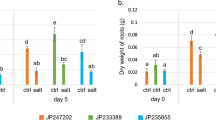
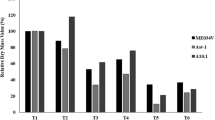
Abstract
The phytohormone abscisic acid (ABA) has been proposed to act as a mediator in plant responses to a range of stresses, including salt stress. Most studies of ABA response apply ABA as a single dose. This may not resemble the prolonged increasing endogenous ABA levels that can occur in association with slowly increasing salinity stresses in nature or field situations. Salt stress response based on method of ABA application was examined in four potato genotypes of varying salt stress resistance: the sensitive ABA-deficient mutant and its normal sibling, a resistant genotype line 9506, and commercial cultivar ‘Norland’ of moderate resistance. ABA was applied by root drench at 0, 50, 75, or 100 μM concentrations through a single dose, or by slowly increasing multiple ABA doses in a sand-based growing system under greenhouse conditions. Salt tolerance was then evaluated after 2 weeks of exposure to 150–180 mM NaCl stress. The method of ABA application had a marked effect on the responses to salt stress. Plant responses to the method of ABA application were differentiated according to (1) growth rate, (2) root water content, and (3) apparent shoot growth response. Under a single dose, growth rate increased in all genotypes under salt stress, whereas slowly increasing multiple ABA applications generally maintained stable growth rates except in the ABA-deficient mutant where there was an upward growth trend. Percent root water content was elevated only under slowly increasing multiple ABA doses in two genotypes, whereas none of the single-dose treatments induced any change. The single ABA dose enhanced vertical growth, whereas the slowly increasing multiple ABA dose applications enhanced lateral shoot growth. Because exogenous application is still an artificial system, endogenous ABA was supplied through grafting of ABA-deficient mutant scions onto rootstocks with known elevated ABA levels. Multiple exogenous ABA applications as low as 50 μM elicited similar shoot water content responses as grafting treatments without ABA application in the mutant genotype but had no effect on the ABA normal sibling. Shoot dry weight was significantly increased through grafting over all exogenous ABA treatments. Our study further indicates that the method of ABA application regime in itself can alter plant responses under salt stress and that certain application regimes may reflect responses to elevated endogenous levels of ABA.



Similar content being viewed by others
References
Ali M, Jensen CR, Mogensen VO, Bahrun A (1999) Drought adaptation of field grown wheat in relation to soil physical conditions. Plant Soil 208:149–159
Amzallag GN, Lerner HR, Poljakoff-Mayber A (1990) Exogenous ABA as a modulator of the response of Sorghum to high salinity. J Exp Bot 41:1529–1534
Borsani O, Cuartero J, Valpuesta V, Botella MA (2002) Tomato tos1 mutation identifies a gene essential for osmotic tolerance and abscisic acid sensitivity. Plant J 32:905–914
Cha-um S, Roytakul S, Sathung T, Maijandang A, Tengsiriwattana S, Kirdmanee C (2007) Exogenous glucose and abscisic acid pre-treatment in indica rice (Oryza sativa L. spp. indica) responses to sodium chloride salt stress. J Plant Sci 2:141–152
Chen CW, Yang YW, Lur HS, Tsai YG, Chang MC (2006a) A novel function of abscisic acid in the regulation of rice (Oryza sativa L.) root growth and development. Plant Cell Physiol 47:1–13
Chen S, Li J, Wang T, Wang S, Polle A, Hüttermann A (2002) Osmotic stress and ion-specific effects on xylem abscisic acid and the relevance to salinity tolerance in poplar. J Plant Growth Regul 21:224–233
Chen THH, Gusta LVG (1983) Abscisic acid induced freezing tolerance in cultured plant cells. Plant Physiol 73:71–75
Chen Y, Ji FF, Xie H, Liang JS (2006b) Overexpression of the regulator of G-protein signalling protein enhances ABA-mediated inhibition of root elongation and drought tolerance in Arabidopsis. J Exp Bot 57:2101–2110
Coleman JS, Schneider KM (1996) Evidence that abscisic acid does not regulate a centralized whole-plant response to low soil-resource availability. Oecologia 106:277–283
Cramer GR, Quarrie SA (2002) Abscisic acid is correlated with the leaf growth inhibition of four genotypes of maize differing in their response to salinity. Funct Plant Biol 29:111–115
De Jong H, Kawchuk LM, Coleman WK, Verhaeghe CA, Russell L, Burns VJ, Tremblay-Deveau E (2001) Development and characterization of an adapted form of droopy, a diploid potato mutant deficient in abscisic acid. Am J Potato Res 78:279–290
Etehadnia M, Waterer D, De Jong H, Tanino KK (2008) Impact of salt stress regime on salinity stress responses in salt sensitive and tolerant potato genotypes. J Plant Growth Regul March 26, 2008 online version
Fambrini A, Castagna F, Vecchia D, Degl’Innocenti E, Ranieri A, Vernieri P, Pardossi A, Guidi L, Rascio N, Pugliesi C (2004) Characterization of a pigment-deficient mutant of sunflower (Helianthus annuus L.) with abnormal chloroplast biogenesis, reduced PS II activity and low endogenous level of abscisic acid. Plant Sci 167:79–89
Fambrini M, Vernieri P, Toncelli ML, Rossi VD, Pugliesi C (1995) Characterization of a wilty sunflower (Helianthus annuus L.) mutant. III. Phenotypic interaction in reciprocal grafts from wilty mutant and wild-type plants. J Exp Bot 46:525–530
Foo E, Turnbull CGN, Beveridge CA (2001) Long-distance signaling and the control of branching in the rms1 mutant of pea. Plant Physiol 126:203–209
Freundl E, Steudle E, Hartung W (1998) Water uptake by roots of maize and sunflower affects the radial transport of abscisic acid and its concentration in the xylem. Planta 207:8–19
Freundl E, Steudle E, Hartung W (2000) Apoplastic transport of abscisic acid through roots of maize: effect of the exodermis. Planta 210:222–231
Fricke W, Akhiyarova G, Veselov D, Kudoyarova G (2004) Rapid and tissue-specific changes in ABA and in growth rate in response to salinity in barley leaves. J Exp Bot 55:1115–1123
Fricke W, Akhiyarova G, Wei W, Alexandersson E, Miller A, Kjellbom PO, Richardson A, Wojciechowski T, Schreiber L, Veselov D, Kudoyarova G, Volkov V (2006) The short-term growth response to salt of the developing barley leaf. J Exp Bot 57:1079–1095
Gómez-Cadenas A, Arbona V, Jacas J, Primo-Millo E, Talon M (2003) Abscisic acid reduces leaf abscission and increases salt tolerance in citrus plants. J Plant Growth Regul 21:234–240
Griffiths A, Jones HG, Tomos AD (1997) Applied abscisic acid, root growth and turgor pressure responses of roots of wild-type and the ABA-deficient mutant, Notabilis, of tomato. J Plant Physiol 151:60–62
Hartung W, Schraut D, Jiang F (2005) Physiology of abscisic acid (ABA) in roots under stress—a review of the relationship between root ABA and radial water and ABA flows. Aust J Agric Res 56:1253–1259
Holappa LD, Walker-Simmons MK, Ho THD, Riechers DE, Beckles DM, Jones RL (2005) A Triticum tauschii protein kinase related to wheat PKABA1 is associated with ABA signaling and is distributed between the nucleus and cytosol. J Cereal Sci 41:333–346
Hose E, Steudle E, Hartung W (2000) Abscisic acid and hydraulic conductivity of maize roots: a study using cell- and root-pressure probes. Planta 211:874–882
Jackson MB (2002) Long-distance signalling from roots to shoots assessed: the flooding story. J Exp Bot 53:175–181
Jia W, Wang Y, Zhang S, Zhang J (2002) Salt-stress-induced ABA accumulation is more sensitively triggered in roots than in shoots. J Exp Bot 53:2201–2206
Khadri M, Tejera NA, Lluch C (2006) Alleviation of salt stress in common bean (Phaseolus vulgaris) by exogenous abscisic acid supply. J Plant Growth Regul 25:110–119
Khadri M, Tejera NA, Lluch C (2007) Sodium chloride-ABA interaction in two common bean (Phaseolus vulgaris) cultivars differing in salinity tolerance. Environ Exp Bot 60:211–218
Kof EM, Vinogradova IA, Oorzhak AS, Karyagin VV, Kalibernaya ZV, Macháčková I, Kondykov IV, Chuvasheva ES (2006) ABA content in shoots and roots of pea mutants af and tl as related to their growth and morphogenesis. Russ J Plant Physiol 53:359–365
Koorneef M, Jorna ML, Brinkhorst-Van der Swan DLC, Karssen CM (1982) The isolation of abscisic acid (ABA) deficient mutants by selection of induced revertants in non-germinating gibberellin sensitive lines of Arabidopsis thaliana (L.). Heynh Theor Appl Genet 61:385–393
Lenzi A, Fambrini M, Barotti S, Pugliesi C, Vernieri P (1995) Seed germination and seedling growth in a wilty mutant of sunflower (Helianthus annuus L.): effect of abscisic acid and osmotic potential. Environ Exp Bot 35:427–434
Liu CL, Chen HP, Liu EE, Peng XX, Lu SY, Guo ZF (2003) Multiple tolerance of rice to abiotic stresses and its relationship with ABA accumulation. Acta Agro Sin 29:725–729
Ma S, Gong Q, Bohnert HJ (2006) Dissecting salt stress pathways. J Exp Bot 57:1097–1107
Mishra G, Zhang W, Deng F, Zhao J, Wang X (2006) A bifurcating pathway directs abscisic acid effects on stomatal closure and opening in Arabidopsis. Science 312:264–266
Montero E, Cabot C, Poschenrieder C, Barceló J (1998) Relative importance of osmotic-stress and ion-specific effects on ABA mediated inhibition of leaf expansion growth in Phaseolus vulgaris. Plant Cell Environ 21:54–62
Moons A, Gielen J, Vandekerckhove J, Van Der Straeten D, Gheysen G, Van Montagu M (1997) An abscisic-acid- and salt-stress-responsive rice cDNA from a novel plant gene family. Planta 202:443–454
Mulholland BJ, Taylor IB, Jackson AC, Thompson AJ (2003) Can ABA mediate responses of salinity stressed tomato? Environ Exp Bot 50:17–28
Neil SJ, Horgan R (1985) Abscisic acid production and water relations in wilty tomato mutants subjected to water deficiency. J Exp Bot 36:1222–1231
Quarrie SA (1982) Droopy: a wilty mutant of potato deficient in abscisic acid. Plant Cell Environ 5:23–26
Robichaud CS, Wong J, Sussex IM (1980) Control of in vitro growth of viviparous embryo mutants of maize by abscisic acid. Dev Genet 1:325–330
Ruan X, Ruggiero B, Koiwa H, Manabe Y, Quist TM, Inan G, Saccardo F, Joly RJ, Hasegawa PM, Bressan RA, Maggio A (2004) Uncoupling the effects of abscisic acid on plant growth and water relations. analysis of sto1/nced3, an abscisic acid-deficient but salt stress-tolerant mutant in Arabidopsis. Plant Physiol 136(2):3134–3147
Salisbury FB, Ross CW (1992) Plant physiology, 4th edn. Wordsworth, California, p 682
Sansberro PA, Mroginski LA, Bottini R (2004) Foliar sprays with ABA promote growth of Ilex paraguariensis by alleviating diurnal water stress. J Plant Growth Regul 42:105–111
Sharma N, Abrams SR, Waterer DR (2005) Uptake, movement, activity, and persistence of an abscisic acid analog (8′ acetylene ABA methyl ester) in marigold and tomato. J Plant Growth Regul 24:28–35
Sharp RE, LeNoble ME (2002) ABA, ethylene and the control of shoot and root growth under water stress. J Exp Bot 53:33–37
Sharp RE, LeNoble ME, Else MA, Thorne ET, Gherardi F (2000) Endogenous ABA maintains shoot growth in tomato independently of effects on plant water balance: evidence for an interaction with ethylene. J Exp Bot 51:1575–1584
Shaterian J, Georges F, Hussain A, Waterer D, De Jong H, Tanino KK (2005a) Root to shoot communication and abscisic acid in calreticulin (CR) gene expression and salt-stress tolerance in grafted diploid potato clones. Environ Exp Bot 53:323–332
Shaterian J, Waterer D, De Jong H, Tanino KK (2005b) Differential stress responses to NaCl salt application in early- and late-maturing diploid potato (Solanum sp.) clones. Environ Exp Bot 54:202–212
Sibole JV, Montero E, Cabot C, Poschenrieder C, Barceló J (2000) Relationship between carbon partitioning and Na+, Cl− and ABA allocation in fruits of salt-stressed bean. J Plant Physiol 157:637–642
Srivastava S, Rahman MH, Shah S, Kav NNV (2006) Constitutive expression of the pea ABA-responsive 17 (ABR17) cDNA confers multiple stress tolerance in Arabidopsis thaliana. Plant Biotech J 4:529–549
Suzuki M, Settles MA, Tseung CW, Li QB, Latshaw S, Wu S, Porch TG, Schmelz EA, James MG, McCarty DR (2006) The maize viviparous15 locus encodes the molybdopterin synthase small subunit. Plant J 45:264–274
Tanino KK, Matthews L, Wasif E, Shenouda A, Gilliland B, Nair MPM (2002) Abscisic acid and estrogen effects on flowering and phenotypic traits in melon (Cucumis melo cv ‘Earligold’) and pumpkin (Cucurbita pepo cv ‘Baby bear’). Acta Hort 588:97–101
Tenhunen JD, Hanano R, Abril M, Weiler EW, Hartung W (1994) Above- and below-ground environmental influences on leaf conductance of Ceanothus thyrsiflorus growing in a chaparral environment: drought response and the role of abscisic acid. Oecologia 99:306–314
Thomas DS, Eamus D (1999) The influence of predawn leaf water potential on stomatal responses to atmospheric water content at constant Ci and on stem hydraulic conductance and foliar ABA concentrations. J Exp Bot 50:243–251
Thompson AJ, Andrews J, Mulholland BJ, McKee JMT, Hilton HW, Horridge JS, Farquhar GD, Smeeton RC, Smillie IRA, Black CR, Taylor IB (2007) Overproduction of abscisic acid in tomato increases transpiration efficiency and root hydraulic conductivity and influences leaf expansion. Plant Physiol 143:1905–1917
Udovenko GV (1995) Physiological basis of plant breeding, vol. 2, pt. 2. St Petersburg: Itz. VIR [in Russian]
Umezawa T, Okamoto M, Kushiro T, Nambara E, Oono Y, Seki M, Kobayashi M, Koshiba T, Kamiya Y, Shinozaki K (2006) CYP707A3, a major ABA 8′-hydroxylase involved in dehydration and rehydration response in Arabidopsis thaliana. Plant J 46:171–182
Vysotskaya LB, Arkhipova TN, Timergalina LN, Dedov AV, Veselov SY, Kudoyarova GR (2004a) Effect of partial root excision on transpiration, root hydraulic conductance and leaf growth in wheat seedlings. Plant Physiol Biochem 42:251–255
Vysotskaya LB, Kudoyarova GR, Veselov S, Jones HG (2004b) Unusual stomatal behaviour on partial root excision in wheat seedlings. Plant Cell Environ 27:69–77
Wahome PK, Jesch HH, Grittner I (2001) Mechanisms of salt stress tolerance in two rose rootstocks: Rosa chinensis ‘Major’ and R. rubiginosa. Sci Hort 87:207–216
Wang TL, Donkin ME, Martin ES (1984) The physiology of a wilty pea: abscisic acid production under water stress. J Exp Bot 35:1222–1232
Wang Z, Huang B, Xu Q (2003) Effects of abscisic acid on drought responses of Kentucky bluegrass. J Amer Soc Hort Sci 128:36–41
Wright STC (1978) Phytohormones and stress phenomena. In: Letham DS, Goodwin PB, Higgins TJV (eds) Phytohormones and related compounds: a comprehensive treatise, vol. 2. Elsevier /North Holland Biomedical Press, Amsterdam, pp 495–536
Acknowledgments
This study was funded by Iranian Agricultural Research Organization and Natural Resources and University of Saskatchewan. The greenhouse assistance by Tom Ward and John Peters and secretarial help by Mary Lee, Sharon Steven, and Carolyn Ouellet are appreciated. Finally, the original donation of potato tubers by Heilke de Jong is greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Etehadnia, M., Waterer, D.R. & Tanino, K.K. The Method of ABA Application Affects Salt Stress Responses in Resistant and Sensitive Potato Lines. J Plant Growth Regul 27, 331–341 (2008). https://doi.org/10.1007/s00344-008-9060-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-008-9060-9