Abstract
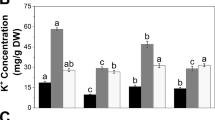
The present study was conducted in order to determine the response of two Egyptian flax cultivars (Sakha 102 and Sakha 105) to salt stress. To this end, seedlings of both cultivars were subjected to 150 mM NaCl condition, and the growth, Na+ and K+ concentrations, proline and hydrogen peroxide concentrations, and antioxidant enzyme activities were measured and compared with plants grown under non-stressed conditions. The results showed that salt stress reduced the overall growth of Sakha 102 by 38% as opposed to almost 50% reduction in Sakha 105. H2O2 significantly increased in Sakha 105, but remained unchanged in Sakha 102. The Na+ concentration in the leaf of Sakha 102 was 30% more than in the leaf of Sakha 105. The K+ concentrations were reduced by the same degree in all tissues of both plants and the Na+/K+ ratios were similar in both cultivars (generally > 1). The proline concentration was significantly more elevated in leaf, stem, and root of Sakha 102 (5.4, 2.5, and ten-fold, respectively) than in Sakha 105 (2.3, 2.3, and 4.0-folds). CAT activity markedly increased in the root of Sakha 102 (3.5-fold), but remained unchanged in Sakha 105. Taken together, these results suggest that Sakha 102 is more salt tolerant than Sakha 105, and that differences in proline concentration and ROS production under stress may account to a greater degree in the differential tolerance.






Similar content being viewed by others
Change history
23 June 2020
The original version of this article unfortunately contained an error in figure legends. The figure legends were misplaced due to an editing error. While Figs. 2, 3, 4, and 5 were shown at the correct position, the figure legends should be rearranged as follows. The original article has been corrected.
References
Abdelaziz MN, Xuan TD, Mekawy AMM, Wang H, Kahnh TD (2018) Relationship of salinity tolerance to Na+ exclusion, proline accumulation, and antioxidant enzyme activity in rice seedlings. Agriculture 8:166. https://doi.org/10.3390/agriculture8110166
Abogadallah GM (2010) Insights into the significance of antioxidative defense under salt stress. Plant Signal Behav 5:369–374. https://doi.org/10.4161/psb.5.4.10873
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59:206–216. https://doi.org/10.1016/j.envexpbot.2005.12.006
Assaha DV, Mekawy AM, Ueda A, Saneoka H (2015) Salinity-induced expression of HKT may be crucial for Na+ exclusion in the leaf blade of huckleberry (Solanum scabrum Mill.), but not of eggplant (Solanum melongena L.). Biochem Biophys Res Commun 460:416–421. https://doi.org/10.1016/j.bbrc.2015.03.048
Assaha DV, Ueda A, Saneoka H, Al-Yahyai R, Yaish MW (2017a) The role of Na+ and K+ transporters in salt stress adaptation in glycophytes. Front Physiol 8:1–19. https://doi.org/10.3389/fphys.2017.00509
Assaha DVM, Mekawy AMM, Liu L, Noori MS, Kokulan KS, Ueda A, Nagaoka T, Saneoka H (2017b) Na+ retention in the root is a key adaptive mechanism to low and high salinity in the glycophyte, Talinum paniculatum (Jacq.) Gaertn. (Portulacaceae). J Agron Crop Sci 203:56–67
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil. https://doi.org/10.1007/bf00018060
Bowler C, Montagu MV, Inze D (1992) Superoxide dismutase and stress tolerance. Ann Rev Plant Physiol Mol Biol 43:83–116. https://doi.org/10.1146/annurev.pp.43.060192.000503
Chakraborty K, Bose J, Shabala L, Shabala S (2016) Difference in root K+ retention ability and reduced sensitivity of K+-permeable channels to reactive oxygen species confer differential salt tolerance in three Brassica species. J Exp Bot 67:4611–4625. https://doi.org/10.1093/jxb/erw236
Chuamnakthong S, Nampei M, Ueda A (2019) Characterization of Na+ exclusion mechanism in rice under saline-alkaline stress conditions. Plant Sci 287:110171.https://doi.org/10.1016/j.plantsci.2019.110171
Cuin TA, Shabala S (2007) Compatible solutes reduce ROS-induced potassium efflux in Arabidopsis roots. Plant Cell Environ 30:875–885. https://doi.org/10.1111/j.1365-3040.2007.01674.x
El-Beltagi HS, Salama ZA, El Hariri DM (2008) Some biochemical markers for evaluation of flax cultivars under salt stress condition. J Nat Fibers 54:316–330. https://doi.org/10.1080/15440470802252487
Elsawy HIA, Mekawy AMM, Elhity MA, Abdel-Dayem SM, Abdelaziz MN, Assaha DVM, Ueda A, Saneoka H (2018) Differential responses of two Egyptian barley (Hordeum vulgare L.) cultivars to salt stress. Plant Physiol Biochem 127:425–435. https://doi.org/10.1016/j.plaphy.2018.04.012
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930. https://doi.org/10.1016/j.plaphy.2010.08.016
Hasegawa PM, Bressan RA, Zhu JK, Bohnert HJ (2000) Plant Cellular and Molecular Responses to High Salinity. Annu Rev Plant Physiol Plant Mol Biol 51:463–499. https://doi.org/10.1146/annurev.arplant.51.1.463
Kaur G, Asthir B (2015) Proline: a key player in plant abiotic stress tolerance. Biol Plant 59:609–619. https://doi.org/10.1007/s10535-015-0549-3
Mekawy AM, Assaha DV, Yahagi H, Tada Y, Ueda A, Saneoka H (2015) Growth, physiological adaptation, and gene expression analysis of two Egyptian rice cultivars under salt stress. Plant Physiol Biochem 87:17–25. https://doi.org/10.1016/j.plaphy.2014.12.007
Mekawy AMM, Abdelaziz MN, Ueda A (2018a) Apigenin pretreatment enhances growth and salinity tolerance of rice seedlings. Plant Physiol Biochem 130:94–104. https://doi.org/10.1016/j.plaphy.2018.06.036
Mekawy AMM, Assaha DVM, Munehiro R, Kohnishi E, Nagaoka T, Ueda A, Saneoka H (2018b) Characterization of type 3 metallothionein-like gene (OsMT-3a) from rice, revealed its ability to confer tolerance to salinity and heavy metal stresses. Environ Exp Bot 147:157–166. https://doi.org/10.1016/j.envexpbot.2017.12.002
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410. https://doi.org/10.1016/S1360-1385(02)02312-9
Munns R (2002) Comparative physiology of salt and water stress. Plant Cell Environ 25:239–250. https://doi.org/10.1046/j.0016-8025.2001.00808.x
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681. https://doi.org/10.1146/annurev.arplant.59.032607.092911
Rhee SG, Chang TC, Jeong W, Kang D (2010) Methods for detection and measurement of hydrogen peroxide inside and outside of cells. Mol Cells 29:539–549. https://doi.org/10.1007/s10059-010-0082-3
Sadak MS, Dawood MG (2014) Role of ascorbic acid and α tocopherol in alleviating salinity stress on flax plant (Linum usitatissimum L.). J Stress Physiol Biochem 10:93–111
Shabala L, Zhang J, Pottosin I, Bose J, Zhu M, Fuglsang AT, Velarde-Buendia A, Massart A, Hill CB, Roessner U, Bacic A, Wu H, Azzarello E, Pandolfi C, Zhou M, Poschenrieder C, Mancuso S, Shabala S (2016) Cell-type-specific H+-ATPase activity in root tissues enables K+ retention and mediates acclimation of barley (Hordeum vulgare) to salinity stress. Plant Physiol 172:2445–2458. https://doi.org/10.1104/pp.16.01347
Sharma P, Jha AB, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot 2012:1–27. https://doi.org/10.1155/2012/217037
Smirnoff N, Cumbes QJ (1989) Hydroxyl radical scavenging activity of compatible solutes. Phytochemistry 28:1057–1060. https://doi.org/10.1016/0031-9422(89)80182-7
Sun J, Dai S, Wang R, Chen S, Li N, Zhou X, Lu C, Shen X, Zheng X, Hu Z, Zhang Z, Song J, Xu Y (2009) Calcium mediates root K+/Na+ homeostasis in poplar species differing in salt tolerance. Tree Physiol 29:1175–1186. https://doi.org/10.1093/treephys/tpp048
Taïbi K, Taïbi F, Abderrahim LA, Ennajah A, Belkhodja M, Mulet JM (2016) Effect of salt stress on growth, chlorophyll content, lipid peroxidation and antioxidant defence systems in Phaseolus vulgaris L. S Afr J Bot 105:306–312. https://doi.org/10.1016/j.sajb.2016.03.011
Takagi H, Yamada S (2013) Roles of enzymes in anti-oxidative response system on three species of chenopodiaceous halophytes under NaCl-stress condition. Soil Sci Plant Nutr 59:603–611. https://doi.org/10.1080/00380768.2013.809600
Tester M, Davenport R (2003) Na+ tolerance and Na+ transport in higher plants. Ann Bot 91:503–527. https://doi.org/10.1093/aob/mcg058
Türkan I, Demiral T (2009) Recent developments in understanding salinity tolerance. Environ Exp Bot 67:2–9. https://doi.org/10.1016/j.envexpbot.2009.05.008
Ueda A, Kanechi M, Uno Y, Inagaki N (2003) Photosynthetic limitations of a halophyte sea aster (Aster tripolium L.) under water stress and NaCl stress. J Plant Res 116:65–70. https://doi.org/10.1007/s10265-002-0070-6
Ueda A, Shi W, Shimada T, Miyake H, Takabe T (2008) Altered expression of barley proline transporter causes different growth responses in Arabidopsis. Planta 227:277–286. https://doi.org/10.1007/s00425-007-0615-y
Ueda A, Yahagi H, Fujikawa Y, Nagaoka T, Esaka M, Calcaño M, González MM, Hernández Martich JD, Saneoka H (2013) Comparative physiological analysis of salinity tolerance in rice. Soil Sci Plant Nutr 59:896–903. https://doi.org/10.1080/00380768.2013.842883
Wang Z, Hobson N, Galindo L, Zhu S, Shi D, McDill J, Yang L, Hawkins S, Neutelings G, Datla R, Lambert G, Galbraith DW, Grassa CJ, Geraldes A, Cronk QC, Cullis C, Dash PK, Kumar PA, Cloutier S, Sharpe AG, Wong GK, Wang J, Deyholos MK (2012) The genome of flax (Linum usitatissimum) assembled de novo from short shotgun sequence reads. Plant J 72:461–473. https://doi.org/10.1111/j.1365-313X.2012.05093.x
Wangsawang T, Chuamnakthong S, Kohnishi E, Sripichitt P, Sreewongchai T, Ueda A (2018) A salinity tolerant japonica cultivar has Na+ exclusion mechanism at leaf sheaths through the function of a Na+ transporter OsHKT1;4 under salinity stress. J Agron Crop Sci 204:274–284. https://doi.org/10.1111/jac.12264
Wu H, Shabala L, Barry K, Zhou M, Shabala S (2013) Ability of leaf mesophyll to retain potassium correlates with salinity tolerance in wheat and barley. Physiol Plant 149:515–527. https://doi.org/10.1111/ppl.12056
Yaish MW (2015) Proline accumulation is a general response to abiotic stress in the date palm tree (Phoenix dactylifera L.). Genet Mol Res 14:9943–9950. https://doi.org/10.4238/2015.August.19.30
Yamaguchi T, Blumwald E (2005) Developing salt-tolerant crop plants: challenges and opportunities. Trends Plant Sci 10:615–620. https://doi.org/10.1016/j.tplants.2005.10.002
Acknowledgements
This research was supported by JSPS KAKENHI Grant Numbers 25850041, 15KK0283, 16K07643 to AU and Ministry of Higher Education and Scientific Research in Egypt to AMMM.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised: “the Figure legends 2, 3, 4, and 5 are rearranged”.
Rights and permissions
About this article
Cite this article
Mekawy, A.M.M., Assaha, D.V.M. & Ueda, A. Differential Salt Sensitivity of Two Flax Cultivars Coincides with Differential Sodium Accumulation, Biosynthesis of Osmolytes and Antioxidant Enzyme Activities. J Plant Growth Regul 39, 1119–1126 (2020). https://doi.org/10.1007/s00344-019-10048-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-019-10048-5