Abstract
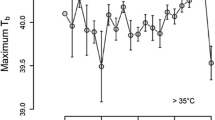
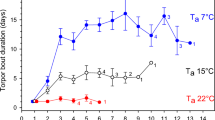
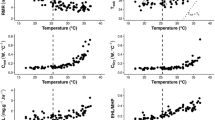
Hibernation is the most effective way to reduce thermoregulatory costs during periods of unfavourable environmental conditions. In preparation to hibernation, fat-storing hibernators accumulate large quantities of body fat, which increases their locomotor costs and also the risk of predation. As a consequence, there should be a strong selective pressure to restrict pre-hibernation fattening to a short-time period before the onset of hibernation. The edible dormouse (Glis glis) is characterized by having adapted its whole life history to the irregularly occurring mast-seeding pattern of the European beech (Fagus sylvaticus). Thus, the question arises how this small endotherm copes with huge differences in food availability between years. Therefore, we investigated body mass and thermal energetics of edible dormice during high and low food years. Our results demonstrate that during periods of low food availability, edible dormice enter an energy-saving mode with reduced body temperature (Tb) and resting metabolic rate (RMR), and high torpor frequencies. During irregularly occurring short events of high food availability in mast years, however, Tb was higher, torpor did not occur, and RMR was drastically elevated possibly due to an enlarged digestive tract and the heat increment of feeding associated with a dietary switch to high-quality food and an increase in the amount of food ingested. This physiological flexibility allows edible dormice to efficiently accumulate body fat reserves under extremely different situations of food availability.





Similar content being viewed by others
References
Barnes BM (1989) Freeze Avoidance in a mammal: body temperatures below 0 °C in an Arctic Hibernator. Science 244:1593–1595
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using {lme4}. J Stat Softw 67:1–48
Bieber C (1998) Population dynamics, sexual activity, and reproduction failure in the fat dormouse (Myoxus glis). J Zool 244:223–229. https://doi.org/10.1111/j.1469-7998.1998.tb00027.x
Bieber C, Ruf T (2009) Summer dormancy in edible dormice (Glis glis) without energetic constraints. Naturwissenschaften 96:165–171. https://doi.org/10.1007/s00114-008-0471-z
Bieber C, Außerlechner K, Skerget C et al (2011) Seasonal changes in liver size in edible dormice (Glis glis): non-invasive measurements using ultrasonography. Eur J Wildl Res 57:657–662. https://doi.org/10.1007/s10344-010-0476-8
Bieber C, Lebl K, Stalder G et al (2014) Body mass dependent use of hibernation: why not prolong the active season, if they can? Funct Ecol 28:167–177. https://doi.org/10.1111/1365-2435.12173
Bieber C, Cornils JS, Hoelzl F et al (2017) The costs of locomotor activity? Maximum body temperatures and the use of torpor during the active season in edible dormice. J Comp Physiol B. https://doi.org/10.1007/s00360-017-1080-y
Bryant AA, Page RE (2005) Timing and causes of mortality in the endangered Vancouver Island marmot (Marmota vancouverensis). Can J Zool 83:674–682. https://doi.org/10.1139/z05-055
Cant JP, McBride BW, Croom WJ (1996) The regulation of intestinal metabolism and its impact on whole animal energetics. J Anim Sci 74:2541. https://doi.org/10.2527/1996.74102541x
Carey HV (2005) Gastrointestinal responses to fasting in mammals: lessons from hibernators. Physiol Ecol Adapt Feed Vertebr Sci Enfield NH 229–254
Carey HV, Andrews MT, Martin SL (2003) Mammalian hibernation: cellular and molecular responses to depressed metabolism and low temperature. Physiol Rev 83:1153–1181. https://doi.org/10.1152/physrev.00008.2003
Clarke A, Fraser KPP (2004) Why does metabolism scale with temperature? Funct Ecol 18:243–251. https://doi.org/10.1111/j.0269-8463.2004.00841.x
Cornils JS, Hoelzl F, Rotter B et al (2017) Edible dormice (Glis glis) avoid areas with a high density of their preferred food plant—the European beech. Front Zool 14:23. https://doi.org/10.1186/s12983-017-0206-0
Cygan T (1985) Seasonal changes in thermoregulation and maximum metabolism in the yellow-necked field mouse. Acta Theriol (Warsz) 30:115–130. https://doi.org/10.4098/AT.arch.85-4
Dausmann KH (2014) Flexible patterns in energy savings: heterothermy in primates. J Zool 292:101–111. https://doi.org/10.1111/jzo.12104
Fietz J, Schlund W, Dausmann KH et al (2004) Energetic constraints on sexual activity in the male edible dormouse (Glis glis). Oecologia 138:202–209. https://doi.org/10.1007/s00442-003-1423-0
Fietz J, Pflug M, Schlund W, Tataruch F (2005) Influences of the feeding ecology on body mass and possible implications for reproduction in the edible dormouse (Glis glis). J Comp Physiol [B] 175:45–55. https://doi.org/10.1007/s00360-004-0461-1
Fietz J, Kager T, Schauer S (2009) Is energy supply the trigger for reproductive activity in male edible dormice (Glis glis)? J Comp Physiol B 179:829–837. https://doi.org/10.1007/s00360-009-0364-2
Fietz J, Klose SM, Kalko EKV (2010) Behavioural and physiological consequences of male reproductive trade-offs in edible dormice (Glis glis). Naturwissenschaften 97:883–890. https://doi.org/10.1007/s00114-010-0704-9
Fietz J, Tomiuk J, Loeschcke V et al (2014) Genetic consequences of forest fragmentation for a highly specialized arboreal mammal—the edible dormouse. PLoS One 9:e88092. https://doi.org/10.1371/journal.pone.0088092
Fox J, Weisberg S (2011) An R companion to applied regression, 2nd edn. Sage, Thousand Oaks
Geiser F, Kenagy GJ (1987) Polyunsaturated lipid diet lengthens torpor and reduces body temperature in a hibernator. Am J Physiol Regul Integr Comp Physiol 252:R897–R901
Geiser F, Hiebert S, Kenagy GJ (1990) Torpor bout duration during the hibernation season of two sciurid rodents: interrelations with temperature and metabolism. Physiol Zool 63:489–503. https://doi.org/10.1086/physzool.63.3.30156224
Havenstein N, Langer F, Fietz J (2017) Life history written in blood: erythrocyte parameters in a small hibernator, the edible dormouse. J Comp Physiol B 1–13. https://doi.org/10.1007/s00360-017-1111-8
Heldmaier G, Elvert R (2004) How to enter torpor: thermodynamic and physiological mechanisms of metabolic depression. Life in the cold: evolution, mechanisms, adaptation, and application 12th international hibernation symposium, pp 185–198
Heldmaier G, Neuweiler G (2013) Vergleichende Tierphysiologie: Gerhard Heldmaier Vegetative Physiologie. Springer, Berlin
Hilton GM, Packham JR (2003) Variation in the masting of common beech (Fagus sylvatica L.) in northern Europe over two centuries (1800–2001). For Int J For Res 76:319–328. https://doi.org/10.1093/forestry/76.3.319
Hoelzl F, Bieber C, Cornils JS et al (2015) How to spend the summer? Free-living dormice (Glis glis) can hibernate for 11 months in non-reproductive years. J Comp Physiol B 185:931–939. https://doi.org/10.1007/s00360-015-0929-1
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biomed J 50:346–363. https://doi.org/10.1002/bimj.200810425
Hume I, Beiglböck C, Ruf T et al (2002) Seasonal changes in morphology and function of the gastrointestinal tract of free-living alpine marmots (Marmota marmota). J Comp Physiol [B] 172:197–207. https://doi.org/10.1007/s00360-001-0240-1
Humphries MM, Thomas DW, Kramer DL (2003) The role of energy availability in mammalian hibernation: a cost-benefit approach. Physiol Biochem Zool 76:165–179. https://doi.org/10.1086/367950
Ims RA, Andreassen HP (2000) Spatial synchronization of vole population dynamics by predatory birds. Nature 408:194–196. https://doi.org/10.1038/35041562
Johns DW, Armitage KB (1979) Behavioral ecology of alpine yellow-bellied marmots. Behav Ecol Sociobiol 5:133–157. https://doi.org/10.1007/BF00293302
Johnston SL, Souter DM, Erwin SS et al (2007) Associations between basal metabolic rate and reproductive performance in C57BL/6J mice. J Exp Biol 210:65–74. https://doi.org/10.1242/jeb.02625
Juškaitis R, Baltrūnaitė L, Augutė V (2015) Diet of the fat dormouse (Glis glis) on the northern periphery of its distributional range. Mammal Res 60:155–161. https://doi.org/10.1007/s13364-015-0213-5
Kager T, Fietz J (2009) Food availability in spring influences reproductive output in the seed-preying edible dormouse (Glis glis). Can J Zool 87:555–565. https://doi.org/10.1139/Z09-040
Kleiber M (1947) Body size and metabolic rate. Physiol Rev 27:511–541
Konarzewski M, Diamond J (1995) Evolution of basal metabolic rate and organ masses in laboratory mice. Evolution 49:1239–1248. https://doi.org/10.2307/2410448
Konarzewski M, Książek A (2013) Determinants of intra-specific variation in basal metabolic rate. J Comp Physiol [B] 183:27–41. https://doi.org/10.1007/s00360-012-0698-z
Körtner G, Geiser F (1995) Body temperature rhythms and activity in reproductive Antechinus (Marsupialia). Physiol Behav 58:31–36. https://doi.org/10.1016/0031-9384(94)00384-H
Krebs HA (1950) Body size and tissue respiration. Biochim Biophys Acta 4:249–269. https://doi.org/10.1016/0006-3002(50)90032-1
Kronfeld-Schor N, Richardson C, Silvia BA et al (2000) Dissociation of leptin secretion and adiposity during prehibernatory fattening in little brown bats. Am J Physiol Regul Integr Comp Physiol 279:R1277–R1281
Kryštufek B (2010) Glis glis (Rodentia: Gliridae). Mamm Species 42:195–206. https://doi.org/10.1644/865.1
Kuznetsova A, Brockhoff PB, Bojesen RH (2015) lmerTest: tests in linear mixed effects models. R package version 2.0–29
Langer F, Fietz J (2014) Ways to measure body temperature in the field. J Therm Biol 42:46–51. https://doi.org/10.1016/j.jtherbio.2014.03.002
Lebl K, Kürbisch K, Bieber C, Ruf T (2010) Energy or information? The role of seed availability for reproductive decisions in edible dormice. J Comp Physiol B 180:447–456. https://doi.org/10.1007/s00360-009-0425-6
Lebl K, Bieber C, Adamík P et al (2011) Survival rates in a small hibernator, the edible dormouse: a comparison across Europe. Ecography 34:683–692. https://doi.org/10.1111/j.1600-0587.2010.06691.x
Lighton JRB (2008) Measuring metabolic rates: a manual for scientists: a manual for scientists. Oxford University Press, USA
Long RA, Martin TJ, Barnes BM (2005) Body temperature and activity patterns in free-living arctic ground squirrels. J Mammal 86:314–322. https://doi.org/10.1644/BRG-224.1
Lovegrove BG (2000) The zoogeography of mammalian basal metabolic rate. Am Nat 156:201–219. https://doi.org/10.1086/303383
Lyman CP (2013) Hibernation and torpor in mammals and birds. Elsevier, Oxford
Maynard LA, Loosli JK (1969) Animal nutrition. McGraw-Hill, New York
Morris PA (1968) Apparent hypothermia in the wood mouse (Apodemus sylvaticus). J Zool 155:235–236. https://doi.org/10.1111/j.1469-7998.1968.tb03044.x
Pilastro A, Tavecchia G, Marin G (2003) Long living and reproduction skipping in the fat dormouse. Ecology 84:1784–1792. https://doi.org/10.1890/0012-9658(2003)084%5B1784:LLARSI%5D2.0.CO;2
Pinheiro J, Bates D, DebRoy S, R Development Core Team (2011) nlme: linear and nonlinear mixed effects models
R Core Team (2011) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rubner M (1902) Die Gesetze des Energieverbrauchs bei der Ernährung. F. Deuticke, Leipzig
Ruf T, Arnold W (2008) Effects of polyunsaturated fatty acids on hibernation and torpor: a review and hypothesis. Am J Physiol Regul Integr Comp Physiol 294:R1044–R1052. https://doi.org/10.1152/ajpregu.00688.2007
Ruf T, Geiser F (2015) Daily torpor and hibernation in birds and mammals. Biol Rev 90:891–926. https://doi.org/10.1111/brv.12137
Ruf T, Heldmaier G (1992) The impact of daily torpor on energy requirements in the djungarian hamster, Phodopus sungorus. Physiol Zool 65:994–1010
Ruf T, Fietz J, Schlund W, Bieber C (2006) High survival in poor years: life history tactics adapted to mast seeding in the edible dormouse. Ecology 87:372–381. https://doi.org/10.1890/05-0672
Sailer MM, Fietz J (2009) Seasonal differences in the feeding ecology and behavior of male edible dormice (Glis glis). Mamm Biol Z Für Säugetierkd 74:114–124. https://doi.org/10.1016/j.mambio.2008.05.005
Schlund W (1997) Die Tibialänge als Maß für Körpergröße und als Hilfsmittel zur Altersbestimmung bei Siebenschläfern (Myoxus glis L.). Mamm Biol Z Für Säugetierkd:187–190
Schlund W, Scharfe F, Stauss MJ, Burkhardt JE (1997) Habitat fidelity and habitat utilization of an arboreal mammal (Myoxus glis) in two different forests. Mamm Biol Z Für Säugetierkd:158–171
Schlund W, Scharfe F, Ganzhorn JU (2002) Long-term comparison of food availability and reproduction in the edible dormouse (Glis glis). Mamm Biol Z Für Säugetierkd 67:219–232. https://doi.org/10.1078/1616-5047-00033
Sheriff MJ, Fridinger RW, Tøien Ø et al (2013) Metabolic rate and prehibernation fattening in free-living arctic ground squirrels. Physiol Biochem Zool 86:515–527. https://doi.org/10.1086/673092
Smit B, Boyles JG, Brigham RM, McKechnie AE (2011) Torpor in dark times: patterns of heterothermy are associated with the lunar cycle in a nocturnal bird. J Biol Rhythms 26:241–248. https://doi.org/10.1177/0748730411402632
Song X, Körtner G, Geiser F (1997) Thermal relations of metabolic rate reduction in a hibernating marsupial. Am J Physiol Regul Integr Comp Physiol 273:R2097–R2104
Sork VL, Bramble J, Sexton O (1993) Ecology of mast-fruiting in three species of North American deciduous oaks. Ecology 74:528–541. https://doi.org/10.2307/1939313
Stawski C, Körtner G, Nowack J, Geiser F (2015) The importance of mammalian torpor for survival in a post-fire landscape. Biol Lett 11:20150134. https://doi.org/10.1098/rsbl.2015.0134
Trombulak SC (1989) Running speed and body mass in belding’s ground squirrels. J Mammal 70:194–197. https://doi.org/10.2307/1381688
Turbill C, Bieber C, Ruf T (2011) Hibernation is associated with increased survival and the evolution of slow life histories among mammals. Proc R Soc B 278:3355–3363. https://doi.org/10.1098/rspb.2011.0190
von Vietinghoff - Riesch A (1960) Der Siebenschlaefer (Glis glis L.). Fischer, Jena
Walton JB, Andrews JF (1981) Torpor induced by food deprivation in the Wood mouse Apodemus sylvaticus. J Zool 194:260–263. https://doi.org/10.1111/j.1469-7998.1981.tb05773.x
Wilz M (1997) Hibernation, Aestivation und täglicher Torpor beim Siebenschläfer (Glis glis L.). PhD Thesis, Philipps-Universität Marburg
Wilz M, Heldmaier G (2000) Comparison of hibernation, estivation and daily torpor in the edible dormouse, Glis glis. J Comp Physiol B 170:511–521. https://doi.org/10.1007/s003600000129
Wolff JO (1996) Population fluctuations of mast-eating rodents are correlated with production of acorns. J Mammal 77:850–856. https://doi.org/10.2307/1382690
Acknowledgements
We thank S. Bigalk, S. Block, J. Flügge, C. Franke, F. Hofmann K. Pilsl, L. Kastner, E. Keppner, J. Saar, and P. Schütz for support in the field. We further thank M. Tschapka for providing additional OxBoxes. James Turner, Thomas Ruf, and Claudia Bieber provided valuable comments on an earlier draft of this paper. The Forestry Office of Nagold kindly provided details on mast phenology. Financial support was provided by the Deutsche Forschungsgemeinschaft (FI 831/3-1; FI 831/5-1 and FI 831/6-1), the Margarete von Wrangell Programme, the German Wildlife Foundation and the Deutsche Bundesstiftung Umwelt all to JF.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Our studies were conducted under license from the Nature Conservancy (Permit Numbers: 55-6/8852.15 and 55-6/8852.15-1) and the Committee on the Ethics of Animal Experiments of the Regional Commission of Tübingen (Permit Numbers: 892 and HOH 25/13).
Additional information
Communicated by G. Heldmaier.
Rights and permissions
About this article
Cite this article
Langer, F., Havenstein, N. & Fietz, J. Flexibility is the key: metabolic and thermoregulatory behaviour in a small endotherm. J Comp Physiol B 188, 553–563 (2018). https://doi.org/10.1007/s00360-017-1140-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-017-1140-3