Abstract
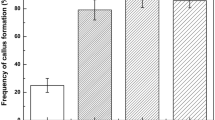
Plants were regenerated from excised adventitious roots of the rose rootstock ‘Moneyway’ via a three step procedure: callus induction, induction of somatic embryos and shoot development. Callus was induced on excised roots after incubation for 4 weeks in the dark on SH-medium (Schenk and Hildebrandt) containing 50 μM 2,4-dichlorophenoxyacetic acid. For embryo induction, calluses were transferred to hormone-free SH-medium and incubated for 8 weeks. The use of Gelrite instead of agar during callus induction stimulated somatic embryogenesis (up to 16% of the explants formed organized structures), whereas the presence of 6-benzylaminopurine in this phase inhibited subsequent regeneration. Yellow solid calluses with embryo-like cotyledons or primordia and friable calluses with embryos were selected, and upon incubation in the light shoots developed. Shoot development was faster and more frequent on solid callus than on friable callus (64% and 21% of the calluses finally formed one or more shoots, respectively). Eleven out of thirteen regenerants developed similarly to control shoots. Finally this regeneration method is compared with other systems for somatic embryogenesis and opportunities for the production of transgenic rose rootstocks and rose cultivare are discussed.
Similar content being viewed by others
Abbreviations
- BAP:
-
6-benzylaminopurine
- BM:
-
basal medium
- BM+:
-
enriched basal medium
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- DAPI-4′,6:
-
diamidino-2-phenylindole
- FeEDDHA:
-
ferric ethylenediamine di(ohydroxyphenylacetate)
- FeEDTA:
-
ferric ethylenediamine tetraacetate
- IBA:
-
indole-3-butyric acid
References
Decout E, Dubois T, Guedira M, Dubois J, Audran JC, Vasseur J (1994) J Exp Bot 45: 1859–1865
De Vries SC, Booij H, Meyerink P, Huisman G, Wilde DH, Thomas TL, Van Kammen A (1988) Planta 176: 196–204
De Wald SG, Litz RE, Moore GA (1989) J Amer Soc Hort Sci 114: 712–716
De Wit JC, Esendam HF, Honkanen JJ, Tuominen U (1990) Plant Cell Rep 9: 456–458
Dudits D, Börge L, Györgyey J (1991) J Cell Sci 99: 475–484
Firoozabady E, Moy Y, Courtney-Gutterson N, Robinson K (1994) Bio/Technology 12: 609–613
Galbraith DW, Harkins KR, Maddox JM, Ayres NM, Sharma DP, Firoozabady E (1983) Science 220: 1049–1051
Kao KN, Michayluk MR (1975) Planta 126: 105–110
Lloyd D, Roberts AV, Short KC (1988) Euphytica 37: 31–36
Noriega C, Söndahl MR (1991) Bio/Technology 9: 991–993
Paek KY, Lee CH, Hwang JK (1985) J Korean Soc Hort Sci 26: 254–260
Quoirin M, Lepoivre Ph, Boxus Ph (1977) In: C. R. Rech. 1976–1977 et Rapports de Synthèse Stat des Cult Fruit et Maraîch. Gembloux Belgium, pp 93–117
Roberts AV, Horan I, Matthews D, Mottley J (1990) In: De Jong J (Ed) Integration of in vitro techniques in ornamental plant breeding. CPO, Wageningen The Netherlands, pp 100–115
Rout GR, Debata BK, Das P (1991) Plant Cell Tiss Org Cult 27: 65–69
Rout GR, Debata BK, Das P (1992) Indian J Exp Biol 30: 15–18
Schenk RU, Hildebrandt AC (1972) Can J Bot 50: 199–204
Sree Ramulu K, Dijkhuis P (1986) Plant Cell Rep 3: 234–237
Van Ark HF, Zaal MACM, Creemers-Molenaar J, Van der Valk P (1991) Plant Cell Tiss Org Cult 27: 275–280
Van der Krieken WM, Breteler H, Visser MHM, Mavridou D (1993) Plant Cell Rep 12: 203–206
Van der Salm TPM, Van der Toorn CJG, Hänisch ten Cate ChH, Dubois LAM, De Vries DP, Dons JJM (1994) Plant Cell Tiss Org Cult 37: 73–77
Wenck AR, Conger BV, Trigiano RN, Sams CE (1988) Plant Physiol 88: 990–992
Author information
Authors and Affiliations
Additional information
Communicated by H. Lörz
Rights and permissions
About this article
Cite this article
van der Salm, T.P.M., van der Toorn, C.J.G., ten Cate Hänisch, C.H. et al. Somatic embryogenesis and shoot regeneration from excised adventitious roots of the rootstock Rosa hybrida L. ‘Moneyway’. Plant Cell Reports 15, 522–526 (1996). https://doi.org/10.1007/BF00232986
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00232986