Abstract
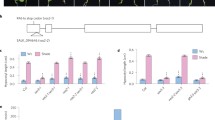
Sunflower hypocotyls elongate as light quality changes from the normal red to far-red (R/FR) ratio of sunlight to a lower R/FR ratio. This low R/FR ratio-induced elongation significantly increases endogenous concentrations of indole-3-acetic acid (IAA) and also of three gibberellins (GAs): GA20, GA1, and GA8. Of these, it is likely GA1 that drives low R/FR-induced growth. Brassinosteroids are also involved in shoot growth. Here we tested three R/FR ratios: high, normal, and low. Significant hypocotyl elongation occurred with this stepwise reduction in R/FR ratio, but endogenous castasterone concentrations in the hypocotyls remained unchanged. Brassinolide was also applied to the seedlings and significantly increased hypocotyl growth, though one that was uniform across all three R/FR ratios. Applied brassinolide increased hypocotyl elongation while significantly reducing (usually) levels of IAA, GA20, and GA8, but not that of GA1, which remained constant. Given the above, we conclude that endogenous castasterone does not mediate the hypocotyl growth that is induced by enriching FR light, relative to R light. Similarly, we conclude that the hypocotyl growth that is induced by applied brassinolide does not result from an interaction of brassinolide with changes in light quality. The ability of applied brassinolide to influence IAA, GA20, and GA8 content, yet have no significant effect on GA1, is hard to explain. One speculative hypothesis, though, could involve the brassinolide-induced reductions that occurred for endogenous IAA, given IAA’s known ability to differentially influence the expression levels of GA20ox, GA3ox, and GA2ox, key genes in GA biosynthesis.




Similar content being viewed by others
References
Asami T, Min YK, Nagata N, Yamagishi K, Takatsuto S, Fujioka S, Murofushi N, Yamaguchi I, Yoshida S (2000) Characterization of brassinazole, a triazole-type brassinosteroid biosynthesis inhibitor. Plant Physiol 123:93–99
Back TG, Baron DL, Luo W, Nakajima SK (1997) Concise, improved procedure for the synthesis of brassinolide and some novel side-chain analogues. J Org Chem 62:1179–1182
Bajguz A, Hayat S (2009) Effects of brassinosteroids on the plant responses to environmental stresses. Plant Physiol Biochem 47:1–8
Cohen D, Meudt WJ (1983) Investigations on the mechanisms of the brassinosteroid response. I. Indole-3-acetic acid metabolism and transport. Plant Physiol 72:691–694
Gamoh K, Sawamoto H, Kakatsuto S, Watabe Y, Arimoto H (1990) Ferroceneboronic acid as a derivatization reagent for the determination of brassinosteroids by high-performance liquid chromatography with electrochemical detection. J Chromatogr 15:227–231
Goda H, Shimada Y, Asami T, Fujioka S, Yoshida S (2002) Microarray analysis of brassinosteroid-regulated genes in Arabidopsis. Plant Physiol 130:1319–1334
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Cal Agr Exp Stat Circ 347:1–32
Katsumi M (1991) Physiological modes of brassinolide action in cucumber hypocotyl growth. In: Cutler HG, Yokota T, Adam G (eds) Brassinosteroids: chemistry, bioactivity and applications. American Chemical Society, Washington, pp 246–254
Kim TW, Hwang JY, Kim YS, Joo SH, Chang SC, Lee JS, Takatsuto S, Kim SK (2005) Arabidopsis CYP85A2, a cytochrome P450, mediates the Baeyer-Villiger oxidation of castasterone to brassinolide in brassinosteroid biosynthesis. Plant Cell 17:2397–2412
Kim TW, Lee SM, Joo SH, Yun HS, Lee Y, Kaufman PB, Kirakosyan A, Kim SH, Nam KH, Lee JS, Chang SC, Kim SK (2007) Elongation and gravitropic responses of Arabidopsis roots are regulated by brassinolide and IAA. Plant Cell Environ 30:679–689
Kinoshita T, Cano-Delgado A, Seto H, Hiranuma S, Fujioka S, Yoshida S, Chory J (2005) Binding of brassinosteroids to the extracellular domain of plant receptor kinase BRI1. Nature 433:167–171
Kurepin LV, Emery RJN, Pharis RP, Reid DM (2007a) The interaction of light quality and irradiance with gibberellins, cytokinins and auxin in regulating growth of Helianthus annuus hypocotyls. Plant Cell Environ 30:147–155
Kurepin LV, Walton LJ, Reid DM (2007b) Interaction of red to far red light ratio and ethylene in regulating stem elongation of Helianthus annuus. Plant Growth Regul 51:53–61
Kurepin LV, Emery RJN, Pharis RP, Reid DM (2007c) Uncoupling light quality from light irradiance effects in Helianthus annuus shoots: putative roles for plant hormones in leaf and internode growth. J Exp Bot 58:2145–2157
Kurepin LV, Walton LJ, Reid DM, Chinnappa CC (2010) Light regulation of endogenous salicylic acid levels in Helianthus annuus hypocotyls. Botany 88:668–674
Li JM, Nagpal P, Vitart V, McMorris TC, Chory J (1996) A role for brassinosteroids in light-dependent development of Arabidopsis. Science 272:398–401
Li J, Wen J, Lease KA, Doke JT, Tax FE, Walker JC (2002) BAK1, an Arabidopsis LRR receptor-like protein kinase, interacts with BRI1 and modulates brassinosteroid signaling. Cell 110:213–222
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Nagata N, Min YK, Nakano T, Asami T, Yoshida S (2000) Treatment of dark-grown Arabidopsis thaliana with a brassinosteroid-biosynthesis inhibitor, brassinazole, induces some characteristics of light-grown plants. Planta 211:781–790
Neff MM, Nguyen SM, Malancharuvil EJ, Fujioka S, Noguchi T, Seto H, Tsubuki M, Honda T, Takatsutoi S, Yoshida S, Chory J (1999) BAS1: A gene regulating brassinosteroid levels and light responsiveness in Arabidopsis. Proc Natl Acad Sci USA 96:15316–15323
Ngo P, Ozga JA, Reinecke DM (2002) Specificity of auxin regulation of gibberellin 20-oxidase gene expression in pea pericarp. Plant Mol Biol 49:439–448
O’Neill DP, Ross JJ (2002) Auxin regulation of the gibberellin pathway in pea. Plant Physiol 130:1974–1982
Ozga JA, Yu J, Reinecke DM (2003) Pollination-, development-, and auxin-specific regulation of gibberellin 3β-hydroxylase gene expression in pea fruit and seeds. Plant Physiol 131:1137–1146
Sakurai A (1999) Biosynthesis. In: Sakurai A, Yokota T, Clouse SD (eds) Brassinosteroids—steroidal plant hormones. Springer-Verlag, Tokyo, pp 91–111
Schultz L, Huub L, Kerckhoffs J, Klahre U, Yokota T, Reid JB (2001) Molecular characterization of the brassinosteroid-deficient lkb mutant in pea. Plant Mol Biol 47:491–498
Smith H (2000) Phytochromes and light signal perception by plants–an emerging synthesis. Nature 407:585–591
Symons GM, Reid JB (2003a) Hormone levels and response during de-etiolation in pea. Planta 216:422–431
Symons GM, Reid JB (2003b) Interactions between light and plant hormones during de-etiolation. J Plant Growth Regul 22:3–14
Takeno K, Pharis RP (1982) Brassinosteroid-induced bending of the leaf lamina of dwarf rice seedlings: an auxin-mediated phenomenon. Plant Cell Physiol 23:1275–1281
Tanaka K, Nakamura Y, Asami T, Yoshida S, Matsuo T, Okamoto S (2003) Physiological roles of brassinosteroids in early growth of Arabidopsis: brassinosteroids have a synergistic relationship with gibberellin as well as auxin in light-grown hypocotyl elongation. J Plant Growth Regul 22:259–271
Wang ZY, Seto H, Fujioka S, Yoshida S, Chory J (2001) BRI1 is a critical component of a plasma-membrane receptor for plant steroids. Nature 410:380–383
Wang L, Wang Z, Xu Y, Joo SH, Kim SK, Xue Z, Xu Z, Wang Z, Chong K (2009) OsGSR1 is involved in crosstalk between gibberellins and brassinosteroids in rice. Plant J 57:498–510
Yang GX, Jan A, Shen SH, Yazaki J, Ishikawa M, Shimatani Z, Kishimoto N, Kikuchi S, Matsumoto H, Komatsu S (2004) Microarray analysis of brassinosteroids- and gibberellin-regulated gene expression in rice seedlings. Mol Genet Genomics 271:468–478
Zhang S, Wei Y, Lu Y, Wang X (2009) Mechanisms of brassinosteroids interacting with multiple hormones. Plant Signal Behav 4:1117–1120
Acknowledgments
This work was funded by the Natural Sciences and Engineering Research Council of Canada Discovery grants to RPP and TGB.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kurepin, L.V., Joo, SH., Kim, SK. et al. Interaction of Brassinosteroids with Light Quality and Plant Hormones in Regulating Shoot Growth of Young Sunflower and Arabidopsis Seedlings. J Plant Growth Regul 31, 156–164 (2012). https://doi.org/10.1007/s00344-011-9227-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-011-9227-7