Abstract
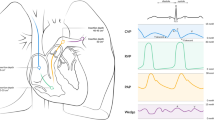
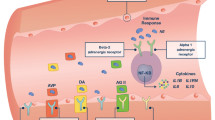
Vasopressors are widely used in resuscitation, ventricular failure, and sepsis, and often induce pulmonary hypertension with undefined mechanisms. We hypothesize that vasopressor-induced pulmonary hypertension is caused by increased pulmonary blood volume and tested this hypothesis in dogs under general anesthesia. In normal hearts (model 1), phenylephrine (2.5 μg/kg/min) transiently increased right but decreased left cardiac output, associated with increased pulmonary blood volume (63% ± 11.8, P = 0.007) and pressures in the left atrium, pulmonary capillary, and pulmonary artery. However, the trans-pulmonary gradient and pulmonary vascular resistance remained stable. These changes were absent after decreasing blood volume or during right cardiac dysfunction to reduce pulmonary blood volume (model 2). During double-ventricle bypass (model 3), phenylephrine (1, 2.5 and 10 μg/kg/min) only slightly induced pulmonary vasoconstriction. Vasopressin (1U and 2U) dose-dependently increased pulmonary artery pressure (52 ± 8.4 and 71 ± 10.3%), but did not cause pulmonary vasoconstriction in normally beating hearts (model 1). Pulmonary artery and left atrial pressures increased during left ventricle dysfunction (model 4), and further increased after phenylephrine injection by 31 ± 5.6 and 43 ± 7.5%, respectively. In conclusion, vasopressors increased blood volume in the lung with minimal pulmonary vasoconstriction. Thus, this pulmonary hypertension is similar to the hemodynamic pattern observed in left heart diseases and is passive, due to redistribution of blood from systemic to pulmonary circulation. Understanding the underlying mechanisms may improve clinical management of patients who are taking vasopressors, especially those with coexisting heart disease.





Similar content being viewed by others
References
Aung K, Htay T (2005) Vasopressin for cardiac arrest: a systematic review and meta-analysis. Arch Intern Med 165:17–24. doi:10.1001/archinte.165.1.17
Brittain EL, Doss LN, Saliba L, Irani W, Byrd BF 3rd, Monahan K (2015) Feasibility and diagnostic potential of pulmonary transit time measurement by contrast echocardiography: a pilot study. Echocardiography 32:1564–1571. doi:10.1111/echo.12906
Caldini P, Permutt S, Waddell JA, Riley RL (1974) Effect of epinephrine on pressure, flow, and volume relationships in the systemic circulation of dogs. Circ Res 34:606–623
Cevasco M, Kwon M, Fiedler A, Lee LS, Shiao J, Shah RV, Worthington AH, Fox JA, Kwong RY, Chen FY (2014) Right heart failure: an ischemic model and restraint therapy for treatment. Ann Thorac Surg 97:1356–1362. doi:10.1016/j.athoracsur.2013.09.112 (discussion 1362-1363)
Crosara S, Ljungvall I, Margiocco ML, Haggstrom J, Tarducci A, Borgarelli M (2012) Use of contrast echocardiography for quantitative and qualitative evaluation of myocardial perfusion and pulmonary transit time in healthy dogs. Am J Vet Res 73:194–201. doi:10.2460/ajvr.73.2.194
Currigan DA, Hughes RJ, Wright CE, Angus JA, Soeding PF (2014) Vasoconstrictor responses to vasopressor agents in human pulmonary and radial arteries: an in vitro study. Anesthesiology 121:930–936. doi:10.1097/ALN.0000000000000430
D’Alto M, Romeo E, Argiento P, Correra A, Santoro G, Gaio G, Sarubbi B, Calabro R, Russo MG (2013) Hemodynamics of patients developing pulmonary arterial hypertension after shunt closure. Int J Cardiol 168:3797–3801. doi:10.1016/j.ijcard.2013.06.036
Eichinger MR, Walker BR (1994) Enhanced pulmonary arterial dilation to arginine vasopressin in chronically hypoxic rats. Am J Physiol 267:H2413–H2419
Evora PR, Pearson PJ, Schaff HV (1993) Arginine vasopressin induces endothelium-dependent vasodilatation of the pulmonary artery. V1-receptor-mediated production of nitric oxide. Chest 103:1241–1245
Garcia-Alvarez A, Pereda D, Garcia-Lunar I, Sanz-Rosa D, Fernandez-Jimenez R, Garcia-Prieto J, Nuno-Ayala M, Sierra F, Santiago E, Sandoval E, Campelos P, Aguero J, Pizarro G, Peinado VI, Fernandez-Friera L, Garcia-Ruiz JM, Barbera JA, Castella M, Sabate M, Fuster V, Ibanez B (2016) Beta-3 adrenergic agonists reduce pulmonary vascular resistance and improve right ventricular performance in a porcine model of chronic pulmonary hypertension. Basic Res Cardiol 111:49. doi:10.1007/s00395-016-0567-0
Guazzi M, Borlaug BA (2012) Pulmonary hypertension due to left heart disease. Circulation 126:975–990. doi:10.1161/CIRCULATIONAHA.111.085761
Haraldsen P, Metzsch C, Lindstedt S, Algotsson L, Ingemansson R (2016) Sevoflurane anesthesia during acute right ventricular ischemia in pigs preserves cardiac function better than propofol anesthesia. Perfusion. doi:10.1177/0267659116637134
Herold IH, Saporito S, Bouwman RA, Houthuizen P, van Assen HC, Mischi M, Korsten HH (2016) Reliability, repeatability, and reproducibility of pulmonary transit time assessment by contrast enhanced echocardiography. Cardiovasc Ultrasound 14:1. doi:10.1186/s12947-015-0044-1
Hochman JS, Buller CE, Sleeper LA, Boland J, Dzavik V, Sanborn TA, Godfrey E, White HD, Lim J, LeJemtel T (2000) Cardiogenic shock complicating acute myocardial infarction–etiologies, management and outcome: a report from the SHOCK Trial Registry. SHould we emergently revascularize Occluded Coronaries for cardiogenic shocK? J Am Coll Cardiol 36:1063–1070
Hoeper MM, Bogaard HJ, Condliffe R, Frantz R, Khanna D, Kurzyna M, Langleben D, Manes A, Satoh T, Torres F, Wilkins MR, Badesch DB (2013) Definitions and diagnosis of pulmonary hypertension. J Am Coll Cardiol 62:D42–D50. doi:10.1016/j.jacc.2013.10.032
Hussain MB, Marshall I (1997) Characterization of alpha1-adrenoceptor subtypes mediating contractions to phenylephrine in rat thoracic aorta, mesenteric artery and pulmonary artery. Br J Pharmacol 122:849–858. doi:10.1038/sj.bjp.0701461
Jin HK, Chen YF, Yang RH, McKenna TM, Jackson RM, Oparil S (1989) Vasopressin lowers pulmonary artery pressure in hypoxic rats by releasing atrial natriuretic peptide. Am J Med Sci 298:227–236
Kwak YL, Lee CS, Park YH, Hong YW (2002) The effect of phenylephrine and norepinephrine in patients with chronic pulmonary hypertension. Anaesthesia 57:9–14
Lam CS, Rienstra M, Tay WT, Liu LC, Hummel YM, van der Meer P, de Boer RA, Van Gelder IC, van Veldhuisen DJ, Voors AA, Hoendermis ES (2016) Atrial fibrillation in heart failure with preserved ejection fraction: association with exercise capacity, left ventricular filling pressures, natriuretic peptides, and left atrial volume. JACC Heart Fail. doi:10.1016/j.jchf.2016.10.005
Leather HA, Segers P, Berends N, Vandermeersch E, Wouters PF (2002) Effects of vasopressin on right ventricular function in an experimental model of acute pulmonary hypertension. Crit Care Med 30:2548–2552. doi:10.1097/01.CCM.0000034696.32358.54
Lopes A, Alnajashi K (2014) Saudi guidelines on the diagnosis and treatment of pulmonary hypertension: pulmonary arterial hypertension associated with congenital heart disease. Ann Thorac Med 9:S21–S25. doi:10.4103/1817-1737.134015
Maseri A, Caldini P, Harward P, Joshi RC, Permutt S, Zierler KL (1972) Determinants of pulmonary vascular volume: recruitment versus distensibility. Circ Res 31:218–228
Mathieu M, El Oumeiri B, Touihri K, Hadad I, Mahmoudabady M, Thoma P, Metens T, Bartunek J, Heyndrickx GR, Brimioulle S, Naeije R, Mc Entee K (2010) Ventricular-arterial uncoupling in heart failure with preserved ejection fraction after myocardial infarction in dogs—invasive versus echocardiographic evaluation. BMC Cardiovasc Disord 10:32. doi:10.1186/1471-2261-10-32
Moreira-Goncalves D, Ferreira R, Fonseca H, Padrao AI, Moreno N, Silva AF, Vasques-Novoa F, Goncalves N, Vieira S, Santos M, Amado F, Duarte JA, Leite-Moreira AF, Henriques-Coelho T (2015) Cardioprotective effects of early and late aerobic exercise training in experimental pulmonary arterial hypertension. Basic Res Cardiol 110:57. doi:10.1007/s00395-015-0514-5
Morimatsu H, Ishikawa K, May CN, Bailey M, Bellomo R (2012) The systemic and regional hemodynamic effects of phenylephrine in sheep under normal conditions and during early hyperdynamic sepsis. Anesth Analg 115:330–342. doi:10.1213/ANE.0b013e31825681ab
Ng GA, Cobbe SM, Smith GL (1998) Non-uniform prolongation of intracellular Ca2+ transients recorded from the epicardial surface of isolated hearts from rabbits with heart failure. Cardiovasc Res 37:489–502
Price LC, Wort SJ, Finney SJ, Marino PS, Brett SJ (2010) Pulmonary vascular and right ventricular dysfunction in adult critical care: current and emerging options for management: a systematic literature review. Crit Care 14:R169. doi:10.1186/cc9264
Pye MP, Black M, Cobbe SM (1996) Comparison of in vivo and in vitro haemodynamic function in experimental heart failure: use of echocardiography. Cardiovasc Res 31:873–881
Rieg AD, Rossaint R, Uhlig S, Martin C (2011) Cardiovascular agents affect the tone of pulmonary arteries and veins in precision-cut lung slices. PLoS One 6:e29698. doi:10.1371/journal.pone.0029698
Russell JA, Walley KR, Singer J, Gordon AC, Hebert PC, Cooper DJ, Holmes CL, Mehta S, Granton JT, Storms MM, Cook DJ, Presneill JJ, Ayers D, Investigators V (2008) Vasopressin versus norepinephrine infusion in patients with septic shock. N Engl J Med 358:877–887. doi:10.1056/NEJMoa067373
Sai Y, Okamura T, Amakata Y, Toda N (1995) Comparison of responses of canine pulmonary artery and vein to angiotensin II, bradykinin and vasopressin. Eur J Pharmacol 282:235–241
Sarkar J, Golden PJ, Kajiura LN, Murata LA, Uyehara CF (2015) Vasopressin decreases pulmonary-to-systemic vascular resistance ratio in a porcine model of severe hemorrhagic shock. Shock 43:475–482. doi:10.1097/SHK.0000000000000325
Scolletta S, Franchi F, Romagnoli S, Carla R, Donati A, Fabbri LP, Forfori F, Alonso-Inigo JM, Laviola S, Mangani V, Maj G, Martinelli G, Mirabella L, Morelli A, Persona P, Payen D, Pulse wave analysis Cardiac Output validation Group (2016) Comparison between Doppler-echocardiography and uncalibrated pulse contour method for cardiac output measurement: a multicenter observational study. Crit Care Med 44:1370–1379. doi:10.1097/CCM.0000000000001663
Shebuski RJ, Fujita T, Ruffolo RR Jr (1986) Evaluation of alpha-1 and alpha-2 adrenoceptor-mediated vasoconstriction in the in situ, autoperfused, pulmonary circulation of the anesthetized dog. J Pharmacol Exp Ther 238:217–223
Simonneau G, Gatzoulis MA, Adatia I, Celermajer D, Denton C, Ghofrani A, Gomez Sanchez MA, Krishna Kumar R, Landzberg M, Machado RF, Olschewski H, Robbins IM, Souza R (2013) Updated clinical classification of pulmonary hypertension. J Am Coll Cardiol 62:D34–D41. doi:10.1016/j.jacc.2013.10.029
Sondergaard L, Reddy V, Kaye D, Malek F, Walton A, Mates M, Franzen O, Neuzil P, Ihlemann N, Gustafsson F (2014) Transcatheter treatment of heart failure with preserved or mildly reduced ejection fraction using a novel interatrial implant to lower left atrial pressure. Eur J Heart Fail 16:796–801. doi:10.1002/ejhf.111
Stene JK, Burns B, Permutt S, Caldini P, Shanoff M (1982) Increased cardiac output following occlusion of the descending thoracic aorta in dogs. Am J Physiol 243:R152–R158
Werner F, Kojonazarov B, Gassner B, Abesser M, Schuh K, Volker K, Baba HA, Dahal BK, Schermuly RT, Kuhn M (2016) Endothelial actions of atrial natriuretic peptide prevent pulmonary hypertension in mice. Basic Res Cardiol 111:22. doi:10.1007/s00395-016-0541-x
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
This work was supported by the National Natural Science Foundation of China (81600006, 81270324 and 81570374) and Sichuan Provincial Scientific Grant (2017SZ0110).
Rights and permissions
About this article
Cite this article
Jiang, C., Qian, H., Luo, S. et al. Vasopressors induce passive pulmonary hypertension by blood redistribution from systemic to pulmonary circulation. Basic Res Cardiol 112, 21 (2017). https://doi.org/10.1007/s00395-017-0611-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-017-0611-8