Abstract
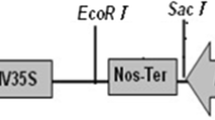
A new method for obtaining transgenic cowpea was developed using positive selection based on the Escherichia coli 6-phosphomannose isomerase gene as the selectable marker and mannose as the selective agent. Only transformed cells were capable of utilizing mannose as a carbon source. Cotyledonary node explants from 4-day-old in vitro-germinated seedlings of cultivar Pusa Komal were inoculated with Agrobacterium tumefaciens strain EHA105 carrying the vector pNOV2819. Regenerating transformed shoots were selected on medium supplemented with a combination of 20 g/l mannose and 5 g/l sucrose as carbon source. The transformed shoots were rooted on medium devoid of mannose. Transformation efficiency based on PCR analysis of individual putative transformed shoots was 3.6%. Southern blot analysis on five randomly chosen PCR-positive plants confirmed the integration of the pmi transgene. Qualitative reverse transcription (qRT-PCR) analysis demonstrated the expression of pmi in T0 transgenic plants. Chlorophenol red (CPR) assays confirmed the activity of PMI in transgenic plants, and the gene was transmitted to progeny in a Mendelian fashion. The transformation method presented here for cowpea using mannose selection is efficient and reproducible, and could be used to introduce a desirable gene(s) into cowpea for biotic and abiotic stress tolerance.







Similar content being viewed by others
Abbreviations
- BAP:
-
6-Benzylaminopurine
- TDZ:
-
Thidiazuron
- pmi :
-
Phosphomannose isomerase
- CPR:
-
Chlorophenol red
References
An G, Evert PR, Mitra A, Ha SB (1988) In: Gelvin SB, Schilperoot RA (eds) Plant molecular biology manual. Kluwer, Dordrecht, pp 1–19
Aswath CR, Mo SY, Kim DH, Park SW (2006) Agrobacterium and biolistic transformation of onion using non-antibiotic selection marker phosphomannose isomerase. Plant Cell Rep 25:92–99
Bakshi S, Sadhukhan A, Mishra S, Sahoo L (2011) Improved Agrobacterium-mediated transformation of cowpea via sonication and vacuum infiltration. Plant cell Rep 30(12):2281–2292. doi:10.1007/s00299-011-1133-8
Ballester A, Cervera M, Pena L (2008) Evaluation of selection strategies alternative to nptII in genetic transformation of citrus. Plant Cell Rep 27:1005–1015
Boscariol RL, Almeida WA, Derbyshire MT, Mourao Filho FA, Mendes BM (2003) The use of the PMI/mannose selection system to recover transgenic sweet orange plants (Citrus sinensis L. Osbeck). Plant Cell Rep 22(2):122–128
Chaudhury D, Madanpotra S, Jaiwal R, Saini R, Kumar PA, Jaiwal PK (2007) Agrobacterium tumefaciens-mediated high frequency genetic transformation of an Indian cowpea (Vigna unguiculata L. Walp.) cultivar and transmission of transgenes into progeny. Plant Sci 172:692–700
Chiang YC, Kiang YT (1988) Genetic analysis of mannose-6-phosphate isomerase in soybeans. Genome 30:808–811
Chilton MD, Currier TC, Farrand SK, Bendich AJ, Gordon MP, Nester EW (1974) Agrobacterium tumefaciens DNA and PS8 bacteriophage DNA not detected in crown gall tumors. Proc Natl Acad Sci USA 71:3672–3676
Degenhardt J, Poppe A, Montag J, Szankowski I (2006) The use of the phosphomannose-isomerase/mannose selection system to recover transgenic apple plants. Plant Cell Rep 25:1149–1156
Dutt M, Lee DH, Grosser JW (2010) Bifunctional selection–reporter systems for genetic transformation citrus: mannose- and kanamycin-based systems. In Vitro Cell Dev Biol-Plant 46:467–476. doi:10.1007/s11627-010-9300-0
Fang J, Chao CT, Roberts PA, Ehlers JD (2007) Genetic diversity of cowpea [Vigna unguiculata (L.) Walp.] in four West African and USA breeding programs as determined by AFLP analysis. Genet Resour Crop Evol 54:1197–1209
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cell. Exp Cell Res 50:151–158
Gurel S, Gurel E, Kaur R, Wong J, Meng L, Tan HQ, Lemaux PG (2009) Efficient, reproducible Agrobacterium-mediated transformation of sorghum using heat treatment of immature embryos. Plant Cell Rep 28:429–444
Hare PD, Chua NH (2002) Excision of selectable marker genes from transgenic plants. Nat Biotechnol 20:575–580
He Z, Fu Y, Si H, Hu G, Zhang S, Yu Y, Sun Z (2004) Phosphomannose-isomerase (pmi) gene as a selectable marker for rice transformation via Agrobacterium. Plant Sci 166:17–22
He Z, Duan Z, Liang W, Chen F, Yao W, Liang H, Yue C, Sun Z, Chen F, Dai J (2006) Mannose selection system used for cucumber transformation. Plant Cell Rep 25(9):953–958
Ivo NL, Nascimento CP, Vieira LS, Campos FAP, Aragao FJL (2008) Biolistic-mediated genetic transformation of cowpea (Vigna unguiculata) and stable Mendelian inheritance of transgenes. Plant Cell Rep 27:1475–1483
Jain M, Chengalrayan K, Abouzid A, Gallo M (2007) Prospecting the utility of a PMI/mannose selection system for the recovery of transgenic sugarcane (Saccharum spp. hybrid) plants. Plant Cell Rep 26:581–590
Joersbo M, Okkels FT (1996) A novel principle for selection of transgenic plant cells: positive selection. Plant Cell Rep 16(3/4):219–221
Joersbo M, Donaldson I, Kreibeg J, Petersen SG, Brunstedt J, Okkels FT (1998) Analysis of mannose selection used for transformation of sugar beet. Mol Breed 4:111–117
Kim JY, Jung M, Kim HS, Lee YH, Choi SH, Lim YP, Min BW, Yang SG, Harn CH (2002) A new selection system for pepper regeneration by mannose. J Plant Biotechnol 4:129–134
Lamblin F, Aime A, Hano C, Roussy I, Domon JM, Van Droogenbroeck B, Laine E (2007) The use of the phosphomannose isomerase gene as alternative selectable marker for Agrobacterium-mediated transformation of flax (Linum usitatissimum). Plant Cell Rep 26:765–772
Lee BT, Matheson NK (1984) Phosphomannoisomerase and phosphoglucoisomerase in seeds of Cassia coluteoides and some other legumes that synthesize galactomannan. Phytochem 23:983–987
Lucca P, Ye X, Potrykus I (2001) Effective selection and regeneration of transgenic rice plants with mannose as selective agent. Mol Breed 7:43–49
Miles JS, Guest JR (1984) Nucleotide sequence and transcriptional start point of the phosphomannose isomerase gene (manA) of Escherichia coli. Gene 32:41–48
Min BW, Cho YN, Song MJ, Noh TK, Kim BK, Chae WK, Park YS, Choi YD, Harn CH (2007) Successful genetic transformation of Chinese cabbage using phosphomannose isomerase as a selection marker. Plant Cell Rep 26:337–344
Murashige T, Skoog S (1962) A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol Plant 15:473–497
Muthukumar B, Mariamma M, Veluthambi K, Gnanam A (1996) Genetic transformation of cotyledon explants of cowpea (Vigna unguiculata L. Walp) using Agrobacterium tumefaciens. Plant Cell Rep 15:980–985
Negrotto D, Jolley M, Beer S, Wenck AR, Hansen G (2000) The use of phosphomannose-isomerase as a selectable marker to recover transgenic maize plants (Zea mays L.) via Agrobacterium transformation. Plant Cell Rep 19:798–803
Obembe OO (2008) Exciting times for cowpea genetic transformation research. Life Sci J 5(2):50–52
O’Kennedy MM, Burger JT, Botha FC (2004) Pearl millet transformation system using the positive selectable marker gene phosphomannose isomerase. Plant Cell Rep 22:684–690
Patil G, Deokar A, Jain PK, Thengane RJ, Srinivasan R (2009) Development of a phosphomannose isomerase-based Agrobacterium-mediated transformation system for chickpea (Cicer arietinum L.). Plant Cell Rep 28:1669–1676
Popelka JC, Gollasch S, Moore A, Molvig L, Higgins TJV (2006) Genetic transformation of cowpea (Vigna unguiculata L.) and stable transmission of the transgenes to progeny. Plant Cell Rep 25:304–312
Privalle LS (2002) Phosphomannose isomerase, a novel plant selection system: potential allergenicity assessment. Ann NY Acad Sci 964:129–138
Qiao GR, Zhou J, Jiang J, Sun YH, Pan LY, Song HG, Jiang JM, Zhuo RY, Wang XJ, Sun ZX (2010) Transformation of Liquidambar formosana L. via Agrobacterium tumefaciens using a mannose selection system and recovery of salt tolerant lines. Plant Cell Tiss Organ Cult 102:163–170
Ramesh SA, Kaiser BN, Franks T, Collins G, Sedgley M (2006) Improved methods in Agrobacterium-mediated transformation of almond using positive (mannose/pmi) or negative (kanamycin resistance) selection-based protocols. Plant Cell Rep 25:821–828. doi:10.1007/s00299-006-0139-0
Reed J, Privalle LS, Powell ML, Meghji M, Dawson J, Dunder E, Suttie J, Wenck A, Launis K, Kramer C, Chang YF, Hansen G, Wright M (2001) Phosphomannose isomerase: an efficient selectable marker for plant transformation. In Vitro Cell Dev Biol Plant 37:127–132
Sambrook KJ, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, New York
Shimelis H, Shiringani R (2010) Variance components and heritabilities of yield and agronomic traits among cowpea genotypes. Euphytica 176:383–389
Sigareva M, Spivey R, Willits MG, Kraimer CM, Chang YF (2004) An efficient mannose selection protocol for tomato that has no adverse effect on the ploidy level of transgenic plants. Plant Cell Rep 23:236–245
Singh NP, Singh YI (2010) Strategy for improving the nutritional quality of grain legumes through genetic engineering. Trends in Biosci 3:1–5
Singh BB, Ehlers JD, Sharma B, Freire-Filho FR (2002) Recent progress in cowpea breeding. In: Fatokun CA, Tarawali SA, Singh BB, Kormawa PM, Tamo M (eds) Proceedings, World Cowpea Conference III, Challenges and opportunities for enhancing sustainable cowpea production. IITA, Ibadan, pp 22–40
Solleti SK, Bakshi S, Purkayastha J, Panda SK, Sahoo L (2008a) Transgenic cowpea (Vigna unguiculata) seeds expressing a bean α-amylase inhibitor 1 confer resistance to storage pests, bruchid beetles. Plant Cell Rep 27:1841–1850
Solleti SK, Bakshi S, Sahoo L (2008b) Additional virulence genes in conjunction with efficient selection scheme, and compatible culture regime enhance recovery of stable transgenic plants in cowpea via Agrobacterium tumefaciens-mediated transformation. J Biotech 135:97–104
Stavolone L, Kononova M, Pauli S, Ragozzino A, De Haan P, Milligan S, Lawton K, Hohn T (2003) Cestrum yellow leaf curling virus promoter: a new strong constitutive promoter for heterologous gene expression in a wide variety of crops. Plant Mol Biol 53:703–713
Waddington SR, Li X, Dixon J, Hyman G, de Vicente MC (2010) Getting the focus right: production constraints for six major food crops in Asian and African farming systems. Nature 2:27–48
Wallbraun M, Sonntag K, Eisenhauer C, Krzcal C, Wang YP (2009) Phosphomannose-isomerase (pmi) gene as a selectable marker for Agrobacterium-mediated transformation of rapeseed. Plant Cell Tiss Organ Cult 99(3):345–351
Wang AS, Evans RA, Altendorf PR, Hanten JA, Doyle MC, Rosichan JL (2000) A mannose selection system for production of fertile transgenic maize plants from protoplasts. Plant Cell Rep 19:654–660
Wright M, Dawson J, Dunder E, Suttie J, Reed J, Kramer C, Chang Y, Novitzky R, Wang H, Artim-Moore L (2001) Efficient biolistic transformation of maize (Zea mays L.) and wheat (Triticum aestivum L.) using the phosphomannose isomerase gene, pmi, as the selectable marker. Plant Cell Rep 20:429–436
Zhang P, Puonti-Kaerlas J (2000) PIG-mediated cassava transformation using positive and negative selection. Plant Cell Rep 19:1041–1048
Zhang P, Potrykus I, Puonti-Kaerlas J (2000) Efficient production of transgenic cassava using negative and positive selection. Transgenic Res 9:405–415
Zhu YJ, Agbayani R, McCafferty H, Albert HH, Moore PH (2005) Effective selection of transgenic papaya plants with the PMI/Man selection system. Plant Cell Rep 24:426–432
Acknowledgments
We thank Syngenta, Switzerland, for providing the plasmid pNOV2819 and Prof. K. Veluthambi, MKU, Madurai, India for the Agrobacterium strain. This work was supported by the grain legume improvement grant and program support grant from the Department of Biotechnology, Government of India. Souvika Bakshi and Nand Kishor Roy are grateful to the Council of Scientific and Industrial Research (CSIR) for Senior and Junior Research Fellowships respectively. Authors are also thankful to the editor for useful suggestions that helped in improving the clarity of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by D. Zaitlin.
Rights and permissions
About this article
Cite this article
Bakshi, S., Saha, B., Roy, N.K. et al. Successful recovery of transgenic cowpea (Vigna unguiculata) using the 6-phosphomannose isomerase gene as the selectable marker. Plant Cell Rep 31, 1093–1103 (2012). https://doi.org/10.1007/s00299-012-1230-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-012-1230-3