Abstract
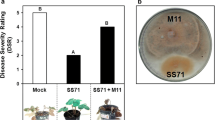
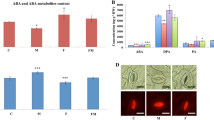
Microbe-associated molecular patterns (MAMPs) are conserved molecules able to trigger plant resistance. The aim of this work was to evaluate the capacity of Azospirillum brasilense REC3 polar flagellin AzFlap as a MAMP, eliciting biochemical, histological, and molecular defense responses that can provide strawberry plants protection against the pathogenic fungus Macrophomina phaseolina. Strawberry plants were treated with AzFlap on leaves or with the isolate REC3 on leaves or roots. Salicylic acid content, biofilm formation, callose and lignin depositions, stomatal closure, ROS, and the expression of defense-related genes such as FaPR1, FaCAT, FaRBOH-D, FaRBOH-F, FaCHI23, FaCHI2-2, and FaGSL5 were evaluated. Phytopathogenic assays in plants treated with AzFlap or REC3 and infected with M. phaseolina were also performed. Results showed that plants leaf treated with AzFlap or root treated with REC3 caused the accumulation of ROS, salicylic acid, callose, lignin, the increase of biofilm formation on leaves, and stomatal closure. The evaluation of the expression of genes associated to defense response indicated the activation of the innate immunity of strawberry plants. The level of gene expression was strongly time and treatment dependent, suggesting a complex regulation of defense signaling. Root inoculations with REC3 or foliar treatment with AzFlap were able to reduce plant mortality, showing the effectiveness of both treatments to control M. phaseolina. These results indicate that flagellin AzFlap from A. brasilense REC3 behaves as a MAMP that activates a defense response against M. phaseolina in strawberry plants.








Similar content being viewed by others
References
Aguirre CM (2012) Diferenciación de aislamientos de Macrophomina phaseolina (Tassi) Goid., agente causal de la podredumbre carbonosa de corona y raíz de frutilla (Fragaria ananassa Duch.). Bachelor thesis, Universidad Nacional de Tucumán, Argentina
Alagna F, Balestrini R, Chitarra W, Marsico AD, Nerva L (2020) Getting ready with the priming: innovative weapons against biotic and abiotic crop enemies in a global changing scenario. In: Hossain MA, Liu F, Burritt DJ, Fujita M, Huang B (eds) Priming-mediated stress and cross-stress tolerance in crop plants, 1st edn. Elsevier Inc, Amsterdam, pp 35–56
Allasia V, Industri B, Ponchet M, Quentin M, Favery B, Keller H (2018) Quantification of salicylic acid (SA) and SA-glucosides in Arabidopsis thaliana. Bio-Protoc 8:1–8
Amsbury S, Kirk P, Benitez-Alfonso Y (2018) Emerging models on the regulation of intercellular transport by plasmodesmata-associated callose. J Exp Bot 69(1):105–115. https://doi.org/10.1093/jxb/erx337
Baino OM, Ramallo AC, Kirschbaum DS (2011) First report of Macrophomina phaseolina causing strawberry crown and root ot in Northwestern Argentina. Plant Dis 95:1477. https://doi.org/10.1094/PDIS-03-ll-0193
Baldani JI, Reis VM, Videira SS, Boddey LH, Divan Baldani VL (2014) The art of isolating nitrogen-fixing bacteria from non-leguminous plants using N-free semi-solid media: a practical guide for microbiologists. Plant Soil 384:413–431. https://doi.org/10.1007/s11104-014-2186-6
Bale JS, Van Lenteren JC, Bigler F (2008) Biological control and sustainable food production. Philos Trans R Soc B Biol Sci 363:761–776. https://doi.org/10.1098/rstb.2007.2182
Bashan Y (1986) Migration of the rhizosphere bacteria Azospirillum brasilense and Pseudomonas fluorescens towards wheat roots in the soil. Microbiology 132:3407–3414. https://doi.org/10.1099/00221287-132-12-3407
Bashan Y, De-Bashan LE (2005) Plant growth-promoting. In: Hillel D (ed) Encyclopedia of soils in the environment, 1st edn. Elsevier, Oxford, pp 103–115
Bashan Y, De-Bashan LE (2010) How the plant growth-promoting bacterium Azospirillum promotes plant growth-a critical assessment. In: Advances in agronomy, 1st edn. Elsevier Inc., Amsterdam, pp 77–136
Bolwell GP, Bindshedler LV, Blee KA, Butt VS, Davies DR, Gardner SL, Gerrish C, Minibayeva F (2002) The apoplastic oxidative burst in response to biotic stress in plants: a three-component system. J Exp Bot 53:1367–1376. https://doi.org/10.1093/jexbot/53.372.1367
Chen C, Bélanger RR, Benhamou N, Paulitz TC (1999) Role of salicylic acid in systemic resistance induced by Pseudomonas spp. against Pythium aphanidermatum in cucumber roots. Eur J Plant Pathol 105:477–486. https://doi.org/10.1023/A:1008743502784
Clay NK, Adio AM, Denoux C, Jander G, Ausubel FM (2009) Glucosinolate metabolites required for an Arabidopsis innate immune response. Science 323:95–101. https://doi.org/10.1126/science.1164627
Conrath U, Beckers GJM, Langenbach CJG, Jaskiewicz MR (2015) Priming for enhanced defense. Annu Rev Phytopathol 53:97–119. https://doi.org/10.1146/annurev-phyto-080614-120132
Croes C, Moens S, Bastelaere E, Vanderleyden J, Michiels KW (1993) The polar flagellum mediates Azospirillum brasilense adsorption to wheat roots. J Gen Microbiol 139:2261–2269. https://doi.org/10.1099/00221287-139-9-2261
D’Ambrogio de Argüeso A (1986) Manual de técnicas de histología vegetal. Editora He, Buenos Aires
De Storme N, Geelen D (2014) Callose homeostasis at plasmodesmata: molecular regulators and developmental relevance. Front Plant Sci 5:138. https://doi.org/10.3389/fpls.2014.00138
Di Rienzo JA (2011) fgStatistics. Statistical software for the analysis of experiments of functional genomics. http://sites.google.com/site/fgStatistics/
Di Rienzo JA, Casanoves F, Balzarini MG, Gonzalez L, Tablada M, Robledo CW (2013) InfoStat versión 2018 Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. http://www.infostat.com.ar
Edmunds LK (1964) Combined relation of plant maturity, temperature, and soil moisture to charcoal stalk rot development in grain sorghum. Phytopathology 54:514–517
Elias JM, Guerrero-Molina MF, Martínez-Zamora MG, Díaz-Ricci JC, Pedraza RO (2018) Role of ethylene and related gene expression in the interaction between strawberry plants and the plant growth-promoting bacterium Azospirillum brasilense. Plant Biol 20:490–496. https://doi.org/10.1111/plb.12697
Ellinger D, Voigt CA (2014) Callose biosynthesis in arabidopsis with a focus on pathogen response: what we have learned within the last decade. Ann Bot 114:1349–1358. https://doi.org/10.1093/aob/mcu120
Freeman S, Katan T (1997) Identification of Colletotrichum species responsible for anthracnose and root necrosis of strawberry in Israel. Phytopathology 87:516–521. https://doi.org/10.1094/PHYTO.1997.87.5.516
Furio RN, Albornoz PL, Coll Y, Martínez-Zamora GM, Salazar SM, Martos GG, Díaz-Ricci JC (2019) Effect of natural and synthetic Brassinosteroids on strawberry immune response against Colletotrichum acutatum. Eur J Plant Pathol 153:227–241. https://doi.org/10.1007/s10658-018-1551-3
Grillo-Puertas M, Delaporte-Quintana P, Pedraza RO, Rapisarda VA (2018) Intracellular polyphosphate levels in Gluconacetobacter diazotrophicus affect tolerance to abiotic stressors and biofilm formation. Microbes Environ 33(2018):440–445. https://doi.org/10.1264/jsme2.ME18044
Guerrero-Molina MF, Winik BC, Pedraza RO (2012) More than rhizosphere colonization of strawberry plants by Azospirillum brasilense. Appl Soil Ecol 61:205–212. https://doi.org/10.1016/j.apsoil.2011.10.011
Guerrero-Molina MF, Lovaisa NC, Salazar SM, Martínez-Zamora MG, Díaz-Ricci JC, Pedraza RO (2015) Physiological, structural and molecular traits activated in strawberry plants after inoculation with the plant growth-promoting bacterium Azospirillum brasilense REC3. Plant Biol 17:766–773. https://doi.org/10.1111/plb.12270
Guidarelli M, Carbone F, Mourgues F, Perrottab G, Rosatib C, Bertolini P, Baraldi E (2011) Colletotrichum acutatum interactions with unripe and ripe strawberry fruits and differential responses at histological and transcriptional levels. Plant Pathol 60:685–697. https://doi.org/10.1111/j.1365-3059.2010.02423.x
Hael-Conrad V, Perato SM, Arias ME, Martínez-Zamora MG, Di Peto PDLA, Martos GG, Castagnaro AP, Díaz-Ricci JC, Chalfoun NR (2018) The elicitor protein AsES induces a systemic acquired resistance response accompanied by systemic microbursts and micro-hypersensitive responses in Fragaria ananassa. Mol Plant Microbe Interact 31(1):46–60. https://doi.org/10.1094/MPMI-05-17-0121-FI
Hall PG, Krieg NR (1983) Swarming of Azospirillum brasilense on solid media. Can J Microbiol 29:1592–1594. https://doi.org/10.1139/m83-243
Jez JM, Bowman ME, Dixon RA, Noel JP (2000) Structure and mechanism of the evolutionarily unique plant enzyme chalcone isomerase. Nat Struct Biol 7(9):786–791
Kawano T, Furuichi T, Muto S (2004) Controlled salicylic acid levels and corresponding signaling mechanisms in plants. Plant Biotechnol 21:319–335. https://doi.org/10.5511/plantbiotechnology.21.319
Koornneef A, Pieterse CMJ (2008) Cross talk in defense signaling. Plant Physiol 146:839–844. https://doi.org/10.1104/pp.107.112029
Lee JY (2015) Plasmodesmata: a signaling hub at the cellular boundary. Curr Opin Plant Biol 27:133–140. https://doi.org/10.1016/j.pbi.2015.06.019
Liu P, Sun F, Gao R, Dong H (2012) RAP26L overexpression delays waterlogging induced premature senescence by increasing stomatal closure more than antioxidant enzyme activity. Plant Mol Biol 79(6):609–622. https://doi.org/10.1007/s11103-012-9936-8
Lu YY, Liu YH, Chen CY (2007) Stomatal closure, callose deposition, and increase of LsGRP1-corresponding transcript in probenazole-induced resistance against Botrytis elliptica in lily. Plant Sci 172(5):913–919. https://doi.org/10.1016/j.plantsci.2006.12.020
Mandal S, Kar I, Mukherjee AK, Acharya P (2013) Elicitor-induced defense responses in Solanum lycopersicum against Ralstonia solanacearum. Sci World J. https://doi.org/10.1155/2013/561056
Martin FW (1959) Staining and observing pollen tubes in the style by means of fluorescence. Biotech Histochem 34:125–128. https://doi.org/10.3109/10520295909114663
Melotto M, Underwood W, Koczan J, Nomura K, He SY (2006) Plant stomata function in innate immunity against bacterial invasion. Cell 126:969–980. https://doi.org/10.1016/j.cell.2006.06.054
Melotto M, Zhang L, Oblessuc PR, He SY (2017) Stomatal defense a decade later. Plant Physiol 174(2):561–571. https://doi.org/10.1104/pp.16.01853
Mittler R, Vanderauwera S, Gollery M, Van Breusegem F (2004) Reactive oxygen gene network of plants. Trends Plant Sci 9:490–498. https://doi.org/10.1016/j.tplants.2004.08.009
Moens S, Michiels K, Keijers V, Van Leuven F, Vanderleyden J (1995a) Cloning, sequencing, and phenotypic analysis of laf1, encoding the flagellin of the lateral flagella of Azospirillum brasilense Sp7. J Bacteriol 177:5419–5426. https://doi.org/10.1128/jb.177.19.5419-5426.1995
Moens S, Michiels K, Vanderleyden J (1995b) Glycosylation of the flagellin of the polar flagellum of Azospirillum brasilense, a gram-negative nitrogen-fixing bacterium. Microbiology 141:2651–2657. https://doi.org/10.1099/13500872-141-10-2651
Mur LAJ, Bi YM, Darby RM, Firek S, Draper J (1997) Compromising early salicylic acid accumulation delays the hypersensitive response and increases viral dispersal during lesion establishment in TMV-infected tobacco. Plant J 12:1113–1126. https://doi.org/10.1046/j.1365-313X.1997.12051113.x
Mur LAJ, Brown IR, Darby RM, Bestwick CS, Bi YM, Mansfield JW, Draper J (2000) A loss of resistance to avirulent bacterial pathogens in tobacco is associated with the attenuation of a salicylic acid-potentiated oxidative burst. Plant J 23:609–621. https://doi.org/10.1046/j.1365-313X.2000.00825.x
Narula K, Elagamey E, Abdellatef MA, Sinha A, Ghosh S, Chakraborty N, Chakraborty S (2020) Chitosan-triggered immunity to Fusarium in chickpea is associated with changes in the plant extracellular matrix architecture, stomatal closure and remodeling of the plant metabolome and proteome. Plant J 103(2):561–583. https://doi.org/10.1111/tpj.14750
Nicholson RL, Hammerschmidt R (1992) Phenolic compounds and their role in disease resistance. Annu Rev Phytopathol 30(30):369–389. https://doi.org/10.1146/annurev.py.30.090192.002101
Pastor V, Luna E, Mauch-Mani B, Ton J, Flors V (2013) Primed plants do not forget. Environ Exp Bot 94:46–56. https://doi.org/10.1016/j.envexpbot.2012.02.013
Pedraza RO, Motok J, Tortora ML, Salazar SM, Díaz-Ricci JC (2007) Natural occurrence of Azospirillum brasilense in strawberry plants. Plant Soil 295:169–178. https://doi.org/10.1007/s11104-007-9273-x
Pedraza RO, Motok J, Salazar SM, Ragout AL, Mentel MI, Tortora ML, Guerrero-Molina MF, Winik BC, Díaz-Ricci JC (2010) Growth-promotion of strawberry plants inoculated with Azospirillum brasilense. World J Microbiol Biotechnol 26:265–272. https://doi.org/10.1007/s11274-009-0169-1
Petrasch S, Knapp SJ, van Kan JAL, Blanco-Ulate B (2019) Grey mold of strawberry, a devastating disease caused by the ubiquitous necrotrophic fungal pathogen Botrytis cinerea. Mol Plant Pathol 20:877–892. https://doi.org/10.1111/mpp.12794
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res 29:2002–2007. https://doi.org/10.1093/nar/29.9.e45
Pieterse CMJ, Van Loon LC (1999) Salicylic acid-independent plant defence pathways. Trends Plant Sci 4:52–58. https://doi.org/10.1016/S1360-1385(98)01364-8
Pieterse CMJ, Leon-Reyes A, Van Der Ent S, Van Wees SCM (2009) Networking by small-molecule hormones in plant immunity. Nat Chem Biol 5:308–316. https://doi.org/10.1038/nchembio.164
Pieterse CMJ, Zamioudis C, Berendsen RL, Weller DM, Van Wees SCM, Bakker PAHM (2014) Induced systemic resistance by beneficial microbes. Annu Rev Phytopathol 52:347–375. https://doi.org/10.1146/annurev-phyto-082712-102340
Qi J, Wang J, Gong Z, Zhou JM (2017) Apoplastic ROS signaling in plant immunity. Curr Opin Plant Biol 38:92–100. https://doi.org/10.1016/j.pbi.2017.04.022
Qi J, Song CP, Wang B, Wang B, Zhou J, Kangasjärvi J, Zhu JK, Gong Z (2018) Reactive oxygen species signaling and stomatal movement in plant responses to drought stress and pathogen attack. J Integr Plant Biol 60:805–826. https://doi.org/10.1111/jipb.12654
Ramos Solano B, Barriuso Maicas J, Pereyra De La Iglesia MT, Domenech J, Gutiérrez-Mañero FJ (2008) Systemic disease protection elicited by plant growth promoting rhizobacteria strains: relationship between metabolic responses, systemic disease protection, and biotic elicitors. Phytopathology 98:451–457. https://doi.org/10.1094/PHYTO-98-4-0451
Ruijter JM, Ramakers C, Hoogaars WMH, Karlen Y, Bakker O, van den Hoff MJB, Moorman AFM (2009) Amplification efficiency: linking baseline and bias in the analysis of quantitative PCR data. Nucleic Acids Res. https://doi.org/10.1093/nar/gkp045
Salazar SM, Castagnaro AP, Arias ME, Chalfoun N, Tonello U, Díaz-Ricci JC (2007) Induction of a defense response in strawberry mediated by an avirulent strain of Colletotrichum. Eur J Plant Pathol 117:109–122. https://doi.org/10.1007/s10658-006-9075-7
Salazar SM, Lovaisa NC, Guerrero Molina MF, Ragout AL, Kirschbaum DS, Díaz-Ricci JC, Pedraza RO (2012) Fruit yield of strawberry plants inoculated with Azospirillum brasilense RLC1 and REC3 under field conditions. Revta Agron NO Argent 32:63–66
Sambrook J, Russell DW (2006) SDS-Polyacrylamide gel electrophoresis of proteins. CSH Protocols. https://doi.org/10.1101/pdb.prot4540
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning. A laboratory manual. Cold Spring Harbor Laboratory Press, NY
Sandino T, López-Kleine L, López C, Marquínez X (2015) Characterization of the morphological response of susceptible and resistant varieties of cassava (Manihot esculenta Crantz) to vascular bacterial blight caused by Xanthomonas axonopodis pv manihotis. Summa Phytopathol 41(2):94–100
Shirokov A, Budanova A, Burygin G, Evseeva N, Matora L, Shchyogolev S (2020) Flagellin of polar flagellum from Azospirillum brasilense Sp245: isolation, structure, and biological activity. Int J Biol Macromol 147:1221–1227. https://doi.org/10.1016/j.ijbiomac.2019.10.092
Shu P, Min D, Ai W, Li J, Zhou J, Li Z, Zhang X, Shi Z, Sun Y, Jiang Y, Li F, Li X, Guo Y (2020) L-Arginine treatment attenuates postharvest decay and maintains quality of strawberry fruit by promoting nitric oxide synthase pathway. Postharv Biol Technol 168:111253. https://doi.org/10.1016/j.postharvbio.2020.111253
Steenhoudt O, Vanderleyden J (2000) Azospirillum, a free-living nitrogen-fixing bacterium closely associated with grasses: genetic, biochemical and ecological aspects. FEMS Microbiol Rev 24:487–506. https://doi.org/10.1016/S0168-6445(00)00036-X
Tikhonovich IA, Provorov NA (2007) Cooperation of plants and microorganisms: getting closer to the genetic construction of sustainable agro-systems. Biotechnol J 2:833–848. https://doi.org/10.1002/biot.200700014
Tomas-Grau RH, Requena-Serra FJ, Hael-Conrad V, Martínez-Zamora MG, Guerrero-Molina MF, Díaz-Ricci JC (2018) Soft mechanical stimulation induces a defense response against Botrytis cinerea in strawberry. Plant Cell Rep 37(2):239–250. https://doi.org/10.1007/s00299-017-2226-9
Torres MA, Dangl JL (2005) Functions of the respiratory burst oxidase in biotic interactions, abiotic stress and development. Curr Opin Plant Biol 8:397–403. https://doi.org/10.1016/j.pbi.2005.05.014
Tortora ML, Díaz-Ricci JC, Pedraza RO (2011) Azospirillum brasilense siderophores with antifungal activity against Colletotrichum acutatum. Arch Microbiol 193:275–286. https://doi.org/10.1007/s00203-010-0672-7
Tortora ML, Díaz-Ricci JC, Pedraza RO (2012) Protection of strawberry plants (Fragaria ananassa Duch.) against anthracnose disease induced by Azospirillum brasilense. Plant Soil 356:279–290. https://doi.org/10.1007/s11104-011-0916-6
Valenzuela-Riffo F, Zúñiga PE, Morales-Quintana L, Lolas M, Cáceres M, Figueroa CR (2020) Priming of defense systems and upregulation of MYC2 and JAZ1 genes after Botrytis cinerea inoculation in methyl jasmonate-treated strawberry fruits. Plants 9(4):447. https://doi.org/10.3390/plants9040447
Vanholme R, Demedts B, Morreel K, Ralph J, Boerjan W (2010) Lignin biosynthesis and structure. Plant Physiol 153:895–905. https://doi.org/10.1104/pp.110.155119
Vellicce GR, Coll Y, Castagnaro AP, Díaz-Ricci JC (2003) Transformation of a strawberry cultivar using a modified regeneration medium. Hortic Sci 38:277–280
Viejobueno J, Albornoz PL, Camacho M, de los Santos B, Martínez-Zamora MG, Salazar SM (2021) Protection of strawberry plants against charcoal rot disease (Macrophomina phaseolina) induced by Azospirillum brasilense. Agronomy 11(2):195. https://doi.org/10.3390/agronomy11020195
Viruega-Góngora VI, Acatitla-Jácome IS, Reyes-Carmona SR, Baca BE, Ramírez-Mata A (2020) Spatio-temporal formation of biofilms and extracellular matrix analysis in Azospirillum brasilense. FEMS Microbiol Lett 367(4):fnaa037. https://doi.org/10.1093/femsle/fnaa037
Zeng W, He SY (2010) A prominent role of the flagellin receptor flagellin-sensing 2 in mediating stomatal response to Pseudomonas syringae pv tomato DC3000 in Arabidopsis. Plant Physiol 153:1188–1198. https://doi.org/10.1104/pp.110.157016
Zhang Y, Li Y, He Y, Hu W, Zhang Y, Wang X, Tang H (2018) Identification of NADPH oxidase family members associated with cold stress in strawberry. FEBS Open Bio 8(4):593–605. https://doi.org/10.1002/2211-5463.12393
Zhulin IB, Armitage JP (1993) Motility, chemokinesis, and methylation-independent chemotaxis in Azospirillum brasilense. J Bacteriol 175:952–958. https://doi.org/10.1128/jb.175.4.952-958.1993
Zipfel C (2009) Early molecular events in PAMP-triggered immunity. Curr Opin Plant Biol 12:414–420. https://doi.org/10.1016/j.pbi.2009.06.003
Zipfel C, Robatzek S, Navarro L, Oakeley EJ, Jones JDG, Felix G, Boller T (2004) Bacterial disease resistance in Arabidopsis through flagellin perception. Nature 428:15–18. https://doi.org/10.1038/nature02485
Acknowledgements
We thank Dr. Fernando Campos-Casal for his help in the lignin surface three dimension plots. This work was partially supported by Secretaría de Ciencia, Arte y Tecnología, Universidad Nacional de Tucumán (Program A621), and by Agencia Nacional de Promoción Científica y Tecnológica (PICT-2017-0653; PICT-2019-02199). JME is fellow of Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET). PLA is member of Fundación Miguel Lillo and JCDR is member of CONICET.
Funding
This study was funded by Secretaría de Ciencia, Arte y Tecnología, Universidad Nacional de Tucumán, Program A621 (ROP), and by Agencia Nacional de Promoción Científica y Tecnológica, PICT-2017-0653(JCDR), PICT-2019-02199 (ROP).
Author information
Authors and Affiliations
Contributions
Concept of the study: ROP, JCDR, and BEB. Analysis and interpretation of manuscript data: all authors. Preparing a draft of the manuscript: JME, ROP, and JCDR. Final approval of manuscript: all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Axel Mithöfer.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Elías, J.M., Ramírez-Mata, A., Albornóz, P.L. et al. The Polar Flagellin of Azospirillum brasilense REC3 Induces a Defense Response in Strawberry Plants Against the Fungus Macrophomina phaseolina. J Plant Growth Regul 41, 2992–3008 (2022). https://doi.org/10.1007/s00344-021-10490-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-021-10490-4