Abstract
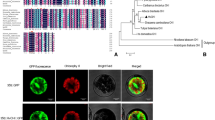
The enzyme that catalyzes the formation of the first stable anthocyanin in the biosynthesis of natural compounds is UDP-glucose: anthocyanidin 3-O-glucosyltransferase (UF3GT). A cDNA clone (Fh3GT1) encoding UF3GT was isolated from Freesia hybrida. Phylogenetic tree analysis indicated that Fh3GT1 was a novel member of glycosyltransferase, which was classified into monocot subgroups. Semi-quantitative RT-PCR analysis detected transcripts of Fh3GT1 in different organs of F. hybrida and in petals of Freesia cultivars of different colors, and the expression level reached the maximum at the fully opened stage of petals. Characterization of the enzymatic assays indicated that Fh3GT1 had a role in anthocyanin glycoside biosyntheses in vitro. To elucidate the function of Fh3GT1, RNA interference vector (pART-Fh3GT1i) was constructed, and introduced into Petunia grandiflora by Agrobacterium-mediated transformation. Integration of the Fh3GT1 in petunia genome was confirmed by PCR and Southern blotting. SqRT-PCR revealed that the endogenous Ph3GT1 mRNA expression levels decreased in transgenic lines compared with the wild-type. The content of total anthocyanin pigments also decreased with the reduction of mRNA transcript levels, and the transgenic petunia plants had significant changes on their flower colors. In summary, this work identified a UF3GT gene from Freesia hybrida and demonstrated a method to modify plant flower color by redirecting the anthocyanin biosynthesis.







Similar content being viewed by others
Abbreviations
- Bp:
-
Base pair
- HPLC:
-
High-performance liquid chromatography
- PSPG:
-
Plant secondary product glycosyltransferase
- PTGS:
-
Post-transcriptional gene silencing
- SqRT-PCR:
-
Semi-quantitative reverse transcription polymerase chain reaction
- UF3GT:
-
UDP-glucose: anthocyanidin 3-O-glucosyltransferase
References
Almeida JR, D’Amico E, Preuss A, Carbone F, de Vos CH, Deiml B, Mourgues F, Perrotta G, Fischer TC, Bovy AG, Martens S, Rosati C (2007) Characterization of major enzymes and genes involved in flavonoid and proanthocyanidin biosynthesis during fruit development in strawberry (Fragaria × ananassa). Arch Biochem Biophys 465:61–71
Arabidopsis GI (2000) Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature 408:796–815
Eamens A, Wang MB, Smith NA, Waterhouse PM (2008) RNA silencing in plants: yesterday, today, and tomorrow. Plant Physiol 147:456–468
Ford CM, Boss PK, Hoj PB (1998) Cloning and characterization of Vitis vinifera UDP-glucose: flavonoid 3-O-glucosyltransferase, a homologue of the enzyme encoded by the maize Bronze-1 locus that may primarily serve to glucosylate anthocyanidins in vivo. J Biol Chem 273:9224–9233
Forkmann G, Martens S (2001) Metabolic engineering and applications of flavonoids. Curr Opin Biotechnol 12:155–160
Fukuchi-Mizutani M, Okuhara H, Fukui Y, Nakao M, Katsumoto Y, Yonekura-Sakakibara K, Kusumi T, Hase T, Tanaka Y (2003) Biochemical and molecular characterization of a novel UDP-glucose: anthocyanin 3′-O-glucosyltransferase, a key enzyme for blue anthocyanin biosynthesis, from gentian. Plant Physiol 132:1652–1663
Gachon CM, Langlois-Meurinne M, Saindrenan P (2005) Plant secondary metabolism glycosyltransferases: the emerging functional analysis. Trends Plant Sci 10:542–549
Gong Z, Yamazaki M, Sugiyama M, Tanaka Y, Saito K (1997) Cloning and molecular analysis of structural genes involved in anthocyanin biosynthesis and expressed in a forma-specific manner in Perilla frutescens. Plant Mol Biol 35:915–927
Goto T, Kondo T, Tamura H, Imagawa H, Lino A, Takeda T (1982) Structure of gentiodelphin, an acylated anthocyanin isolated from Gentiana Makinoi, that is stable in dilute aqueous solution. Tetrahedron Lett 23:3695–3698
Griesser M, Hoffmann T, Bellido ML, Rosati C, Fink B, Kurtzer R, Aharoni A, Munoz-Blañco J, Schwab W (2008) Redirection of flavonoid biosynthesis through the downregulation of an anthocyanidin glucosyltransferase in ripening strawberry fruit. Plant Physiol 146:1528–1539
Grotewold E, Davis KM (2008) Trafficking and sequestration of anthocyanins. Nat Prod Commun 3:1251–1258
Harborne JB, Williams CA (2000) Advances in flavonoid research since 1992. Phytochemistry 55:481–504
Hodges DM, Delong JM, Forney CF, Prange RK (1999) Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207:604–611
Holsters M, de Waele D, Depicker A, Messens E, van Montagu M, Schell J (1978) Transfection and transformation of Agrobacterium tumefaciens. Mol Gen Genet 163:181–187
Holton TA, Cornish EC (1995) Genetics and biochemistry of anthocyanin biosynthesis. Plant Cell 7:1071–1083
Hondo T, Yoshida K, Nakagawa A, Kawai T, Tamura H, Goto T (1992) Structural basis of blue-color development in flower petals from Commelina communis. Nature 358:515–518
Huang JQ, Wei ZM, An HL, Zhu YX (2001) Agrobacterium tumefaciens-mediated transformation of rice with the spider insecticidal gene conferring resistance to leaf folder and striped stem borer. Cell Res 11:149–155
Kaminaga Y, Sahin FP, Mizukami H (2004) Molecular cloning and characterization of a glucosyltransferase catalyzing glucosylation of curcumin in cultured Catharanthus roseus cells. FEBS Lett 567:197–202
Kapitonov D, Yu RK (1999) Conserved domains of glycosyltransferases. Glycobiology 9:961–978
Katsumoto Y, Fukuchi-Mizutani M, Fukui Y et al (2007) Engineering of the rose flavonoid biosynthetic pathway successfully generated blue-hued flowers accumulating delphinidin. Plant Cell Physiol 48:1589–1600
Kerschen A, Napoli CA, Jorgensen RA, Müller AE (2004) Effectiveness of RNA interference in transgenic plants. FEBS Lett 566:223–228
Kim JH, Kim BG, Ko JH, Lee Y, Hur HG, Lim Y, Ahn JH (2006) Molecular cloning, expression, and characterization of a flavonoid glycosyltransferase from Arabidopsis thaliana. Plant Sci 170:897–903
Klein AO, Hagen CW (1961) Anthocyanin production in detached petals of impatiens balsamina L. Plant Physiol 36:1–9
Ko JH, Kima BG, Kim JH, Kim H, Lim CE, Lim J, Lee C, Lim Y, Ahn JH (2008) Four glucosyltransferases from rice: cDNA cloning, expression, and characterization. J Plant Physiol 165:435–444
Kubo A, Arai Y, Nagashima S, Yoshikawa T (2004) Alteration of sugar donor specificities of plant glycosyltransferases by a single point mutation. Arch Biochem Biophys 429:198–203
Li Y, Baldauf S, Lim EK, Bowles DJ (2001) Phylogenetic analysis of the UDP-glycosyltransferase multigene family of Arabidopsis thaliana. J Biol chem 276:4338–4343
Liang HR, Vuorela P, Vuorela H, Hiltunen R (1997) Isolation and immunomodulating effect of flavonol glycosides from Epimedium hunanense. Planta Med 63:316–319
Mackenzie PI, Owens IS, Burchell B et al (1997) The UDP glycosyltransferase gene superfamily: recommended nomenclature update based on evolutionary divergence. Pharmacogenetics 7:255–269
Marrs KA, Alfenito MR, Lloyd AM, Walbot V (1995) A glutathione S-transferase involved in vacuolar transfer encoded by the maize gene Bronze-2. Nature 375:397–400
Martin C, Gerats T (1992) The control of flower coloration. In: Jordan B (ed) Molecular Biology of Flowering. CIB International, Wallingford, UK, pp 219–255
Martin C, Prescott A, Mackay S, Bartlett J, Vrijlandt E (1991) Control of anthocyanin biosynthesis in flowers of Antirrhinum majus. Plant J 1:37–49
Modolo LV, Blount JW, Achnine L et al (2007) A functional genomics approach to (iso)flavonoid glycosylation in the model legume Medicago truncatula. Plant Mol Biol 64:499–518
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Offen W, Martinez-Fleites C, Yang M et al (2006) Structure of a flavonoid glucosyltransferase reveals the basis of plant natural product modification. EMBO J 25:1396–1405
Ossowski S, Schwab R, Weigel D (2008) Gene silencing in plants using artificial microRNAs and other small RNAs. Plant J 53:674–690
Proudfoot NJ, Brownlee GG (1976) 3′ non-coding region sequences in eukaryotic messenger RNA. Nature 263:211–214
Ross J, Li Y, Lim EK, Bowles DJ (2001) Higher plant glycosyltransferases. Genome Biol 2(2): reviews 3004 http://genomebiology.com/2001/2/2/reviews/3004
Saito K, Yamazaki M (2002) Biochemistry and molecular biology of the late-stage of biosynthesis of anthocyanin: lessons from Perilla frutescens as a model plant. New Phytol 155:9–23
Sharp PA (2001) RNA interference-2001. Genes Dev 15:485–490
Smith NA, Singh SP, Wang MB, Stoutjesdijk PA, Green AG, Waterhouse PM (2000) Total silencing by intron-spliced hairpin RNAs. Nature 407:319–320
Tanaka Y, Yonekura K, Fukuchi-Mizutani M, Fukui Y, Fujiwara H, Ashikari T, Kusumi T (1996) Molecular and biochemical characterization of three anthocyanin synthetic enzymes from Gentiana triflora. Plant Cell Physiol 37:711–716
Tanaka Y, Katsumoto Y, Brugliera F, Mason J (2005) Genetic engineering in floriculture. Plant Cell Tiss Org 80:1–24
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Tsuda S, Fukui Y, Nakamura N et al (2004) Flower color modification of Petunia hybrida commercial varieties by metabolic engineering. Plant Biotechnol 21:377–386
Vaucheret H, Béclin C, Fagard M (2001) Post-transcriptional gene silencing in plants. J Cell Sci 114:3083–3091
Vogt T, Jones P (2000) Glycosyltransferase in plant natural product synthesis: characterization of a supergene family. Trends Plant Sci 5:380–386
Wesley SV, Helliwell CA, Smith NA et al (2001) Construct design for efficient, effective and high-throughput gene silencing in plants. Plant J 27:581–590
Winkel-Shirley B (2001) Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol 126:485–493
Wise RP, Rohde W, Salamini F (1990) Nucleotide sequence of the Bronze-1 homologous gene from Hordeum vulgare. Plant Mol Biol 14:277–279
Yamazaki M, Yamagishi E, Gong Z et al (2002) Two flavonoid glucosyltransferases from Petunia hybrida: molecular cloning, biochemical properties and developmentally regulated expression. Plant Mol Biol 48:401–411
Yonekura-Sakakibara K, Nakayama T, Yamazaki M, Saito K (2009) Modification and stabilization of anthocyanins. In: Gould K, Davies K, Winefield C (eds) Anthocyanins biosynthesis functions and applications. Springer, New York, pp 169–190
Yoshida K, Toyama Y, Kameda K, Kondo T (2000) Contribution of each caffeoyl residue of the pigment molecule of gentiodelphin to blue color development. Phytochemistry 54:85–92
Yoshihara N, Imayama T, Fukuchi-Mizutanic M, Okuhara H, Tanaka Y, Ino I, Yabuya T (2005) cDNA cloning and characterization of UDP-glucose: anthocyanidin 3-O-glucosyltransferase in Iris hollandica. Plant Sci 169:496–501
Yu O, Jez JM (2008) Nature’s assembly line: biosynthesis of simple phenylpropanoids and polyketides. Plant J 54:750–762
Acknowledgments
This research was supported by the National Natural Science Foundation of the People’s Republic of China (grant# 30970280) and Jilin provincial Government of the People’s Republic of China (grant# 20085030). We also want to thank Professor Lixia Liu for providing the plant expression vector pKANNIBAL and pART27 and Agrobacterium tumefaciens strain EHA105, and Professors Chuanbin Mao and Xingzhi Wang for helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by B. Li.
Rights and permissions
About this article
Cite this article
Sui, X., Gao, X., Ao, M. et al. cDNA cloning and characterization of UDP-glucose: anthocyanidin 3-O-glucosyltransferase in Freesia hybrida . Plant Cell Rep 30, 1209–1218 (2011). https://doi.org/10.1007/s00299-011-1029-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-011-1029-7