Abstract
Purpose
The co-occurrence of Hirschsprung’s disease (HSCR) and multiple endocrine neoplasia type 2 (MEN2) is a relatively rare event. The basis for this association is the presence of a “Janus” mutation in the RET proto-oncogene––a mutation that acts simultaneously as both a gain-in-function and a loss-of-function mutation. To date, four mutations in the exon 10 region of RET that are known to cause MEN2A have been implicated in this association: C620, C618, C611 and C609. We performed a systematic review of the published literature on this association to determine its incidence, the prevalence and phenotype of HSCR associated with the 4 RET mutations mentioned above.
Methods
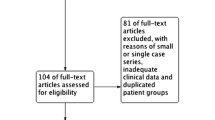
A systematic literature-based search for relevant articles was conducted using three online databases. After exclusion of ineligible publications, we recorded data on all patients with a diagnosis of HSCR or MEN2A with a “Janus” RET mutation, as well as those who carried the mutation but were unaffected. Statistical analysis was performed using SPSS.
Results
The literature search yielded 885 publications, of which 36 articles, incorporating data on 341 individuals, were eligible for inclusion in the final analysis. Co-occurrence of HSCR and MEN2A was recorded in 84 cases (24.6 %). HSCR occurred alone in 64 carriers of a “Janus” mutation (18.8 %) and MEN2A occurred in isolation in 173 cases (50.7 %). Twenty individuals (5.9 %) were found to carry a “Janus” mutation after screening on the basis of family history but were unaffected by either MEN2A or HSCR. The most common mutation recorded was the C620 mutation [114 cases (48.1 %)]. There was a relatively high incidence of long-segment aganglionosis (29.3 %) and total colonic aganglionosis (17.3 %) in this cohort. This trend was particularly notable in those with C620 mutations, only 33 % of whom had short-segment disease.
Conclusion
While the overall incidence of HSCR co-occurring with MEN2A is low, both conditions occur with a relatively high frequency in families with a RET mutation at exon 10. The proportion of cases of long-segment HSCR and total colonic aganglionosis is higher than that in the general population with HSCR in those with C620 and C618 mutations. These findings re-inforce the importance of RET mutation testing in HSCR when a family history of either HSCR or MEN2 is present. In families with MEN2A and known exon 10 RET mutations, the threshold for investigation for HSCR in those with gastrointestinal symptoms should be very low. High-quality prospective longitudinal studies of large HSCR populations are required to shed greater light on this rare but important phenomenon.


Similar content being viewed by others
References
Sipple JH (1961) The association of pheochromocytoma with carcinoma of the thyroid gland. Am J Med 31:163–166
Williams ED, Pollock DJ (1966) Multiple mucosal neuromata with endocrine tumours: a syndrome allied to von Recklinghausen’s disease. J Pathol Bacteriol 91(1):71–80. doi:10.1002/path.1700910109
Verdy M, Weber AM, Roy CC, Morin CL, Cadotte M, Brochu P (1982) Hirschsprung’s disease in a family with multiple endocrine neoplasia type 2. J Pediatr Gastroenterol Nutr 1(4):603–607
Godbole K (2004) Many faces of Hirschsprung’s disease. Indian Pediatr 41(11):1115–1123
Pasini B, Rossi R, Ambrosio MR, Zatelli MC, Gullo M, Gobbo M, Collini P, Aiello A, Pansini G, Trasforini G, degli Uberti EC (2002) RET mutation profile and variable clinical manifestations in a family with multiple endocrine neoplasia type 2A and Hirschsprung’s disease. Surgery 131(4):373–381
Arighi E, Popsueva A, Degl’Innocenti D, Borrello MG, Carniti C, Perala NM, Pierotti MA, Sariola H (2004) Biological effects of the dual phenotypic Janus mutation of ret cosegregating with both multiple endocrine neoplasia type 2 and Hirschsprung’s disease. Mol Endocrinol 18(4):1004–1017. doi:10.1210/me.2003-0173
Kenny SE, Tam PK, Garcia-Barcelo M (2010) Hirschsprung’s disease. Semin Pediatr Surg 19(3):194–200. doi:10.1053/j.sempedsurg.2010.03.004
Emison ES, Garcia-Barcelo M, Grice EA, Lantieri F, Amiel J, Burzynski G, Fernandez RM, Hao L, Kashuk C, West K, Miao X, Tam PK, Griseri P, Ceccherini I, Pelet A, Jannot AS, de Pontual L, Henrion-Caude A, Lyonnet S, Verheij JB, Hofstra RM, Antinolo G, Borrego S, McCallion AS, Chakravarti A (2010) Differential contributions of rare and common, coding and noncoding ret mutations to multifactorial Hirschsprung disease liability. Am J Hum Genet 87(1):60–74. doi:10.1016/j.ajhg.2010.06.007
Pandey R, Thurow T, de WMR (2011) Hirschsprung disease of the colon, a vaginal mass and medullary thyroid cancer––a RET oncogene driven problem. J Gastrointest Oncol 2(4):254–257. doi:10.3978/j.issn.2078-6891.2011.028
Quedas EP, Longuini VC, Sekiya T, Coutinho FL, Toledo SP, Tannuri U, Toledo RA (2012) RET haplotype, not linked to the C620R activating mutation, associated with Hirschsprung disease in a novel MEN2 family. Clinics 67(Suppl 1):57–61
Moore SW, Zaahl M (2010) Familial associations in medullary thyroid carcinoma with Hirschsprung disease: the role of the RET-C620 “Janus” genetic variation. J Pediatr Surg 45(2):393–396. doi:10.1016/j.jpedsurg.2009.10.080
Moore SW, Zaahl M (2009) Clinical and genetic differences in total colonic aganglionosis in Hirschsprung’s disease. J Pediatr Surg 44(10):1899–1903. doi:10.1016/j.jpedsurg.2009.04.026
Vaclavikova E, Dvorakova S, Sykorova V, Bilek R, Dvorakova K, Vlcek P, Skaba R, Zelinka T, Bendlova B (2009) RET mutation Tyr791Phe: the genetic cause of different diseases derived from neural crest. Endocrine 36(3):419–424. doi:10.1007/s12020-009-9242-7
Fialkowski EA, DeBenedetti MK, Moley JF, Bachrach B (2008) RET proto-oncogene testing in infants presenting with Hirschsprung disease identifies 2 new multiple endocrine neoplasia 2A kindreds. J Pediatr Surg 43(1):188–190. doi:10.1016/j.jpedsurg.2007.09.043
Butter A, Gagne J, Al-Jazaeri A, Emran MA, Deal C, St-Vil D (2007) Prophylactic thyroidectomy in pediatric carriers of multiple endocrine neoplasia type 2A or familial medullary thyroid carcinoma: mutation in C620 is associated with Hirschsprung’s disease. J Pediatr Surg 42(1):203–206. doi:10.1016/j.jpedsurg.2006.09.019
Dvorakova S, Dvorakova K, Malikova M, Skaba R, Vlcek P, Bendlova B (2005) A novel Czech kindred with familial medullary thyroid carcinoma and Hirschsprung’s disease. J Pediatr Surg 40(6):e1–e6. doi:10.1016/j.jpedsurg.2005.03.031
Fitze G, Schreiber M, Hohenberger W, Hummer HP, Roesner D, Schackert HK (2004) Interaction of RET proto-oncogene codon 609 germline mutations with RET haplotypes characterized by c.135G>A alleles modifying MEN 2A or HSCR phenotypes. Am J Med Genet Part A 129A(3):323–325. doi:10.1002/ajmg.a.30230
Nishikawa M, Murakumo Y, Imai T, Kawai K, Nagaya M, Funahashi H, Nakao A, Takahashi M (2003) Cys611Ser mutation in RET proto-oncogene in a kindred with medullary thyroid carcinoma and Hirschsprung’s disease. Eur J Hum Genet 11(5):364–368. doi:10.1038/sj.ejhg.5200971
Cohen, Phay JE, Albinson C, DeBenedetti MK, Skinner MA, Lairmore TC, Doherty GM, Balfe DM, Wells SA Jr, Moley JF (2002) Gastrointestinal manifestations of multiple endocrine neoplasia type 2. Ann Surgery 235(5):648–654 (discussion 654–645)
Sasaki Y, Shimotake T, Go S, Iwai N (2001) Total thyroidectomy for hereditary medullary thyroid carcinoma 12 years after correction of Hirschsprung’s disease. Eur J Surg 167(6):467–469. doi:10.1080/110241501750243851
Lore F, Talidis F, Di Cairano G, Renieri A (2001) Multiple endocrine neoplasia type 2 syndromes may be associated with renal malformations. J Inter Med 250(1):37–42
Inoue K, Shimotake T, Inoue K, Tokiwa K, Iwai N (1999) Mutational analysis of the RET proto-oncogene in a kindred with multiple endocrine neoplasia type 2A and Hirschsprung’s disease. J Pediatr Surg 34(10):1552–1554
Sijmons RH, Hofstra RM, Wijburg FA, Links TP, Zwierstra RP, Vermey A, Aronson DC, Tan-Sindhunata G, Brouwers-Smalbraak GJ, Maas SM, Buys CH (1998) Oncological implications of RET gene mutations in Hirschsprung’s disease. Gut 43(4):542–547
Borrego S, Eng C, Sanchez B, Saez ME, Navarro E, Antinolo G (1998) Molecular analysis of the ret and GDNF genes in a family with multiple endocrine neoplasia type 2A and Hirschsprung disease. J Clin Endocrinol Metab 83(9):3361–3364. doi:10.1210/jcem.83.9.5093
Romeo G, Ceccherini I, Celli J, Priolo M, Betsos N, Bonardi G, Seri M, Yin L, Lerone M, Jasonni V, Martucciello G (1998) Association of multiple endocrine neoplasia type 2 and Hirschsprung disease. J Intern Med 243(6):515–520
Decker RA, Peacock ML, Watson P (1998) Hirschsprung disease in MEN 2A: increased spectrum of RET exon 10 genotypes and strong genotype-phenotype correlation. Hum Mol Genet 7(1):129–134
Peretz H, Luboshitsky R, Baron E, Biton A, Gershoni R, Usher S, Grynberg E, Yakobson E, Graff E, Lapidot M (1997) Cys 618 Arg mutation in the RET proto-oncogene associated with familial medullary thyroid carcinoma and maternally transmitted Hirschsprung’s disease suggesting a role for imprinting. Hum Mutat 10(2):155–159. doi:10.1002/(SICI)1098-1004(1997)10:2<155:AID-HUMU7>3.0.CO;2-J
Blank RD, Sklar CA, Dimich AB, LaQuaglia MP, Brennan MF (1996) Clinical presentations and RET protooncogene mutations in seven multiple endocrine neoplasia type 2 kindreds. Cancer 78(9):1996–2003
Caron P, Attie T, David D, Amiel J, Brousset F, Roger P, Munnich A, Lyonnet S (1996) C618R mutation in exon 10 of the RET proto-oncogene in a kindred with multiple endocrine neoplasia type 2A and Hirschsprung’s disease. J Clin Endocrinol Metab 81(7):2731–2733. doi:10.1210/jcem.81.7.8675603
Borst MJ, VanCamp JM, Peacock ML, Decker RA (1995) Mutational analysis of multiple endocrine neoplasia type 2A associated with Hirschsprung’s disease. Surgery 117(4):386–391
Mulligan LM, Eng C, Attie T, Lyonnet S, Marsh DJ, Hyland VJ, Robinson BG, Frilling A, Verellen-Dumoulin C, Safar A et al (1994) Diverse phenotypes associated with exon 10 mutations of the RET proto-oncogene. Hum Mol Genet 3(12):2163–2167
Rakover Y, Dharan M, Luboshitsky R (1994) Hirschsprung’s disease associated with isolated familial medullary carcinoma of the thyroid. J Pediatr Endocrinol 7(4):373–377
Hubner A, Holschneider AM (1987) Multiple endocrine neoplasias in 3 generations. Langenbecks Arch Chir 372:747–750
Le Marec B, Roussey M, Le Clech G, Kerisit J, Allanic H (1983) Thyroid cancer with amyloid stroma, sipple syndrome, congenital megacolon with plexus hyperplasia: one and the same autosomal dominant disease with complete penetrance. J Genet Hum 31((1):11–12
Olivares Munoz M, Julia Masip MV, Oriola J, Martorell Sampol L, Parareda Salles A, Ribo Cruz JM (2012) Hirschsprung’s disease and medullary carcinoma of the thyroids: two diseases in a monogenetic disorder. Cir Pediatr 25(2):87–90
Oliva-Rodriguez R, Guerrero-VazquezR, Del Castillo Tous M, Martinez-Brocca MA, Martos-Martinez JM, Leal-Cerro A, Navarro-Gonzalez E (2011) Descriptive study of the clinical behavior of multiple endocrine neoplasia 2A. Endocrine reviews. https://www.endocrine.org/~/media/endosociety/Files/Meetings/Abstracts/2011/02TES2011.pdf
Lallier M, St-Vil D, Giroux M, Huot C, Gaboury L, Oligny L, Desjardins JG (1998) Prophylactic thyroidectomy for medullary thyroid carcinoma in gene carriers of MEN2 syndrome. J Paediat Surg 33(6):846–848
Frilling A, Becker H, Roeher HD (1992) Unusual features of multiple endocrine neoplasia. Henry Ford Hosp Med J 40(3–4):253–255
Moore SW, Appfelstaedt J, Zaahl MG (2007) Familial medullary carcinoma prevention, risk evaluation, and RET in children of families with MEN2. J Pediatr Surg 42(2):326–332. doi:10.1016/j.jpedsurg.2006.10.005
Virtanen VB, Pukkala E, Kivisaari R, Salo PP, Koivusalo A, Arola J, Miettinen PJ, Rintala RJ, Perola M, Pakarinen MP (2013) Thyroid cancer and co-occurring RET mutations in Hirschsprung disease. Endocr Relat Cancer 20(4):595–602. doi:10.1530/ERC-13-0082
Decker RA, Peacock ML (1998) Occurrence of MEN 2a in familial Hirschsprung’s disease: a new indication for genetic testing of the RET proto-oncogene. J Pediatr Surg 33(2):207–214
Angrist M, Bolk S, Thiel B, Puffenberger EG, Hofstra RM, Buys CH, Cass DT, Chakravarti A (1995) Mutation analysis of the RET receptor tyrosine kinase in Hirschsprung disease. Hum Mol Genet 4(5):821–830
Decker RA (1992) Long-term follow-up of a large North American kindred with multiple endocrine neoplasia type 2A. Surg 112(6):1066–1072 (Discussion 1072–1063)
Hofstra RM, Wu Y, Stulp RP, Elfferich P, Osinga J, Maas SM, Siderius L, Brooks AS, vd Ende JJ, Heydendael VM, Severijnen RS, Bax KM, Meijers C, Buys CH (2000) RET and GDNF gene scanning in Hirschsprung patients using two dual denaturing gel systems. Hum Mutat 15(5):418–429. doi:10.1002/(SICI)1098-1004(200005)15:5<418:AID-HUMU3>3.0.CO;2-2
Eng C, Clayton D, Schuffenecker I, Lenoir G, Cote G, Gagel RF, van Amstel HK, Lips CJ, Nishisho I, Takai SI, Marsh DJ, Robinson BG, Frank-Raue K, Raue F, Xue F, Noll WW, Romei C, Pacini F, Fink M, Niederle B, Zedenius J, Nordenskjold M, Komminoth P, Hendy GN, Mulligan LM et al (1996) The relationship between specific RET proto-oncogene mutations and disease phenotype in multiple endocrine neoplasia type 2. International RET mutation consortium analysis. JAMA 276(19):1575–1579
Carniti C, Belluco S, Riccardi E, Cranston AN, Mondellini P, Ponder BA, Scanziani E, Pierotti MA, Bongarzone I (2006) The Ret(C620R) mutation affects renal and enteric development in a mouse model of Hirschsprung’s disease. Am J Pathol 168(4):1262–1275. doi:10.2353/ajpath.2006.050607
Edery P, Pelet A, Mulligan LM, Abel L, Attie T, Dow E, Bonneau D, David A, Flintoff W, Jan D et al (1994) Long segment and short segment familial Hirschsprung’s disease: variable clinical expression at the RET locus. J Med Genet 31(8):602–606
Badner JA, Sieber WK, Garver KL, Chakravarti A (1990) A genetic study of Hirschsprung disease. Am J Hum Genet 46(3):568–580
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Coyle, D., Friedmacher, F. & Puri, P. The association between Hirschsprung’s disease and multiple endocrine neoplasia type 2a: a systematic review. Pediatr Surg Int 30, 751–756 (2014). https://doi.org/10.1007/s00383-014-3538-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-014-3538-2