Abstract
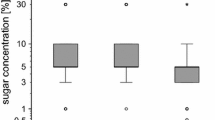
The parasitic mite Varroa destructor influences flight behavior, orientation and returning success of forager honeybees (Apis mellifera) infested as adults. As impaired orientation toward the nest entrance might be due to deficiency in recognition and responsiveness to stimuli in the environment, we examined effects of V. destructor on sensory responsiveness, non-associative and associative learning of honey bee foragers by using proboscis extension reaction paradigm (PER). Although infested and uninfested workers were initially equally responsive to different concentrations of sugar water, we found differences in non-associative learning. In habituation, PER to repeated sugar stimulation of the antennae occurred faster in infested foragers compared to uninfested foragers. In sensitization, infested foragers showed a lower response to an odor stimulus following sugar stimulation than non-infested foragers. Differences in non-associative paradigms were more pronounced in bees with lower responsiveness to sucrose. In conditioning learning experiments, a significant reduction in proboscis extension response was found 1 min but not 12 min after a single conditioning trial indicating that V. destructor predominantly affects the non-associative components of learning and its underlying neural and molecular processes.




Similar content being viewed by others
References
Bicker G, Hahnlein I (1994) Long-term habituation of an appetitive reflex in the honeybee. Neuroreport 30:54–56
Bitterman ME, Menzel R, Fietz A, Schäfer S (1983) Classical conditioning of proboscis extension in honeybees (Apis mellifera). J Comp Psychol 97:107–119
Boecking O, Drescher W (1992) The removal response of Apis mellifera L. colonies to brood in wax and plastic cells after artificial and natural infestation with Varroa jacobsoni Oud. and to freeze-killed brood. Exp Appl Acarol 16:321–329
Braun G, Bicker G (1992) Habituation of an appetitive reflex in the honeybee. J Neurophysiol 67:588–598
Büchler R, Drescher W, Tornier I (1992) Grooming behaviour of Apis cerana, Apis mellifera and Apis dorsata and its effect on the parasitic mites Varroa jacobsoni and Tropilaelaps clareae. Exp Appl Acarol 16:313–319
Contzen C, Garedew A, Schmolz E (2003) The influence of Varroa destructor mites on energy content, haemolymph volume and protein concentration of bee pupae. Apidologie 34:503–504
Fries I, Huazhen W, Wei S, Jin CS (1996) Grooming behaviour and damaged mites (Varroa jacobsoni) in Apis cerana and Apis mellifera ligustica. Apidologie 27:3–11
Fuchs F, Kutschker C (2000) The role of forager bee infestation on population dynamic of Varroa destructor (Anderson Trueman 2000). Pszczelnicze Zeszyty Naukowe 44:199–208
Gilliam M, Taber S, Richardson GV (1983) Hygienic behavior of honey bees in relation to chalkbrood disease. Apidologie 14:29–39
Gregory PG, Evans JD, Rinderer T, De Guzman L (2005) Conditional immune-gene suppression of honeybees parasitized by Varroa mites. J Insect Sci 5:1–5
Hamilton WD, Zuk M (1982) Heritable true fitness and bright birds: a role for parasites? Science 218:384–387
Hammer M, Braun G, Mauelshagen J (1994) Food-induced arousal and nonassociative learning in honeybees:dependence of sensitization on the application site and duration of food stimulation. Behav Neural Biol 62:210–223
Jog M, Watve M (2005) Role of parasites and commensals in shaping host behaviour. Curr Sci 89:1184–1191
Kandel ER (1991) Cellular mechanisms of learning and the biological basis of individuality. In: Kandel ER, Schwartz J, Jessel TM (eds) Principles of neural sciences, 3rd edn. Appleton and Lange, East Norwalk, pp 1010–1030
Kralj J (2004) Parasite–host interactions between V. destructor Anderson and Trueman and Apis mellifera L.: influence of parasitism on flight behaviour and on the loss of infested foragers. Dissertation, Fachbereich Biologie und Informatik der Johann Wolfgang Goethe Universitat Frankfurt am Main, p 122
Kralj J, Fuchs S (2006) Parasitic Varroa destructor mites influence flight duration and homing ability of infested Apis mellifera foragers. Apidologie 37 (in print)
Kuwabara M (1957) Bildung des bedingten Reflexes von Pavlovs Typus bei der Honigbene, Apis mellifica. J Fac Sci Hokkaido Univ Zool 13:458–464
Mallon EB, Brockmann A, Schmidt-Hempel P (2003) Immune response inhibits associative learning in insects. Proc R Soc Lond B 270:2471–2473
McAllister MK, Roitberg BD, Weldon KL (1990) Adaptive suicide in pea aphids: decisions are cost sensitive. Anim Behav 40:167–175
Menzel R (1999) Memory dynamics in the honeybee. J Comp Physiol A 185:323–340
Menzel R, Heyne A, Kinzel C, Gerber B, Fiala A (1999) Pharmacological dissociation between reinforcing, sensitizing, and response releasing function of reward in honeybee classical conditioning. Behav Neurosci 113:744–754
Moore J (2002) Behavioral alternations and avoiding parasites. In: May RM, Harvey PH (eds) Parasites and the behavior of animals. Oxford University Press, Oxford, pp 89–118
Moore J, Freehling M (2002) Cockroach hosts in thermal gradients suppress parasite development. Behav Ecol 133:261–266
Müller CB, Schmid-Hempel P (1993) Exploitation of cold temperature as defence against parasitoids in bumblebees. Nature 363:65–67
Page RE, Erber J, Fondrk MK (1998) The effect of genotype on response thresholds to sucrose and foraging behavior of honey bees (Apis mellifera L.). J Comp Physiol A 182:489–500
Pankiw T (2004) Cued in: honey bees pheromones as information flow and collective decision-making. Apidologie 35:217–226
Pankiw T, Page RE (1999) The effect of genotype, age, sex and caste on response thresholds to sucrose and foraging behavior of honey bees (Apis mellifera L.). J Comp Physiol A 185:207–213
Pankiw T, Page RE (2000) Response thresholds to sucrose predict foraging division of labor in honeybees. Behav Ecol Sociobiol 47:265–267
Peng YS, Fang Y, Shaoyu X, Ge L (1987) The resistance mechanism of the Asian honey bee, Apis cerana Fabr., to an ectoparasitic mite, Varroa jacobsoni Oudemans. J Invertebr Pathol 49:54–60
Poulin R (1995) “Adaptive” changes in the behaviour of parasitized animals: a critical review. Int J Parasitol 25:1371–1383
Riddel CE, Mallon EB (2006) Insect psychoneuroimmunology: immune response reduces learning in protein starved bumblebees (Bombus terrestris). Brain Behav Immun 20:135–138
Sakofski F (1990) Quantitative investigations on transfer of Varroa jacobsoni Oud. In: Ritter W, Van Laere O, Jacobs F, De Wael L (eds) Proceedings of the international symposium on recent research on bee pathology. Apimondia, Gent, Belgium, pp 70–72
Scheiner R (2004) Responsiveness to sucrose and habituation of the proboscis extension response in honey bees. J Comp Physiol A 190:727–733
Scheiner R, Erber J, Page RE (1999) Tactile learning and the individual evaluation of the reward in honey bees (Apis mellifera L.). J Comp Physiol A 185:1–10
Scheiner R, Page RE, Erber J (2001a) Responsiveness to sucrose affects tactile and olfactory learning in preforaging honey bees of two genetic strains. Behav Brain Res 120:67–73
Scheiner R, Page RE, Erber J (2001b) The effect of genotype foraging role and sucrose responsiveness on the tactile learning performance of honey bees (Apis mellifera L.). Neurobiol Learn Mem 76:138–150
Scheiner R, Barnert M, Erber J (2003) Variation in water and sucrose responsiveness during the foraging season affects proboscis extension learning in honey bees. Apidologie 34:67–72
Scheiner R, Page RE, Erber J (2004) Sucrose responsiveness and behavioural plasticity in honey bees (Apis mellifera). Apidologie 35:133–142
Smith-Trail DR (1980) Behavioral interactions between parasites and hosts: host suicide and the evolution of complex life cycles. Am Nat 116:77–91
Spivak M, Reuter GS (1997) Performance of hygienic colonies in a commercial apiary. Am Bee J 137:228
Takeda K (1961) Classical olfactory conditioned response. J Insect Physiol 6:168–179
Tautz J, Maier S, Groh C, Rössler W, Brockmann A (2003) Behavioral performance in adult honey bees is influenced by the temperature experienced during their pupal development. PNAS 100:7343–7347
Yang X, Cox-Foster DL (2005) Impact of an ectoparasite on the immunity and pathology of an invertebrate: evidence for host immunosupression and viral amplification. PNAS 102:7470–7475
Acknowledgments
We want to thank the people of the BEEgroup, Würzburg for their support and Dr. Ricarda Scheiner, TU Berlin, for useful suggestions on a previous draft. The experiments comply with the principles of animal care, publication No. 86–23, revised 1985, of the National Institute of Health and also with the current laws of the country in which the experiments were performed.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jasna Kralj and Axel Brockmann have contributed equally to this study.
Rights and permissions
About this article
Cite this article
Kralj, J., Brockmann, A., Fuchs, S. et al. The parasitic mite Varroa destructor affects non-associative learning in honey bee foragers, Apis mellifera L.. J Comp Physiol A 193, 363–370 (2007). https://doi.org/10.1007/s00359-006-0192-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-006-0192-8