Abstract
Purpose
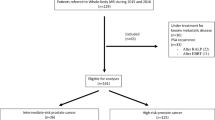
It is generally accepted that when metastases develop in a patient with biochemical recurrence of prostate cancer (PCa), they follow a centrifuge pattern of seeding from the pelvis and that most patients enter the disease as oligometastatic. In this study, we used whole-body magnetic resonance imaging (WB-MRI) to assess the anatomical distribution of oligo- and polymetastatic disease and the impact of the initial treatment on this distribution in patients.
Materials and methods
WB-MRI examinations of patients with a rising prostate-specific antigen (PSA) after radical treatment by surgery or/and radiotherapy were analyzed for disease recurrence. The patients were separated into three groups, based on the primary treatment: patients treated by radical prostatectomy without radiotherapy and with/without lymph node dissection (RP), patients treated only by radiotherapy or hormono-radiotherapy (RT) and patients treated with radical prostatectomy and adjuvant or salvage radiotherapy (RP + RT). Patients with ≤ 5 bone or/and node metastases were considered oligometastatic. Regional distributions of bone and lymph nodes metastases were reported using anatomical diagrams. Univariate and multivariable logistic regressions were performed to identify prognostic factors of relapse.
Results
The primary treatment (RP, RT, RP + RT), Gleason score, PSA at relapse, time between first diagnosis and recurrence did not influence the metastatic status (oligo vs. polymetastatic). Oligometastatic patients showed different distribution of bone metastases compared to the polymetastatic ones and the distribution of the oligometastatic disease was not influenced by the primary treatment.
Conclusions
In this WB-MRI-based study, there was no evidence that the primary treatment influenced the metastatic status of the patient or the distribution of the oligometastatic disease.







Similar content being viewed by others
References
Roehl KA et al (2004) Cancer progression and survival rates following anatomical radical retropubic prostatectomy in 3478 consecutive patients: long-term results. J Urol 172(3):910–914
Hellman S, Weichselbaum RR (1995) Oligometastases. J Clin Oncol 13(1):8–10
Padhani AR et al (2017) Rationale for modernising imaging in advanced prostate cancer. Eur Urol Focus 3(2–3):223–239
Lecouvet FE et al (2012) Can whole-body magnetic resonance imaging with diffusion-weighted imaging replace Tc 99 m bone scanning and computed tomography for single-step detection of metastases in patients with high-risk prostate cancer? Eur Urol 62(1):68–75
Gupta SK et al (2017) Prostate-specific membrane antigen positron emission tomography-computed tomography for prostate cancer: distribution of disease and implications for radiation therapy planning. Int J Radiat Oncol Biol Phys 99(3):701–709
Lepinoy A et al (2014) Pattern of occult nodal relapse diagnosed with (18)F-fluoro–choline PET/CT in prostate cancer patients with biochemical failure after prostate-only radiotherapy. Radiother Oncol 111(1):120–125
Ost P et al (2016) Pattern of progression after stereotactic body radiotherapy for oligometastatic prostate cancer nodal recurrences. Clin Oncol R Coll Radiol 28(9):e115–e120
Sobol I et al (2017) Contemporary mapping of post-prostatectomy prostate cancer relapse with (11)C-Choline positron emission tomography and multiparametric magnetic resonance imaging. J Urol 197(1):129–134
Larbi A et al (2016) Whole body MRI (WB-MRI) assessment of metastatic spread in prostate cancer: therapeutic perspectives on targeted management of oligometastatic disease. Prostate 76(11):1024–1033
Eisenhauer EA et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45(2):228–247
Koh DM, Hughes M, Husband JE (2006) Cross-sectional imaging of nodal metastases in the abdomen and pelvis. Abdom Imaging 31(6):632–643
Ahmed KA et al (2012) Stereotactic body radiation therapy in the treatment of oligometastatic prostate cancer. Front Oncol 2:215
Tabata K et al (2012) Radiotherapy for oligometastases and oligo-recurrence of bone in prostate cancer. Pulm Med 2012:541656
Mehta CR, Patel NR (1983) A network algorithm for performing Fisher’s exact test in r c contingency tables. J Am Stat Assoc 78:427–434
Newcombe RG (1998) Two-sided confidence intervals for the single proportion: comparison of seven methods. Stat Med 17(8):857–872
Pampel FC (2000) Logistic regression: a primer. Quantitative applications in the social sciences. Thousand oaks. Sage Publications, California
Tosoian JJ et al (2017) Oligometastatic prostate cancer: definitions, clinical outcomes, and treatment considerations. Nat Rev Urol 14(1):15–25
Singh D et al (2004) Is there a favorable subset of patients with prostate cancer who develop oligometastases? Int J Radiat Oncol Biol Phys 58(1):3–10
Solloway MJ et al (1998) Mice lacking Bmp6 function. Dev Genet 22(4):321–339
Gillessen S et al (2018) Management of patients with advanced prostate cancer: the report of the advanced prostate cancer consensus conference APCCC 2017. Eur Urol 73(2):178–211
Parker CC et al (2018) Radiotherapy to the primary tumour for newly diagnosed, metastatic prostate cancer (STAMPEDE): a randomised controlled phase 3 trial. Lancet 392:2353–2366
Tamoto E et al (2004) Gene-expression profile changes correlated with tumor progression and lymph node metastasis in esophageal cancer. Clin Cancer Res 10(11):3629–3638
Lussier YA et al (2011) MicroRNA expression characterizes oligometastasis(es). PLoS One 6(12):e28650
Gundem G et al (2015) The evolutionary history of lethal metastatic prostate cancer. Nature 520(7547):353–357
Murphy DG, Sweeney CJ, Tombal B (2017) “Gotta Catch ‘em All”, or do we? Pokemet approach to metastatic prostate cancer. Eur Urol 72(1):1–3
Ost P et al (2018) Surveillance or metastasis-directed therapy for oligometastatic prostate cancer recurrence: a prospective, randomized, multicenter phase II trial. J Clin Oncol 36(5):446–453
Muacevic A et al (2013) Safety and feasibility of image-guided robotic radiosurgery for patients with limited bone metastases of prostate cancer. Urol Oncol 31(4):455–460
Richard PJ, Rengan R (2016) Oligometastatic non-small-cell lung cancer: current treatment strategies. Lung Cancer (Auckl) 7:129–140
Simmonds PC et al (2006) Surgical resection of hepatic metastases from colorectal cancer: a systematic review of published studies. Br J Cancer 94(7):982–999
Graves A et al (2013) Metastatic renal cell carcinoma: update on epidemiology, genetics, and therapeutic modalities. Immunotargets Ther 2:73–90
Lecouvet FE et al (2007) Magnetic resonance imaging of the axial skeleton for detecting bone metastases in patients with high-risk prostate cancer: diagnostic and cost-effectiveness and comparison with current detection strategies. J Clin Oncol 25(22):3281–3287
Hirmas N, Al-Ibraheem A, Herrmann K, Alsharif A, Muhsin H, Khader J, Al-Daghmin A, Salah S (2018) [68Ga]PSMA PET/CT improves initial staging and management plan of patients with high-risk prostate cancer. Mol Imaging Biol. https://doi.org/10.1007/s11307-018-1278-8
Padhani AR et al (2017) METastasis reporting and data system for prostate cancer: practical guidelines for acquisition, interpretation, and reporting of whole-body magnetic resonance imaging-based evaluations of multiorgan involvement in advanced prostate cancer. Eur Urol 71(1):81–92
Park SY et al (2018) Gallium 68 PSMA-11 PET/MR imaging in patients with intermediate- or high-risk prostate cancer. Radiology 288(2):495–505
Dyrberg E et al (2018) (68)Ga-PSMA-PET/CT in comparison with (18)F-fluoride-PET/CT and whole-body MRI for the detection of bone metastases in patients with prostate cancer: a prospective diagnostic accuracy study. Eur Radiol 29:1221–1230
Pasoglou V et al (2014) One-step TNM staging of high-risk prostate cancer using magnetic resonance imaging (MRI): toward an upfront simplified “all-in-one” imaging approach? Prostate 74(5):469–477
Pasoglou V et al (2015) Whole-body 3D T1-weighted MR imaging in patients with prostate cancer: feasibility and evaluation in screening for metastatic disease. Radiology 275(1):155–166
Lecouvet FE et al (2018) Use of modern imaging methods to facilitate trials of metastasis-directed therapy for oligometastatic disease in prostate cancer: a consensus recommendation from the EORTC imaging group. Lancet Oncol 19(10):e534–e545
Parker WP et al (2017) Identification of site-specific recurrence following primary radiation therapy for prostate cancer using C-11 choline positron emission tomography/computed tomography: a nomogram for predicting extrapelvic disease. Eur Urol 71(3):340–348
Soldatov A et al (2019) Patterns of progression after (68)Ga-PSMA-ligand PET/CT-guided radiation therapy for recurrent prostate cancer. Int J Radiat Oncol Biol Phys 103(1):95–104
Rischke HC et al (2015) Adjuvant radiotherapy after salvage lymph node dissection because of nodal relapse of prostate cancer versus salvage lymph node dissection only. Strahlenther Onkol 191(4):310–320
Meijer HJ et al (2013) Geographical distribution of lymph node metastases on MR lymphography in prostate cancer patients. Radiother Oncol 106(1):59–63
Author information
Authors and Affiliations
Contributions
VP, NM, JVD, SVN, BT, FEL: protocol/project development, data collection or management, data analysis, manuscript writing/editing. PT: data collection or management. MH: drawings
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pasoglou, V., Michoux, N., Van Damme, J. et al. Pattern of metastatic deposit in recurrent prostate cancer: a whole-body MRI-based assessment of lesion distribution and effect of primary treatment. World J Urol 37, 2585–2595 (2019). https://doi.org/10.1007/s00345-019-02700-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-019-02700-2