Abstract
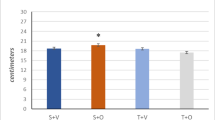
The regulation of energy metabolism is one of the major functions of steroid hormones. This study was performed to explore whether testosterone can regulate the aerobic capacity of skeletal muscles via myoglobin expression. To study this, changes in testosterone level were quantified, and the level of myoglobin protein was analyzed using Western blot in mice subjected to 6 weeks of training (T) or testosterone administration (A). Both treatments significantly increased the plasma testosterone level when compared to the untrained (U) or control (C) group. Training induced a significant increase in the myoglobin content in gastrocnemius and plantaris muscles (287 and 83%, respectively). Testosterone administration increased myoglobin concentration in plantaris (183%) but not in gastrocnemius. In extensor digitorum longus muscle the protein content decreased slightly after exercise, but increased 78% after testosterone administration. In soleus and rectus femoris muscles the myoglobin content was unchanged after both treatments. The data show that testosterone and training have differential effects on the concentration of myoglobin in some, but not all muscles. This may have an influence on the aerobic capacity in mouse skeletal muscles. The data demonstrated that both testosterone administration and training induced an increase in plasma testosterone level. However, the effects of the treatments on the myoglobin concentration differ.


Similar content being viewed by others
Abbreviations
- EDL:
-
Musculus extensor digitorum longus
- GAS:
-
Musculus gastrocnemius
- Mb:
-
Myoglobin
- PL:
-
Musculus plantaris
- RF:
-
Musculus rectus femoris
- SO:
-
Musculus soleus
References
Allen DL, Harrison BC, Maass A, Bell ML, Byrnes WC, Leinwand LA (2001) Cardiac and skeletal muscle adaptations to voluntary wheel running in the mouse. J Appl Physiol 90:1900–1908
Ariano MA, Armstrong RB, Edgerton VR (1973) Hindlimb muscles fiber populations of five mammals. J Histochem Cytochem 21:51–55
Baum O, Da Silva-Azevedo L, Willerding G, Wöckel A, Planitzer G, Gossrau R, Pries AR, Zakrzewicz A (2004) Endothelial NOS is main mediator for shear stress-dependent angiogenesis in skeletal muscle after prazosin administration. Am J Physiol 287:H2300–H2308
Bhasin S, Woodhouse L, Storer TW (2001) Hormones and sport: proof of the effect of testosterone on skeletal muscle. J Endocrinol 170:27–38
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bricout V-A, Serrurier BD, Bigard AX, Guzennec CY (1999) Effects of hindlimb suspension and androgen treatment on testosterone receptors in rat skeletal muscles. Eur J Appl Physiol 79:443–448
Chaĭkovskiĭ VS, Basharina OB, Shaliapina IV, Rogozkin VA (1987) Effect of physical exercise on myoglobin and tropomyosin levels in skeletal muscles and myoglobin level in rat blood. Vopr Med Khim 33:79–83
Conley KE, Jones C (1996) Myoglobin content and oxygen diffusion: model analysis of horse and steer muscle. Am J Physiol 271:C2027–C2036
Conley KE, Ordway GA, Richardson RS (2000) Deciphering the mysteries of myoglobin in striated muscle. Acta Physiol Scand 168:623–634
Conley KE, Kemper WF, Crowther GJ (2001) Limits to sustainable muscle performance: interaction between glycolysis and oxidative phosphorylation. J Exp Biol 204:3189–3194
Demirel HA, Powers SK, Naito H, Hughes M, Coombes JS (1999) Exercise-induced alterations in skeletal muscle myosin heavy chain phenotype: dose-response relationship. J Appl Physiol 86:1002–1008
Devlin BH, Wefald FC, Kraus WE, Bernard TS, Williams RS (1989) Identification of a muscle-specific enhancer within the 5′ flanking region of the human myoglobin gene. J Biol Chem 264:13896–13900
Egginton S, Hudlická O, Brown MD, Walter H, Weiss JB, Bate A (1998) Capillary growth in relation to blood flow and performance in overloaded rat skeletal muscle. J Appl Physiol 85:2025–2032
Garry DJ, Ordway GA, Lorenz JN, Radford NB, Chin ER, Grange RW, Bassel-Duby R, Williams RS (1998) Mice without myoglobin. Nature 395:905–908
Grange RW, Meeson A, Chin E, Lau KS, Stull JT, Shelton JM, Sanders-Williams R, Garry DJ (2001) Functional and molecular adaptations in skeletal muscle of myoglobin-mutant mice. Am J Physiol Cell Physiol 281:1487–1494
Guezennec CY, Ferre P, Serrurier B, Merino D, Aymonod M, Pesquies PC (1984) Metabolic effects of testosterone during prolonged physical exercise and fasting. Eur J Appl Physiol Occup Physiol 52:300–304
Gödecke A, Flögel U, Zanger K, Zanger K, Ding Z, Hirchenhain J, Decking UK, Schrader J (1999) Disruption of myoglobin in mice induces multiple compensatory mechanisms. Proc Natl Acad Sci USA 96:10495–10500
Hackney AC, Szczepanowska E, Viru AM (2003) Basal testicular testosterone production in endurance-trained men is suppressed. Eur J Appl Physiol 89:198–201
Harms SJ, Hickson RC (1983) Skeletal muscle mitochondria and myoglobin, endurance, and intensity of training. J Appl Physiol 54:798–802
Hartgens F, Kuipers H (2004) Effects of androgenic-anabolic steroids in athletes. Sports Med 34:513–554
Heineman FW, Balaban RS (1990) Control of mitochondrial respiration in the heart in vivo. Annu Rev Physiol 52:523–542
Hepple RT, Vogell JE (2004) Anatomic capillarization is maintained in relative excess of fiber oxidative capacity in some skeletal muscles of late middle-aged rats. J Appl Physiol 96:2257–2264
Hickson RC (1981) Skeletal muscle cytochrome c and myoglobin, endurance, and frequency of training. J Appl Physiol 51:746–749
Holmäng A, Svedberg J, Jennische E, Björntorp P (1990) Effects of testosterone on muscle insulin sensitivity and morphology in female rats. Am J Physiol 259:E555–E560
Joubert Y, Tobin C (1995) Testosterone treatment results in quiescent satellite cells being activated and recruited into cell cycle in rat levator ani muscle. Dev Biol 169:286–294
Kraemer WJ, Patton JF, Gordon SE, Harman EA, Deschenes MR, Reynolds K, Newton RU, Triplett NT, Dziados JE (1995) Compatibility of high-intensity strength and endurance training on hormonal and skeletal muscle adaptations. J Appl Physiol 78:976–989
Kyröläinen H, Takala TE, Komi PV (1998) Muscle damage induced by stretch-shortening cycle exercise. Med Sci Sports Exerc 30:415–420
Laemmli UK (1970) Cleavage of structural proteins during assembly of head of bacteriophage T4. Nature 227:680–685
Lamb DR (1989) Anabolic steroids and athletic performance. In: Laron Z, Rogol A (eds) Hormones and sport. Raven, New York, pp 257–273
Lin J, Wu R, Tarr PT, Zhang C-W, Wu Z, Boss O, Michael LF, Puigserver P, Isotani E, Olson EN, Lowell BB, Bassel-Duby R, Spiegleman BM (2002) Transcriptional co-activator PGC-1a drives the formation of slow-twitch muscle fibers. Nature 418:797–801
Mänttäri S, Anttila K, Kaakinen M, Järvilehto M (2006) Effects of low intensity training on dihydropyridine and ryanodine receptor content in skeletal muscle of mouse. J Physiol Biochem 62:293–302
Martinez JA, Buttery PJ, Pearson JT (1984) The mode of action of anabolic agents, the effects of testosterone on growth rates and muscle protein metabolism in female rats. Br J Nutr 54:515–521
McDonald R, Hegenauer J, Sucec A, Saltman P (1984) Effects of deficiency and exercise on myoglobin in rats. Eur J Appl Physiol 52:414–419
Merx MW, Gödecke A, Flögel U, Schrader J (2005) Oxygen supply and nitric oxide scavenging by myoglobin contribute to exercise endurance and cardiac function. FASEB J 19:1015–1017
Millikan GA (1937) Experiments on muscle hemoglobin in vivo: the instantaneous measurement of muscle metabolism. Proc Royal Soc Ser B 123:218–241
Nicolopoulos-Stournaras S, Iles JF (1984) Hindlimb muscle activity during locomotion in the rat (Rattus norvegicus) (Rodentia: Muridae). J Zool Lond 203:427–440
Ordway GA, Garry DJ (2004) Myoglobin: an essential hemoprotein in striated muscle. J Exp Biol 207:3441–3446
Pansarasa O, D’Antona G, Gualea MR, Marzani B, Pellegrino MA, Marzatico F (2002) “Oxidative stress”: effects of mild endurance training and testosterone treatment on rat gastrocnemius muscle. Eur J Appl Physiol 87:550–555
Pette D, Staron RS (2001) Transitions of muscle fiber phenotypic profiles. Histochem Cell Biol 115:359–372
Prezant DJ, Karwa MJ, Kim HH, Maggiore D, Chung V, Valentine DE (1997) Short- and long-term effects of testosterone on diaphragm in castrated and normal male rats. J Appl Physiol 82:134–143
Richardson RS, Noyszewski EA, Kendrick KF, Leigh JS, Wagner PD (1995) Myoglobin O2 desaturation during exercise: evidence of limited O2 transport. J Clin Invest 96:1916–1926
Rogozkin VA (1979) Anabolic steroid metabolism in skeletal muscle. J Steroid Biochem 11:923–926
Scheller K, Sekeris CE (2003) The effects of steroid hormones on the transcription of genes encoding enzymes of oxidative phosphorylation. Exp Physiol 88:129–140
Scow RO, Roe JH (1953) Effect of testosterone propionate on the weight and myoglobin content of striated muscles in gonadoectomized guinea pigs. Am J Physiol 173:22–28
Sinha-Hikim I, Cornford M, Gaytan H, Lee ML, Bhasin S (2006) Effects of testosterone supplementation on skeletal muscle fiber hypertrophy and satellite cells in community-dwelling, older men. J Clin Endocrin Metab 91:3024–3033
Stuart MK, Howley ET, Gladden LB, Cox RH (1981) Efficiency of trained subjects differing in maximal oxygen uptake and type of training. J Appl Physiol 50:444–449
Tagarakis CVM, Bloch W, Hartmann G, Hollmann W, Addicks K (2000) Testosterone-propionate impairs the response of the cardiac capillary bed to exercise. Med Sci Sports Exerc 32:946–953
Totsuka Y, Nagao Y, Horii T, Yonekawa H, Imai H, Hatta H, Izaike Y, Tokunaga T, Atomi Y (2003) Physical performance and soleus muscle fiber composition in wild-derived and laboratory inbred mouse strains. J Appl Physiol 95:720–727
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc Natl Acad Sci USA 76:4350–4354
Tsika RW, Herrick RE, Baldwin KM (1987) Effect of anabolic steroids in skeletal muscle mass during hindlimb suspension. J Appl Physiol 63:2122–2127
Underwood LE, Williams RS (1987) Pretranslational regulation of myoglobin gene expression. Am J Physiol Cell Physiol 252:C450–C453
Üstunel I, Akkoyunlu G, Demir R (2003) The effect of testosterone on gastrocnemius muscle fibers in growing and adult male and female rats: a histochemical, morphometric and ultrastructural study. Anat Histol Embryol 32:70–79
Wilson JD, Griffin JE (1980) The use and misuse of androgens. Metabolism 29:1278–1295
Wittenberg JB (1970) Myoglobin-facilitated oxygen diffusion: role of myoglobin in oxygen entry into muscle. Physiol Rev 50:559–636
Wittenberg JB, Wittenberg BA (2003) Myoglobin function reassessed. J Exp Biol 206:2011–2020
Yoshioka M, Boivin A, Ye P, Labrie F, St-Amand J (2006) Effects of dihydrosterone on skeletal muscle transcriptome in mice measured by serial analysis of gene expression. J Mol Endocrinol 36:247–259
Zhan WZ, Swallow JG, Garland T Jr, Proctor DN, Carter PA, Sieck GC (1999) Effects of genetic selection and voluntary activity on the medial gastrocnemius muscle in house mice. J Appl Physiol 87:2326–2333
Acknowledgments
The authors express appreciation to Dr. Esa Hohtola for his careful reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Heldmaier.
Rights and permissions
About this article
Cite this article
Mänttäri, S., Anttila, K. & Järvilehto, M. Testosterone stimulates myoglobin expression in different muscles of the mouse. J Comp Physiol B 178, 899–907 (2008). https://doi.org/10.1007/s00360-008-0280-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-008-0280-x