Abstract
With more than 450 studied species, coral reproduction is a well-known research field. However, the vast majority of coral reproduction research has focused exclusively on shallow reefs. The incentive for the present study was: (1) the recent accelerated global degradation of coral reefs; (2) the growing interest in mesophotic coral ecosystems (MCEs; 30–120 m depth) and their potential to serve as a larval source for shallow reefs; and (3) the lack of information on MCE coral reproduction. Here, we compare the reproduction and ecology of the depth-generalist coral Paramontastraea peresi between shallow (5–10 m) and mesophotic (40–45 m) habitats in the Gulf of Eilat/Aqaba, Red Sea. Field surveys were conducted to assess the living cover, abundance, and size frequency distribution of P. peresi. Four to six colonies from each habitat were sampled monthly between April 2015 and January 2017, and the gametogenesis cycles, fecundity, and oocyte sizes were measured. The reproductive cycle in the MCEs was shorter than in the shallow reef. Despite having larger polyps, the mesophotic colonies contained significantly smaller and fewer oocytes per polyp. In spite of the relatively stable environmental conditions of the MCEs, which may contribute to coral survival, scarcity of sunlight is probably a major energetic impediment to investment in reproduction by P. peresi at mesophotic depths. Further intensive reproductive studies in mesophotic reefs are thus required to assess the ability of corals in this environment to reproduce and constitute a larval source for depleted shallow-water reefs.








Similar content being viewed by others
References
Akkaynak D, Treibitz T, Shlesinger T, Tamir R, Loya Y, Iluz D (2017) What is the space of attenuation coefficients in underwater computer vision? Proceedings of the IEEE Computer Vision and Pattern Recognition conference (CVPR), 21–26 July 2017, Honolulu, Hawaii, pp 4931–4940
Anthony KRN, Hoegh-Guldberg O (2003) Variation in coral photosynthesis, respiration and growth characteristics in contrasting light microhabitats: an analogue to plants in forest gaps and understoreys. Funct Ecol 17:246–259
Babcock RC, Bull G, Harrison PL, Heyward A, Oliver J, Wallace C, Willis B (1986) Synchronous spawnings of 105 scleractinian coral species on the Great Barrier Reef. Mar Biol 90:379–394
Baird AH, Guest JR, Willis BL (2009) Systematic and biogeographical patterns in the reproductive biology of scleractinian corals. Annu Rev Ecol Evol Syst 40:551–571
Bak RP, Nieuwland G, Meesters EH (2005) Coral reef crisis in deep and shallow reefs: 30 years of constancy and change in reefs of Curacao and Bonaire. Coral Reefs 24:475–479
Bongaerts P, Ridgway T, Sampayo E, Hoegh-Guldberg O (2010) Assessing the ‘deep reef refugia’ hypothesis: focus on Caribbean reefs. Coral Reefs 29:309–327
Bongaerts P, Riginos C, Brunner R, Englebert N, Smith SR, Hoegh-Guldberg O (2017) Deep reefs are not universal refuges: reseeding potential varies among coral species. Sci Adv 3:e1602373
Cox EF, Ward S (2002) Impact of elevated ammonium on reproduction in two Hawaiian scleractinian corals with different life history patterns. Mar Pollut Bull 44:1230–1235
Eyal G, Eyal-Shaham L, Cohen I, Tamir R, Ben-Zvi O, Sinniger F, Loya Y (2016) Euphyllia paradivisa, a successful mesophotic coral in the northern Gulf of Eilat/Aqaba, Red Sea. Coral Reefs 35:91–102
Eyal-Shaham L, Eyal G, Tamir R, Loya Y (2016) Reproduction, abundance and survivorship of two Alveopora spp. in the mesophotic reefs of Eilat, Red Sea. Sci Rep 6:20964
Faure G, Pichon M (1978) Description de Favites peresi, nouvelle espèce de Scleractiniaire hermatypique de l’océan Indien (Cnidaria, Anthozoa, Scleractinia). Bulletin du Muséum National d’Histoire Naturelle, Paris, 3e série 513:107–127
Fricke HW, Schuhmacher H (1983) The depth limits of Red Sea stony corals: an ecophysiological problem (a deep diving survey by submersible). Mar Ecol 4:163–194
Fryxell PA (1957) Mode of reproduction of higher plants. Bot Rev 23:135–233
Glynn PW (1993) Coral reef bleaching: ecological perspectives. Coral Reefs 12:1–17
Glynn PW (1996) Coral reef bleaching: facts, hypotheses and implications. Glob Chang Biol 2:495–509
Harrison PL (2011) Sexual reproduction of scleractinian corals. In: Dubinsky Z, Stambler N (eds) Coral reefs: an ecosystem in transition. Springer, Netherlands, pp 59–85
Harrison PL, Wallace CC (1990) Reproduction, dispersal and recruitment of scleractinian corals. In: Dubinsky Z (ed) Coral reefs, ecosystems of the world 25. Elsevier, Amsterdam, pp 133–207
Holstein DM, Smith TB, Gyory J, Paris CB (2015) Fertile fathoms: deep reproductive refugia for threatened shallow corals. Sci Rep 5:12407
Holstein DM, Smith TB, Paris CB (2016) Depth-independent reproduction in the reef coral Porites astreoides from shallow to mesophotic zones. PLoS One 11:e0146068
Huang D, Benzoni F, Fukami H, Knowlton N, Smith ND, Budd AF (2014) Taxonomic classification of the reef coral families Merulinidae, Montastraeidae, and Diploastraeidae (Cnidaria: Anthozoa: Scleractinia). Zool J Linn Soc 171:277–355
Hughes TP, Connell JH (1999) Multiple stressors on coral reefs: a long-term perspective. Limnol Oceanogr 44:932–940
Hughes TP, Kerry JT, Álvarez-Noriega M, Álvarez-Romero JG, Anderson KD, Baird AH, Babcock RC, Beger M, Bellwood DR, Berkelmans R, Bridge TCL, Butler I, Byrne M, Cantin NE, Comeau S, Connolly SR, Cumming GS, Dalton SJ, Diaz-Pulido G, Eakin M, Figueira W, Gilmour J, Harrison HB, Heron SF, Hoey AS, Hobbs J-PA, Hoogenboom MO, Kennedy EV, Kuo C-Y, Lough JM, Lowe RJ, Liu G, McCulloch MT, Malcolm H, McWilliam M, Pandolfi JM, Pears R, Pratchett MS, Schoepf V, Simpson T, Skirving W, Sommer B, Torda G, Wachenfeld D, Willis BL, Wilson SK (2017) Global warming and recurrent mass bleaching of corals. Nature 543:373–377
Huston M (1985) Variation in coral growth rates with depth at Discovery Bay, Jamaica. Coral Reefs 4:19–25
Jerlov NG (1976) Marine optics. Elsevier, Amsterdam, p 231
Jokiel PL, Ito RY, Liu PM (1985) Night irradiance and synchronization of lunar release of planula larvae in the reef coral Pocillopora damicornis. Mar Biol 88:167–174
Kahng S, Copus J, Wagner D (2014) Recent advances in the ecology of mesophotic coral ecosystems (MCEs). Curr Opin Environ Sustain 7:72–81
Kojis BL, Quinn NJ (1981) Aspects of sexual reproduction and larval development in the shallow water hermatypic coral Goniastrea australensis (Edwards and Haime, 1857). Bull Mar Sci 31:558–573
Kojis BL, Quinn NJ (1984) Seasonal and depth variation in fecundity of Acropora palifera at two reefs in Papua New Guinea. Coral Reefs 3:165–172
Kramarsky-Winter E, Loya Y (1998) Reproductive strategies of two fungiid corals from the northern Red Sea: environmental constraints? Mar Ecol Prog Ser 174:175–182
Lesser MP, Slattery M, Leichter JJ (2009) Ecology of mesophotic coral reefs. J Exp Mar Bio Ecol 375:1–8
Loya Y (1975) Possible effects of water pollution on the community structure of Red Sea corals. Mar Biol 29:177–185
Loya Y, Rinkevich B (1979) Abortion effect in corals induced by oil pollution. Mar Ecol Prog Ser 1:77–80
Loya Y, Rinkevich B (1980) Effects of oil pollution on coral reef communities. Mar Ecol Prog Ser 3:167–180
Loya Y, Sakai K (2008) Bidirectional sex change in mushroom stony corals. Proc R Soc Lond B Biol Sci 275:2335–2343
Loya Y, Lubinevsky H, Rosenfeld M, Kramarsky-Winter E (2004) Nutrient enrichment caused by in situ fish farms at Eilat, Red Sea is detrimental to coral reproduction. Mar Pollut Bull 49:344–353
Loya Y, Eyal G, Treibitz T, Lesser MP, Appeldoorn R (2016) Theme section on mesophotic coral ecosystems: advances in knowledge and future perspectives. Coral Reefs 35:1–9
Madin JS, Anderson KD, Andreasen MH, Bridge TC, Cairns SD, Connolly SR, Darling ES, Diaz M, Falster DS, Franklin EC, Gates RD, Hoogenboom MO, Huang D, Keith SA, Kosnik MA, Kuo CY, Lough JM, Lovelock CE, Luiz O, Martinelli J, Mizerek T, Pandolfi JM, Pochon X, Pratchett MS, Putnam HM, Roberts TE, Stat M, Wallace CC, Widman E, Baird AH (2016) The Coral Trait Database, a curated database of trait information for coral species from the global oceans. Sci Data 3:160017
Mass T, Einbinder S, Brokovich E, Shashar N, Vago R, Erez J, Dubinsky Z (2007) Photoacclimation of Stylophora pistillata to light extremes: metabolism and calcification. Mar Ecol Prog Ser 334:93–102
Nir O, Gruber D, Einbinder S, Kark S, Tchernov D (2011) Changes in scleractinian coral Seriatopora hystrix morphology and its endocellular Symbiodinium characteristics along a bathymetric gradient from shallow to mesophotic reef. Coral Reefs 30:1089
Nozawa Y (2012) Annual variation in the timing of coral spawning in a high-latitude environment: influence of temperature. Biol Bull 222:192–202
Pandolfi JM, Connolly SR, Marshall DJ, Cohen AL (2011) Projecting coral reef futures under global warming and ocean acidification. Science 333:418–422
Pennisi E (2007) Spawning for a better life. Science 318:1712–1717
Prasetia R, Sinniger F, Harii S (2016) Gametogenesis and fecundity of Acropora tenella (Brook 1892) in a mesophotic coral ecosystem in Okinawa, Japan. Coral Reefs 35:53–62
Prasetia R, Sinniger F, Hashizume K, Harii S (2017) Reproductive biology of the deep brooding coral Seriatopora hystrix: implications for shallow reef recovery. PLoS One 12:e0177034
Rapuano H, Brickner I, Shlesinger T, Meroz-Fine E, Tamir R, Loya Y (2017) Reproductive strategies of the coral Turbinaria reniformis in the northern Gulf of Aqaba (Red Sea). Sci Rep 7:42670
Richmond RH, Hunter CL (1990) Reproduction and recruitment of corals: comparisons among the Caribbean, the Tropical Pacific, and the Red Sea. Mar Ecol Prog Ser 60:185–203
Rinkevich B, Loya Y (1979a) Reproduction of the Red Sea coral Stylophora pistillata. 1. Gonads and planulae. Mar Ecol Prog Ser 1:133–144
Rinkevich B, Loya Y (1979b) The reproduction of the Red Sea coral Stylophora pistillata. II. Synchronization in breeding and seasonality of planulae shedding. Mar Ecol Prog Ser 1:145–152
Rinkevich B, Loya Y (1987) Variability in the pattern of sexual reproduction of the coral Stylophora pistillata at Eilat, Red Sea: a long-term study. Biol Bull 173:335–344
Sakai K (1998) Effect of colony size, polyp size, and budding mode on egg production in a colonial coral. Biol Bull 195:319–325
Shlesinger Y, Loya Y (1985) Coral community reproductive patterns: Red Sea versus the Great Barrier Reef. Science 228:1333–1335
Shlesinger T, Loya Y (2016) Recruitment, mortality, and resilience potential of scleractinian corals at Eilat, Red Sea. Coral Reefs 35:1357–1368
Shlesinger Y, Goulet T, Loya Y (1998) Reproductive patterns of scleractinian corals in the northern Red Sea. Mar Biol 132:691–701
Slattery M, Lesser MP, Brazeau D, Stokes MD, Leichter JJ (2011) Connectivity and stability of mesophotic coral reefs. J Exp Mar Bio Ecol 408:32–41
Smith TB, Gyory J, Brandt ME, Miller WJ, Jossart J, Nemeth RS (2016) Caribbean mesophotic coral ecosystems are unlikely climate change refugia. Glob Chang Biol 22:2756–2765
Spirlet C, Grosjean P, Jangoux M (2000) Optimization of gonad growth by manipulation of temperature and photoperiod in cultivated sea urchins, Paracentrotus lividus (Lamarck) (Echinodermata). Aquaculture 185:85–99
Szmant AM (1986) Reproductive ecology of Caribbean reef corals. Coral Reefs 5:43–53
Szmant-Froelich AM (1985) The effect of colony size on the reproductive ability of the Carribean coral Montastrea annularis (Ellis and Solander). Proc 5th Int Coral Reef Symp 4:295–300
Veron JEN (2000) The corals of the world, vol 3. Townsville, Australian Institute of Marine Science, p 166
Vize PD (2006) Deepwater broadcast spawning by Montastraea cavernosa, Montastraea franksi, and Diploria strigosa at the Flower Garden Banks, Gulf of Mexico. Coral Reefs 25:169–171
Wilkinson C, Souter D (2008) The status of Caribbean coral reefs after bleaching and hurricanes in 2005. Coral Reef Monitoring Network and Australian Institute of Marine Science, Townsville
Willis BL, Babcock RC, Harrison PL, Oliver JK, Wallace CC (1985) Patterns in the mass spawning of corals on the Great Barrier Reef from 1981 to 1984. Proc 5th Int Coral Reef Symp 4:343–348
Wolgast LJ, Zeide B (1983) Reproduction of trees in a variable environment. Bot Gaz 144:260–262
Acknowledgements
We thank the Interuniversity Institute for Marine Sciences at Eilat (IUI) for the logistical support. We are indebted to R. Tamir, O. Ben-Zvi, H. Rapuano, M. Grinblat, L. Eyal-Shaham, G. Eyal, and I. Brickner for their help with the field and laboratory work. This study was partially supported by the Israel Science Foundation (ISF) No. 341/12 and the US Middle East Regional Cooperation (MERC) Program Agency for International Development (MERC/USAID) No. M32-037 to Y.L.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Biology Editor Dr. Anastazia Banaszak
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
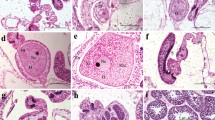
Photomicrograph of oogenesis stages II–V and spermary stages II and III in Paramontastraea peresi (stages defined in ESM Table S1). (a) Stage II oocytes (b) Stage III oocytes (c) Stage III and IV oocytes (d) Stage IV and V oocytes (e) Stage II spermaries (f) Stage III spermaries. NUC: nucleus; NUL: nucleolus; RM: reservoir materials FLG: flagella (TIFF 6818 kb)
Rights and permissions
About this article
Cite this article
Feldman, B., Shlesinger, T. & Loya, Y. Mesophotic coral-reef environments depress the reproduction of the coral Paramontastraea peresi in the Red Sea. Coral Reefs 37, 201–214 (2018). https://doi.org/10.1007/s00338-017-1648-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-017-1648-8