the renal microvasculature in metabolic renovascular disease in swine. Cell Transplant 27:1080–1095. https://doi.org/10.1177/0963689718780942
Eirin A, Zhu XY, Krier JD, Tang H, Jordan KL, Grande JP, Lerman A, Textor SC, Lerman LO (2012) Adipose tissue-derived mesenchymal stem cells improve revascularization outcomes to restore renal function in swine atherosclerotic renal artery stenosis. Stem Cells 30:1030–1041. https://doi.org/10.1002/stem.1047
Eirin A, Zhu XY, Puranik AS, Tang H, McGurren KA, van Wijnen AJ, Lerman A, Lerman LO (2017) Mesenchymal stem cell-derived extracellular vesicles attenuate kidney inflammation. Kidney Int 92:114–124. https://doi.org/10.1016/j.kint.2016.12.023
Eirin A, Zhu XY, Puranik AS, Woollard JR, Tang H, Dasari S, Lerman A, van Wijnen AJ, Lerman LO (2017) Integrated transcriptomic and proteomic analysis of the molecular cargo of extracellular vesicles derived from porcine adipose tissue-derived mesenchymal stem cells. PLoS ONE 12:e0174303. https://doi.org/10.1371/journal.pone.0174303
Gansevoort RT, Correa-Rotter R, Hemmelgarn BR, Jafar TH, Heerspink HJ, Mann JF, Matsushita K, Wen CP (2013) Chronic kidney disease and cardiovascular risk: epidemiology, mechanisms, and prevention. Lancet 382:339–352. https://doi.org/10.1016/S0140-6736(13)60595-4
Haarer J, Johnson CL, Soeder Y, Dahlke MH (2015) Caveats of mesenchymal stem cell therapy in solid organ transplantation. Transpl Int 28:1–9. https://doi.org/10.1111/tri.12415
Halberg N, Khan T, Trujillo ME, Wernstedt-Asterholm I, Attie AD, Sherwani S, Wang ZV, Landskroner-Eiger S, Dineen S, Magalang UJ, Brekken RA, Scherer PE (2009) Hypoxia-inducible factor 1alpha induces fibrosis and insulin resistance in white adipose tissue. Mol Cell Biol 29:4467–4483. https://doi.org/10.1128/MCB.00192-09
Hayer MK, Price AM, Liu B, Baig S, Ferro CJ, Townend JN, Steeds RP, Edwards NC (2018) Diffuse myocardial interstitial fibrosis and dysfunction in early chronic kidney disease. Am J Cardiol 121:656–660. https://doi.org/10.1016/j.amjcard.2017.11.041
He J, Wang Y, Lu X, Zhu B, Pei X, Wu J, Zhao W (2015) Micro-vesicles derived from bone marrow stem cells protect the kidney both in vivo and in vitro by microRNA-dependent repairing. Nephrology (Carlton) 20:591–600. https://doi.org/10.1111/nep.12490
Higuchi Y, Otsu K, Nishida K, Hirotani S, Nakayama H, Yamaguchi O, Matsumura Y, Ueno H, Tada M, Hori M (2002) Involvement of reactive oxygen species-mediated NF-kappa B activation in TNF-alpha-induced cardiomyocyte hypertrophy. J Mol Cell Cardiol 34:233–240. https://doi.org/10.1006/jmcc.2001.1505
Jurk D, Wang C, Miwa S, Maddick M, Korolchuk V, Tsolou A, Gonos ES, Thrasivoulou C, Saffrey MJ, Cameron K, von Zglinicki T (2012) Postmitotic neurons develop a p21-dependent senescence-like phenotype driven by a DNA damage response. Aging Cell 11:996–1004. https://doi.org/10.1111/j.1474-9726.2012.00870.x
Katsiki N, Athyros VG, Karagiannis A, Mikhailidis DP (2014) Metabolic syndrome and non-cardiac vascular diseases: an update from human studies. Curr Pharm Des 20:4944–4952
Khangura KK, Eirin A, Kane GC, Misra S, Textor SC, Lerman A, Lerman LO (2014) Cardiac function in renovascular hypertensive patients with and without renal dysfunction. Am J Hypertens 27:445–453. https://doi.org/10.1093/ajh/hpt203
Kingma JG, Simard D, Rouleau JR, Drolet B, Simard C (2017) The physiopathology of cardiorenal syndrome: a review of the potential contributions of inflammation. J Cardiovasc Dev Dis. https://doi.org/10.3390/jcdd4040021
Kirkland JL, Tchkonia T (2017) Cellular senescence: a translational perspective. EBioMedicine 21:21–28. https://doi.org/10.1016/j.ebiom.2017.04.013
Kunter U, Rong S, Boor P, Eitner F, Muller-Newen G, Djuric Z, van Roeyen CR, Konieczny A, Ostendorf T, Villa L, Milovanceva-Popovska M, Kerjaschki D, Floege J (2007) Mesenchymal stem cells prevent progressive experimental renal failure but maldifferentiate into glomerular adipocytes. J Am Soc Nephrol 18:1754–1764. https://doi.org/10.1681/ASN.2007010044
Lawson C, Kovacs D, Finding E, Ulfelder E, Luis-Fuentes V (2017) Extracellular vesicles: evolutionarily conserved mediators of intercellular communication. Yale J Biol Med 90:481–491
Lerman LO, Schwartz RS, Grande JP, Sheedy PF, Romero JC (1999) Noninvasive evaluation of a novel swine model of renal artery stenosis. J Am Soc Nephrol 10:1455–1465
Lin J, Zhu X, Chade AR, Jordan KL, Lavi R, Daghini E, Gibson ME, Guglielmotti A, Lerman A, Lerman LO (2009) Monocyte chemoattractant proteins mediate myocardial microvascular dysfunction in swine renovascular hypertension. Arterioscler Thromb Vasc Biol 29:1810–1816. https://doi.org/10.1161/ATVBAHA.109.190546
Machado MV, Vieira AB, da Conceicao FG, Nascimento AR, da Nobrega ACL, Tibirica E (2017) Exercise training dose differentially alters muscle and heart capillary density and metabolic functions in an obese rat with metabolic syndrome. Exp Physiol 102:1716–1728. https://doi.org/10.1113/EP086416
Malaquin N, Martinez A, Rodier F (2016) Keeping the senescence secretome under control: molecular reins on the senescence-associated secretory phenotype. Exp Gerontol 82:39–49. https://doi.org/10.1016/j.exger.2016.05.010
Minamino T, Orimo M, Shimizu I, Kunieda T, Yokoyama M, Ito T, Nojima A, Nabetani A, Oike Y, Matsubara H, Ishikawa F, Komuro I (2009) A crucial role for adipose tissue p53 in the regulation of insulin resistance. Nat Med 15:1082–1087. https://doi.org/10.1038/nm.2014
Nascimento AR, Machado M, de Jesus N, Gomes F, Lessa MA, Bonomo IT, Tibirica E (2013) Structural and functional microvascular alterations in a rat model of metabolic syndrome induced by a high-fat diet. Obesity (Silver Spring) 21:2046–2054. https://doi.org/10.1002/oby.20358
Pawar AS, Zhu XY, Eirin A, Tang H, Jordan KL, Woollard JR, Lerman A, Lerman LO (2015) Adipose tissue remodeling in a novel domestic porcine model of diet-induced obesity. Obesity (Silver Spring) 23:399–407. https://doi.org/10.1002/oby.20971
Reinders ME, Leuning DG, de Fijter JW, Hoogduijn MJ, Rabelink TJ (2014) Mesenchymal stromal cell therapy for cardio renal disorders. Curr Pharm Des 20:2412–2429
Ronco C, Di Lullo L (2017) Cardiorenal syndrome in western countries: epidemiology, diagnosis and management approaches. Kidney Dis (Basel) 2:151–163. https://doi.org/10.1159/000448749
Saad A, Dietz AB, Herrmann SMS, Hickson LJ, Glockner JF, McKusick MA, Misra S, Bjarnason H, Armstrong AS, Gastineau DA, Lerman LO, Textor SC (2017) Autologous mesenchymal stem cells increase cortical perfusion in renovascular disease. J Am Soc Nephrol 28:2777–2785. https://doi.org/10.1681/ASN.2017020151
Saad A, Herrmann SM, Crane J, Glockner JF, McKusick MA, Misra S, Eirin A, Ebrahimi B, Lerman LO, Textor SC (2013) Stent revascularization restores cortical blood flow and reverses tissue hypoxia in atherosclerotic renal artery stenosis but fails to reverse inflammatory pathways or glomerular filtration rate. Circ Cardiovasc Interv 6:428–435. https://doi.org/10.1161/CIRCINTERVENTIONS.113.000219
Sluijter JPG, Davidson SM, Boulanger CM, Buzas EI, de Kleijn DPV, Engel FB, Giricz Z, Hausenloy DJ, Kishore R, Lecour S, Leor J, Madonna R, Perrino C, Prunier F, Sahoo S, Schiffelers RM, Schulz R, Van Laake LW, Ytrehus K, Ferdinandy P (2018) Extracellular vesicles in diagnostics and therapy of the ischaemic heart: position paper from the Working Group on Cellular Biology of the Heart of the European Society of Cardiology. Cardiovasc Res 114:19–34. https://doi.org/10.1093/cvr/cvx211
Smith GL, Masoudi FA, Shlipak MG, Krumholz HM, Parikh CR (2008) Renal impairment predicts long-term mortality risk after acute myocardial infarction. J Am Soc Nephrol 19:141–150. https://doi.org/10.1681/ASN.2007050554
Sorrentino JA, Sanoff HK, Sharpless NE (2014) Defining the toxicology of aging. Trends Mol Med 20:375–384. https://doi.org/10.1016/j.molmed.2014.04.004
Stenvinkel P, Luttropp K, McGuinness D, Witasp A, Qureshi AR, Wernerson A, Nordfors L, Schalling M, Ripsweden J, Wennberg L, Soderberg M, Barany P, Olauson H, Shiels PG (2017) CDKN2A/p16INK4(a) expression is associated with vascular progeria in chronic kidney disease. Aging (Albany NY) 9:494–507. https://doi.org/10.18632/aging.101173
Sun R, Zhu B, Xiong K, Sun Y, Shi D, Chen L, Zhang Y, Li Z, Xue L (2017) Senescence as a novel mechanism involved in beta-adrenergic receptor mediated cardiac hypertrophy. PLoS ONE 12:e0182668. https://doi.org/10.1371/journal.pone.0182668
Suthahar N, Meijers WC, Sillje HHW, de Boer RA (2017) From inflammation to fibrosis-molecular and cellular mechanisms of myocardial tissue remodelling and perspectives on differential treatment opportunities. Curr Heart Fail Rep 14:235–250. https://doi.org/10.1007/s11897-017-0343-y
Thomas G, Sehgal AR, Kashyap SR, Srinivas TR, Kirwan JP, Navaneethan SD (2011) Metabolic syndrome and kidney disease: a systematic review and meta-analysis. Clin J Am Soc Nephrol 6:2364–2373. https://doi.org/10.2215/CJN.02180311
Uchida Y, Ichimiya S, Ishii H, Kanashiro M, Watanabe J, Yoshikawa D, Takeshita K, Sakai S, Amano T, Matsubara T, Murohara T (2012) Impact of metabolic syndrome on various aspects of microcirculation and major adverse cardiac events in patients with ST-segment elevation myocardial infarction. Circ J 76:1972–1979
Urbieta-Caceres VH, Zhu XY, Jordan KL, Tang H, Textor K, Lerman A, Lerman LO (2012) Selective improvement in renal function preserved remote myocardial microvascular integrity and architecture in experimental renovascular disease. Atherosclerosis 221:350–358. https://doi.org/10.1016/j.atherosclerosis.2011.10.005
Wali RK, Wang GS, Gottlieb SS, Bellumkonda L, Hansalia R, Ramos E, Drachenberg C, Papadimitriou J, Brisco MA, Blahut S, Fink JC, Fisher ML, Bartlett ST, Weir MR (2005) Effect of kidney transplantation on left ventricular systolic dysfunction and congestive heart failure in patients with end-stage renal disease. J Am Coll Cardiol 45:1051–1060. https://doi.org/10.1016/j.jacc.2004.11.061
Welnicki MT, Sliz DI, Szeligowska J, Duda-Krol WB, Chomiuk T, Dabrowska D, Drozdz J, Mamcarz AJ (2018) The influence of metabolic syndrome coexistence on the prognosis of patients with heart failure without atrial fibrillation. Analysis of Polish data from the pilot survey for the ESC Heart Failure Registry. Kardiol Pol 76:794–796. https://doi.org/10.5603/KP.2018.0077
Yuan F, Hedayat AF, Ferguson CM, Lerman A, Lerman LO, Eirin A (2018) Mitoprotection attenuates myocardial vascular impairment in porcine metabolic syndrome. Am J Physiol Heart Circ Physiol 314:H669–H680. https://doi.org/10.1152/ajpheart.00431.2017
Zhao L, Cheng G, Jin R, Afzal MR, Samanta A, Xuan YT, Girgis M, Elias HK, Zhu Y, Davani A, Yang Y, Chen X, Ye S, Wang OL, Chen L, Hauptman J, Vincent RJ, Dawn B (2016) Deletion of interleukin-6 attenuates pressure overload-induced left ventricular hypertrophy and dysfunction. Circ Res 118:1918–1929. https://doi.org/10.1161/CIRCRESAHA.116.308688
Zhu XY, Daghini E, Chade AR, Napoli C, Ritman EL, Lerman A, Lerman LO (2007) Simvastatin prevents coronary microvascular remodeling in renovascular hypertensive pigs. J Am Soc Nephrol 18:1209–1217. https://doi.org/10.1681/ASN.2006090976
Zhu XY, Ebrahimi B, Eirin A, Woollard JR, Tang H, Jordan KL, Ofori M, Saad A, Herrmann SM, Dietz AB, Textor SC, Lerman A, Lerman LO (2015) Renal vein levels of microRNA-26a are lower in the poststenotic kidney. J Am Soc Nephrol 26:1378–1388. https://doi.org/10.1681/ASN.2014030248
Zhu XY, Urbieta-Caceres V, Krier JD, Textor SC, Lerman A, Lerman LO (2013) Mesenchymal stem cells and endothelial progenitor cells decrease renal injury in experimental swine renal artery stenosis through different mechanisms. Stem Cells 31:117–125. https://doi.org/10.1002/stem.1263
Funding
This study was partly supported by NIH Grant Numbers: DK120292, DK104273, HL123160, and DK102325, and DK106427.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Dr. Lerman receives grant funding from Novo Nordisk, and is an advisor to Weijian Technologies and AstraZeneca. All the other authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhang, L., Zhu, XY., Zhao, Y. et al. Selective intrarenal delivery of mesenchymal stem cell-derived extracellular vesicles attenuates myocardial injury in experimental metabolic renovascular disease. Basic Res Cardiol 115, 16 (2020). https://doi.org/10.1007/s00395-019-0772-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-019-0772-8