Abstract
Objective
To investigate the influence of different volume of interest (VOI) delineation strategies on machine learning–based predictive models for discrimination between low and high nuclear grade clear cell renal cell carcinoma (ccRCC) on dynamic contrast-enhanced CT.
Methods
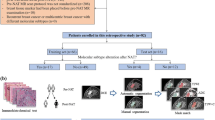
This study retrospectively collected 177 patients with pathologically proven ccRCC (124 low-grade; 53 high-grade). Tumor VOI was manually segmented, followed by artificially introducing uncertainties as: (i) contour-focused VOI, (ii) margin erosion of 2 or 4 mm, and (iii) margin dilation (2, 4, or 6 mm) inclusive of perirenal fat, peritumoral renal parenchyma, or both. Radiomics features were extracted from four-phase CT images (unenhanced phase (UP), corticomedullary phase (CMP), nephrographic phase (NP), excretory phase (EP)). Different combinations of four-phasic features for each VOI delineation strategy were used to build 176 classification models. The best VOI delineation strategy and superior CT phase were identified and the top-ranked features were analyzed.
Results
Features extracted from UP and EP outperformed features from other single/combined phase(s). Shape features and first-order statistics features exhibited superior discrimination. The best performance (ACC 81%, SEN 67%, SPE 87%, AUC 0.87) was achieved with radiomics features extracted from UP and EP based on contour-focused VOI.
Conclusion
Shape and first-order features extracted from UP + EP images are better feature representations. Contour-focused VOI erosion by 2 mm or dilation by 4 mm within peritumor renal parenchyma exerts limited impact on discriminative performance. It provides a reference for segmentation tolerance in radiomics-based modeling for ccRCC nuclear grading.
Key Points
• Lesion delineation uncertainties are tolerated within a VOI erosion range of 2 mm or dilation range of 4 mm within peritumor renal parenchyma for radiomics-based ccRCC nuclear grading.
• Radiomics features extracted from unenhanced phase and excretory phase are superior to other single/combined phase(s) at differentiating high vs low nuclear grade ccRCC.
• Shape features and first-order statistics features showed superior discriminative capability compared to texture features.





Similar content being viewed by others
Abbreviations
- ACC:
-
Accuracy
- AUC:
-
Area under the curve
- ccRCC:
-
Clear cell renal cell carcinoma
- CECT:
-
Contrast-enhanced computed tomography
- CMP:
-
Corticomedullary phase
- CT:
-
Computed tomography
- EP:
-
Excretory phase
- NP:
-
Nephrographic phase
- PACS:
-
Picture archiving and communication system
- RCC:
-
Renal cell carcinoma
- ROC:
-
Receiver operating characteristic
- SEN:
-
Sensitivity
- SMOTE:
-
Synthetic minority oversampling technique
- SPE:
-
Specificity
- UP:
-
Unenhanced phase
- VOI:
-
Volume of interest
References
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA Cancer J Clin 70:7–30
Capitanio U, Bensalah K, Bex A et al (2019) Epidemiology of renal cell carcinoma. Eur Urol 75:74–84
Znaor A, Lortet-Tieulent J, Laversanne M, Jemal A, Bray F (2015) International variations and trends in renal cell carcinoma incidence and mortality. Eur Urol 67:519–530
Leibovich BC, Lohse CM, Crispen PL et al (2010) Histological subtype is an independent predictor of outcome for patients with renal cell carcinoma. J Urol 183:1309–1315
Ljungberg B, Albiges L, Abu-Ghanem Y et al (2019) European Association of Urology Guidelines on Renal Cell Carcinoma: the 2019 update. Eur Urol 75:799–810
Fuhrman SA, Lasky LC, Limas C (1982) Prognostic significance of morphologic parameters in renal cell carcinoma. Am J Surg Pathol 6:655–663
Delahunt B (2009) Advances and controversies in grading and staging of renal cell carcinoma. Mod Pathol 22(Suppl 2):S24-36
Jewett MA, Mattar K, Basiuk J et al (2011) Active surveillance of small renal masses: progression patterns of early stage kidney cancer. Eur Urol 60:39–44
Kunkle DA, Egleston BL, Uzzo RG (2008) Excise, ablate or observe: the small renal mass dilemma–a meta-analysis and review. J Urol 179:1227–1233
Zini L, Perrotte P, Capitanio U et al (2009) Radical versus partial nephrectomy: effect on overall and noncancer mortality. Cancer 115:1465–1471
Campbell N, Rosenkrantz AB, Pedrosa I (2014) MRI phenotype in renal cancer: is it clinically relevant? Top Magn Reson Imaging 23:95–115
Marconi L, Dabestani S, Lam TB et al (2016) Systematic review and meta-analysis of diagnostic accuracy of percutaneous renal tumour biopsy. Eur Urol 69:660–673
Kutikov A, Smaldone MC, Uzzo RG, Haifler M, Bratslavsky G, Leibovich BC (2016) Renal mass biopsy: always, sometimes, or never? Eur Urol 70:403–406
Gillies RJ, Kinahan PE, Hricak H (2016) Radiomics: images are more than pictures, they are data. Radiology 278:563–577
Lambin P, Leijenaar RTH, Deist TM et al (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14:749–762
Erichson BJ, Korfiatis P, Akkus Z, Kline TL (2017) Machine learning for medical imaging. Radiographics 37:505–515
Bektas CT, Kocak B, Yardimci AH et al (2019) Clear cell renal cell carcinoma: machine learning-based quantitative computed tomography texture analysis for prediction of fuhrman nuclear grade. Eur Radiol 29:1153–1163
Kocak B, Durmaz ES, Ates E, Kaya OK, Kilickesmez O (2019) Unenhanced CT texture analysis of clear cell renal cell carcinomas: a machine learning-based study for predicting histopathologic nuclear grade. AJR Am J Roentgenol 212:W132–W139
van Timmeren JE, Cester D, Tanadini-Lang S, Alkadhi H, Baessler B (2020) Radiomics in medical imaging-“how-to” guide and critical reflection. Insights Imaging 11:91
He X, Wei Y, Zhang H et al (2020) Grading of clear cell renal cell carcinomas by using machine learning based on artificial neural networks and radiomic signatures extracted from multidetector computed tomography images. Acad Radiol 27:157–168
Ding J, Xing Z, Jiang Z et al (2018) CT-based radiomic model predicts high grade of clear cell renal cell carcinoma. Eur J Radiol 103:51–56
Gill TS, Varghese BA, Hwang DH et al (2019) Juxtatumoral perinephric fat analysis in clear cell renal cell carcinoma. Abdom Radiol (NY) 44:1470–1480
van Griethuysen JJM, Fedorov A, Parmar C et al (2017) Computational radiomics system to decode the radiographic phenotype. Cancer Res 77:e104–e107
Chawla N, Bowyer K, Hall L, Kegelmeyer W (2002) SMOTE: synthetic minority over-sampling technique. J Artif Intell Res 16:321–357
Lin F, Cui EM, Lei Y, Luo LP (2019) CT-based machine learning model to predict the Fuhrman nuclear grade of clear cell renal cell carcinoma. Abdom Radiol (NY) 44:2528–2534
Kocak B, Ates E, Durmaz ES, Ulusan MB, Kilickesmez O (2019) Influence of segmentation margin on machine learning-based high-dimensional quantitative CT texture analysis: a reproducibility study on renal clear cell carcinomas. Eur Radiol 29:4765–4775
Yap FY, Varghese BA, Cen SY et al (2021) Shape and texture-based radiomics signature on CT effectively discriminates benign from malignant renal masses. Eur Radiol 31:1011–1021
Humphrey PA (2014) Grading renal cell carcinoma: the International Society of Urological Pathology grading system. J Urol 191:798–799
Delahunt B, Cheville JC, Martignoni G et al (2013) The International Society of Urological Pathology (ISUP) grading system for renal cell carcinoma and other prognostic parameters. Am J Surg Pathol 37:1490–1504
Shu J, Wen D, Xi Y et al (2019) Clear cell renal cell carcinoma: machine learning-based computed tomography radiomics analysis for the prediction of WHO/ISUP grade. Eur J Radiol 121:108738
Dagher J, Delahunt B, Rioux-Leclercq N et al (2017) Clear cell renal cell carcinoma: validation of World Health Organization/International Society of Urological Pathology grading. Histopathology 71:918–925
Zhou H, Mao H, Dong D et al (2020) Development and external validation of radiomics approach for nuclear grading in clear cell renal cell carcinoma. Ann Surg Oncol 27:4057–4065
Cui E, Li Z, Ma C et al (2020) Predicting the ISUP grade of clear cell renal cell carcinoma with multiparametric MR and multiphase CT radiomics. Eur Radiol 30:2912–2921
Funding
This study has received funding from the National Natural Science Foundation of China [grant numbers 81971574, 81874216], the Natural Science Foundation of Guangdong Province, P.R. China [grant number 2021A1515011350], the Science and Technology Project of Guangzhou, P.R. China [grant numbers 201904010422, 202002030268, 202102010025], the Guangzhou Key Laboratory of Molecular Imaging and Clinical Translational Medicine and the Construction of High-level Key Clinical Specialty (Medical Imaging) in Guangzhou.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is Dr. Ruimeng Yang.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was waived by the institutional review board.
Ethical approval
Institutional review board approval was obtained.
Methodology
• retrospective
• observational
• multicenter study
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Luo, S., Wei, R., Lu, S. et al. Fuhrman nuclear grade prediction of clear cell renal cell carcinoma: influence of volume of interest delineation strategies on machine learning-based dynamic enhanced CT radiomics analysis. Eur Radiol 32, 2340–2350 (2022). https://doi.org/10.1007/s00330-021-08322-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-021-08322-w