Abstract
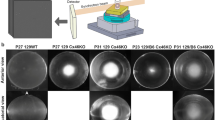
Scheimpflug imaging has recently been established for in vivo imaging of the anterior eye segment and quantitative determination of lens transparency in the mouse. This enables more effective investigations of cataract formation with the mouse model, including longitudinal studies. In order to enable recognition of disease-associated irregularities, we performed Scheimpflug measurements with the common laboratory inbred lines C57BL/6J, C3HeB/FeJ, FVB/NCrl, BALB/cByJ, and 129/SvJ in a period between 2 and 12 months of age. C57BL/6J mice showed lowest mean lens densities during the test period. Progressive cortical lens opacification was generally observed, with the earliest onset in C57BBL/6J, C3HeB/FeJ, and 129/SvJ, between 2 and 6 months after birth. Moreover, lenses of these inbred lines developed nuclear opacities. Calculated mean lens density significantly increased between 6 and 12 months of age in all inbred strains except 129/SvJ. Lens densities (and the corresponding standard deviations) of FVB/NCrl and 129/SvJ increased most likely because of differences in the genetic background. Albinism as confounder might be excluded since the albino Balb/cByJ mice are more similar to the C57BL/6J or C3Heb/FeJ mice. We further identified strain-specific anterior lens opacities (C57BL/6J) and cloudy corneal lesions (C57BL/6J, FVB/NCrl, and BALB/cByJ) at later stages. In conclusion, our results indicate that there are lifelong opacification processes in the mouse lens. The highest lens transparency and a dark coat color, which prevents interference from light reflections, make mice with the C57BL/6J background most suitable for cataract research by Scheimpflug imaging. We show that lens densitometry by Scheimpflug imaging in mouse eyes can resolve differences of less than 1 %, making it possible to detect differences in cataract development in different mouse strains, even if they are small.






Similar content being viewed by others
References
Alizadeh A, Clark J, Seeberger T, Hess J, Blankenship T, FitzGerald PG (2004) Characterization of a mutation in the lens-specific CP49 in the 129 strain of mouse. Invest Ophthalmol Vis Sci 45:884–891
Cho YK, Chang HS, La TY, Ji D, Kim H, Choi JA, Kim MS (2010) Anterior segment parameters using Pentacam and prediction of corneal endothelial cell loss after cataract surgery. Korean J Ophthalmol 24:284–290
Churchill A, Graw J (2011) Clinical and experimental advances in congenital and paediatric cataracts. Philos Trans R Soc Lond B Biol Sci 366:1234–1249
Dalke C, Puk O, Neuhäuser-Klaus A, Favor J, Graw J (2006) Eye disorders. In: Hrabĕ de Angelis M, Chambon P, Brown S (eds) Standards of mouse model phenotyping. Wiley, Weinheim, pp 283–309
Fuchs H, Gailus-Durner V, Adler T, Aguilar-Pimentel JA, Becker L et al (2011) Mouse phenotyping. Methods 53:120–135
Graw J (2009) Mouse models of cataract. J Genet 88:469–486
Grewal DS, Brar GS, Grewal SP (2009) Correlation of nuclear cataract lens density using Scheimpflug images with Lens Opacities Classification System III and visual function. Ophthalmology 116:1436–1443
Gupta R, Asomugha CO, Srivastava OP (2011) The common modification in alphaA-crystallin in the lens, N101D, is associated with increased opacity in a mouse model. J Biol Chem 286:11579–11592
Johnson GJ, Foster A (2004) Prevalence, incidence and distribution of visual impairment. In: Johnson GJ, Minassian DC, Weale RA, West SK (eds) The epidemiology of eye disease. Arnold, London, pp 3–28
Jun G, Guo H, Klein BE, Klein R, Wang JJ, Mitchell P et al (2009) EPHA2 is associated with age-related cortical cataract in mice and humans. PLoS Genet 5:e1000584
Magalhães FP, Costa EF, Cariello AJ, Rodrigues EB, Hofling-Lima AL (2011) Comparative analysis of the nuclear lens opalescence by the Lens Opacities Classification System III with nuclear density values provided by Oculus Pentacam: a cross-section study using Pentacam Nucleus Staging software. Arq Bras Oftalmol 74:110–113
Michael R, Bron AJ (2011) The ageing lens and cataract: a model of normal and pathological aging. Philos Trans R Soc Lond B Biol Sci 366:1278–1292
Okano Y, Asada M, Fujimoto A, Ohtake A, Murayama K, Hsiao KJ et al (2001) A genetic factor for age-related cataract: identification and characterization of a novel galactokinase variant, “Osaka”, in Asians. Am J Hum Genet 68:1036–1042
Pal-Gosh S, Tadvalkar G, Jurjus RA, Zieske JD, Stepp MA (2008) BALB/c and C57BL6 mouse strains vary in their ability to heal corneal epithelial debridement wounds. Exp Eye Res 87:478–486
Pendergrass W, Penn P, Possin D, Wolf N (2005) Accumulation of DNA, nuclear and mitochondrial debris, and ROS at sites of age-related cortical cataract in mice. Invest Ophthalmol Vis Sci 46:4661–4670
Pendergrass W, Zitnik G, Urfer SR, Wolf N (2011) Age-related retention of fiber cell nuclei and nuclear fragments in the lens of multiple species. Mol Vis 17:2672–2684
Puk O, Dalke C, Calzada-Wack J, Ahmad N, Klaften M et al (2009) Reduced corneal thickness and enlarged anterior chamber depth in a novel ColIIIa2G257D mutant mouse. Invest Ophthalmol Vis Sci 50:5653–5661
Puk O, Ahmad N, Wagner S, Hrabé de Angelis M, Graw J (2011a) Microphakia and congenital cataract formation in a novel Lim2(C51R) mutant mouse. Mol Vis 17:1164–1171
Puk O, Möller G, Geerlof A, Krowiorz K, Ahmad N, Wagner S, Adamski J, Hrabé de Angelis M, Graw J (2011b) The pathologic effect of a novel neomorphic Fgf9(Y162C) allele is restricted to decreased vision and retarded lens growth. PLoS One 6:e23678. doi:10.1371/journal.pone.0023678
Regenfuss B, Onderka J, Bock F, Hos D, Maruyama K, Cursiefen C (2010) Genetic heterogeneity of lymphangiogenesis in different mouse strains. Am J Pathol 177:501–510
Sandilands A, Wang X, Hutcheson AM, James J, Prescott AR, Wegener A, Pekny M, Gong X, Quinlan RA (2004) Bsfp2 mutation found in 129 strains causes the loss of CP49’ and induces vimentin-dependent changes in the lens fibre cell cytoskeleton. Exp Eye Res 78:875–889
Santana A, Waiswo M (2011) The genetic and molecular basis of congenital cataract. Arq Bras Oftalmol 74:136–142
Shi Y, Shi X, Jin Y, Miao A, Bu L, He J, Jiang H, Lu Y, Kong X, Hu L (2008) Mutation screening of HSF4 in 150 age-related cataract patients. Mol Vis 14:1850–1855
Simirskii VN, Lee RS, Wawrousek EF, Duncan MK (2006) Inbred FVB/N mice are mutant at the cp49/Bsfp2 locus and lack beaded filament proteins in the lens. Invest Ophthalmol Vis Sci 47:4931–4934
Thaung C, West K, Clark BJ, McKie L, Morgan JE et al (2002) Novel ENU-induced mutations in the mouse: models for human eye disease. Hum Mol Genet 11:755–767
Ueda Y, Duncan MK, David LL (2002) Lens proteomics: the accumulation of crystallin modifications in the mouse lens with age. Invest Ophthalmol Vis Sci 43:205–215
West SK (2000) Looking forward to 20/20: a focus on the epidemiology of eye diseases. Epidemiol Rev 22:64–70
Wolf N, Pendergrass W, Singh N, Swisshelm K, Schwartz J (2008) Radiation cataracts: mechanisms involved in their long delayed occurrence but then rapid progression. Mol Vis 14:274–285
Zhang Y, Zhang L, Sun D, Li Z, Wang L, Liu P (2011) Genetic polymorphisms of superoxide dismutases, catalase, and glutathione peroxidase in age-related cataract. Mol Vis 17:2325–2332
Zhang Y, Zhang L, Song Z, Sun DL, Liu HR, Fu SB, Liu DR, Liu P (2012) Genetic polymorphisms in DNA repair genes OGG1, APE1, XRCC1, and XPD and the risk of age-related cataract. Ophthalmology 119:900–906
Zhou Z, Wang B, Hu S, Zhang C, Ma X, Qi Y (2011) Genetic variations in GJA3, GJA8, LIM2, and age-related cataract in the Chinese population: a mutation screening study. Mol Vis 17:621–626
Acknowledgments
We thank Erika Bürkle, Maria Kugler, and Nadine Senger for expert technical assistance and Lillian Garrett for critically reading the manuscript. The work was supported by National Genome Network Grant NGFN plus (BMBF 01GS0850) and Infrafrontier (Grants BMBF 01KX1011 and BMBF 01KX1012).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Puk, O., de Angelis, M.H. & Graw, J. Lens density tracking in mice by Scheimpflug imaging. Mamm Genome 24, 295–302 (2013). https://doi.org/10.1007/s00335-013-9470-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00335-013-9470-2