Abstract
Background and aims
Patients with metastatic colorectal cancer have a limited life expectancy and are at risk for life-threatening tumor-related obstruction, perforation, and hemorrhage. Though surgical resection is performed frequently in this setting, its true benefit is not well-established.
Materials and methods
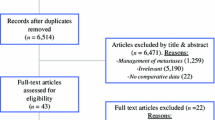
We reviewed the medical literature from 1996–2006 using the search terms metastatic colorectal cancer and primary resection to find studies that evaluated the management of primary tumors in metastatic colorectal cancer. All search results were included in our analysis and were assessed on the basis of methodologic quality.
Results/findings
Twelve relevant studies were identified; ten were single-institution retrospective reviews and two were population-based studies using National Cancer Institute’s Surveillance, Epidemiology, and End-Results database. No prospective or randomized studies were identified. Approximately 70% of patients diagnosed with metastatic colorectal cancer in the USA undergo primary tumor resection; only a minority have this done for tumor-related symptoms or as part of potentially curative resection. The postoperative mortality ranged from 9.0–11.2% in large cancer registries but was often lower in major cancer centers. Resection of asymptomatic primary tumors was frequently associated with prolonged survival but was not found to reduce significantly the incidence of life-threatening tumor-related complications.
Interpretation/conclusion
Retrospective data suggest that non-curative resection of asymptomatic colorectal primary tumors may prolong survival; however, selection bias and unaccounted clinical factors may explain this observation. Prospective, randomized surgical trials are needed to test the role of primary tumor resection in this setting, especially because of its current widespread use, and its associated cost, morbidity, and high postoperative mortality.
Similar content being viewed by others
References
ACS (2007) http://www.cancer.org
SEER (2007) http://seer.cancer.gov
Adam R, Avisar E, Ariche A, Giachetti S, Azoulay D, Castaing D, Kunstlinger F, Levi F, Bismuth F (2001) Five-year survival following hepatic resection after neoadjuvant therapy for nonresectable colorectal. Ann Surg Oncol 8:347–353
Ballantyne GH, Quin J (1993) Surgical treatment of liver metastases in patients with colorectal cancer. Cancer 71:4252–4266
Poston GJ, Adam R, Alberts S, Curley S, Figueras J, Haller D, Kunstlinger F, Mentha G, Nordlinger B, Patt Y, Primrose J, Roh M, Rougier P, Ruers T, Schmoll HJ, Valls C, Vauthey NJ, Cornelis M, Kahan JP (2005) OncoSurge: a strategy for improving resectability with curative intent in metastatic colorectal cancer. J Clin Oncol 23:7125–7134
Sjovall A, Jarv V, Blomqvist L, Singnomklao T, Cedermark B, Glimelius B, Holm T (2004) The potential for improved outcome in patients with hepatic metastases from colon cancer: a population-based study. Eur J Surg Oncol 30:834–841
de Gramont A, Figer A, Seymour M, Homerin M, Hmissi A, Cassidy J, Boni C, Cortes-Funes H, Cervantes A, Freyer G, Papamichael D, Le Bail N, Louvet C, Hendler D, de Braud F, Wilson C, Morvan F, Bonetti A (2000) Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol 18:2938–2947
Douillard JY, Cunningham D, Roth AD, Navarro M, James RD, Karasek P, Jandik P, Iveson T, Carmichael J, Alakl M, Gruia G, Awad L, Rougier P (2000) Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet 355:1041–1047
Kohne CH, van Cutsem E, Wils J, Bokemeyer C, El-Serafi M, Lutz MP, Lorenz M, Reichardt P, Ruckle-Lanz H, Frickhofen N, Fuchs R, Mergenthaler HG, Langenbuch T, Vanhoefer U, Rougier P, Voigtmann R, Muller L, Genicot B, Anak O, Nordlinger B (2005) Phase III study of weekly high-dose infusional fluorouracil plus folinic acid with or without irinotecan in patients with metastatic colorectal cancer: European organisation for research and treatment of cancer gastrointestinal group study 40986. J Clin Oncol 23:4856–4865
American Society of Clinical Oncology (ASCO, 2007) http://www.asco.org/portal/site/ASCO/menuitem.34d60f5624ba07fd506fe310ee37a01d/?vgnextoid=76f8201eb61a7010VgnVCM100000ed730ad1RCRD&vmview=abst_detail_view&confID=40&abstractID=33907
Giantonio B, Levy D, O’Dwyer P, Meropol N, Catalano P, Benson A 3rd (2006) A phase II study of high-dose bevacizumab in combination with irinotecan, 5-fluorouracil, leucovorin, as initial therapy for advanced colorectal cancer: results from the eastern cooperative oncology group study E2200. Ann Oncol 17:1399–1403
Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, Berlin J, Baron A, Griffing S, Holmgren E, Ferrara N, Fyfe G, Rogers B, Ross R, Kabbinavar F (2004) Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med 350:2335–2342
Bacon HE, Martin PV (1964) The rationale of palliative resection for primary cancer of the colon and rectum complicated by liver and lung metastasis. Dis Colon Rectum 36:211–217
Joffe J, Gordon PH (1981) Palliative resection for colorectal carcinoma. Dis Colon Rectum 24:355–360
Longo WE, Ballantyne GH, Bilchik AJ, Modlin IM (1988) Advanced rectal cancer. What is the best palliation? Dis Colon Rectum 31:842–847
Makela J, Haukipuro K, Laitinen S, Kairaluoma MI (1990) Palliative operations for colorectal cancer. Dis Colon Rectum 33:846–850
Moran MR, Rothenberger DA, Lahr CJ, Buls JG, Goldberg SM (1987) Palliation for rectal cancer. Resection? Anastomosis? Arch Surg 122:640–643
Oxley EM, Ellis H (1969) Prognosis of carcinoma of the large bowel in the presence of liver metastases. Br J Surg 56:149–152
Nordic Gastrointestinal Tumor Adjuvant Therapy Group (1992) Expectancy or primary chemotherapy in patients with advanced asymptomatic colorectal cancer: a randomized trial. J Clin Oncol 10:904–911
Allendorf JD, Bessler M, Kayton ML, Oesterling SD, Treat MR, Nowygrod R, Whelan RL (1995) Increased tumor establishment and growth after laparotomy vs laparoscopy in a murine model. Arch Surg 130:649–653
Carter JJ, Feingold DL, Kirman I, Oh A, Wildbrett P, Asi Z, Fowler R, Huang E, Whelan RL (2003) Laparoscopic-assisted cecectomy is associated with decreased formation of postoperative pulmonary metastases compared with open cecectomy in a murine model. Surgery 134:432–436
Da Costa ML, Redmond HP, Finnegan N, Flynn M, Bouchier-Hayes D (1998) Laparotomy and laparoscopy differentially accelerate experimental flank tumour growth. Br J Surg 85:1439–1442
Cook AD, Single R, McCahill LE (2005) Surgical resection of primary tumors in patients who present with stage IV colorectal cancer: an analysis of surveillance, epidemiology, and end results data, 1988 to 2000. Ann Surg Oncol 12:637–645
Temple LK, Hsieh L, Wong WD, Saltz L, Schrag D (2004) Use of surgery among elderly patients with stage IV colorectal cancer. J Clin Oncol 22:3475–3484
Law WL, Chu KW (2006) Outcomes of resection of stage IV rectal cancer with mesorectal excision. J Surg Oncol 93:523–528
Nash GM, Saltz LB, Kemeny NE, Minsky B, Sharma S, Schwartz GK, Ilson DH, O’Reilly E, Kelsen DP, Nathanson DR, Weiser M, Guillem JG, Wong WD, Cohen AM, Paty PB (2002) Radical resection of rectal cancer primary tumor provides effective local therapy in patients with stage IV disease. Ann Surg Oncol 9:954–960
Rosen SA, Buell JF, Yoshida A, Kazsuba S, Hurst R, Michelassi F, Millis JM, Posner MC (2000) Initial presentation with stage IV colorectal cancer: how aggressive should we be? Arch Surg 135:530–534 (discussion 534–535)
Yamamura T, Tsukikawa S, Akaishi O, Tanaka K, Matsuoka H, Hanai A, Oikawa H, Ozasa T, Kikuchi K, Matsuzaki H, Yamaguchi S (1997) Multivariate analysis of the prognostic factors of patients with unresectable synchronous liver metastases from colorectal cancer. Dis Colon Rectum 40:1425–1429
Sarela AI, Guthrie JA, Seymour MT, Ride E, Guillou PJ, O’Riordain DS (2001) Non-operative management of the primary tumour in patients with incurable stage IV colorectal cancer. Br J Surg 88:1352–1356
Liu SK, Church JM, Lavery IC, Fazio VW (1997) Operation in patients with incurable colon cancer—is it worthwhile? Dis Colon Rectum 40:11–14
Ruo L, Gougoutas C, Paty PB, Guillem JG, Cohen AM, Wong WD (2003) Elective bowel resection for incurable stage IV colorectal cancer: prognostic variables for asymptomatic patients. J Am Coll Surg 196:722–728
Scoggins CR, Meszoely IM, Blanke CD, Beauchamp RD, Leach SD (1999) Nonoperative management of primary colorectal cancer in patients with stage IV disease. Ann Surg Oncol 6:651–657
Stelzner S, Hellmich G, Koch R, Ludwig K (2005) Factors predicting survival in stage IV colorectal carcinoma patients after palliative treatment: a multivariate analysis. J Surg Oncol 89:211–217
Tebbutt NC, Norman AR, Cunningham D, Hill ME, Tait D, Oates J, Livingston S, Andreyev J (2003) Intestinal complications after chemotherapy for patients with unresected primary colorectal cancer and synchronous metastases. Gut 52:568–573
Alves A, Panis Y, Mathieu P, Mantion G, Kwiatkowski F, Slim K (2005) Postoperative mortality and morbidity in French patients undergoing colorectal surgery: results of a prospective multicenter study. Arch Surg 140:278–283 (discussion 284)
Mella J, Biffin A, Radcliffe AG, Stamatakis JD, Steele RJ (1997) Population-based audit of colorectal cancer management in two UK health regions. Colorectal Cancer Working Group, Royal College of Surgeons of England Clinical Epidemiology and Audit Unit. Br J Surg 84:1731–1736
Riedl S, Wiebelt H, Bergmann U, Hermanek P Jr (1995) Postoperative complications and fatalities in surgical therapy of colon carcinoma. Results of the German multicenter study by the colorectal carcinoma study group. Chirurgia 66:597–606
Wyrzykowski AD, Feliciano DV, George TA, Tremblay LN, Rozycki GS, Murphy TW, Dente CJ (2005) Emergent right hemicolectomies. Am Surg 71:653–656 (discussion 656–657)
Abdalla EK, Vauthey JN, Ellis LM, Ellis V, Pollock R, Broglio KR, Hess K, Curley SA (2004) Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann Surg 239:818–825 (discussion 825–817)
Curley SA (2005) Outcomes after surgical treatment of colorectal cancer liver metastases. Semin Oncol 32:S109–S111
Glehen O, Osinsky D, Beaujard AC, Gilly FN (2003) Natural history of peritoneal carcinomatosis from nongynecologic malignancies. Surg Oncol Clin N Am 12:729–739 xiii
Geller DA, Tsung A, Marsh JW, Dvorchik I, Gamblin TC, Carr BI (2006) Outcome of 1000 liver cancer patients evaluated at the UPMC Liver Cancer Center. J Gastrointest Surg 10:63–68
Wong YN, Mitra N, Hudes G, Localio R, Schwartz JS, Wan F, Montagnet C, Armstrong K (2006) Survival associated with treatment vs observation of localized prostate cancer in elderly men. JAMA 296:2683–2693
Alberts SR, Horvath WL, Sternfeld WC, Goldberg RM, Mahoney MR, Dakhil SR, Levitt R, Rowland K, Nair S, Sargent DJ, Donohue JH (2005) Oxaliplatin, fluorouracil, and leucovorin for patients with unresectable liver-only metastases from colorectal cancer: a North Central Cancer Treatment Group phase II study. J Clin Oncol 23:9243–9249
Acknowledgment
Dr. Neugut is the recipient of a grant from the American Cancer Society (RSGT-01–024–04-CPHPS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eisenberger, A., Whelan, R.L. & Neugut, A.I. Survival and symptomatic benefit from palliative primary tumor resection in patients with metastatic colorectal cancer: a review. Int J Colorectal Dis 23, 559–568 (2008). https://doi.org/10.1007/s00384-008-0456-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-008-0456-6