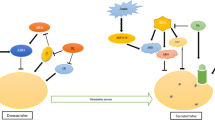
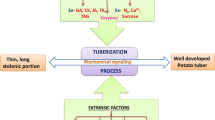
Abstract
Tuberization is one of the multiple outputs of a single-input phytochrome B sensory system, involving several regulatory genes. Phytochrome B- and GA-mediated photoperiodic perception occurs in the leaf, and then the RNA acts as a systemic signal in the long-distance signaling pathway to initiate tuberization in the subapical region of an underground stolon. There is good evidence that flowering and tuberizing signals might be similar. Is there a cross-talk with an oxidative burst-mediated redox signaling pathway during tuberization? Is the lipoxygenase cascade involved in the formation of the perimedullary tissue in a growing tuber? Do aquaporins regulate cell division, expansion and elongation during stolon growth and tuber induction in potato? Is the adaptive diversity for tuberization under varying photoperiods a micro-evolutionary indicator of differential transduction of cell-to-cell signal molecules under spatial and temporal expression of regulatory genes encoding transcriptional activators? Taking these views into consideration, the review presents an interim synthesis of a signaling network regulating in planta tuberization in potato.


Similar content being viewed by others
References
Ahn YJ, Zimmerman JL (2006) Introduction of the carrot HSP 17.7 into potato (Solanum tuberosum L.) enhances cellular membrane stability and tuberization in vitro. Plant Cell Environ 29:95–104
Alleva K, Niemietz CM, Sutka M, Maurel C, Parisi M, Tyerman SD, Amodeo G (2006) Plasma membrane of Beta vulgaris storage root shows high water channel activity regulated by cytoplasmic pH and a dual range of calcium concentrations. J Exp Bot 57:609–621
Amador V, Monte E, Garcia-Martínez J-L, Prat S (2001) Gibberellins signal nuclear import of PHOR1, a photoperiod-responsive protein with homology to Drosophila armadillo. Cell 106:343–354
Atkin OK, Loveys BR, Atkinson LJ, Pons TL (2006) Phenotypic plasticity and growth temperature: understanding interspecific variability. J Exp Bot 57:267–281
Bachem CWB, van der Hoeven, Lucker J, Oomen R, Casarini E, Jacobsen E, Visser RGF (2000) Functional genomic analysis of potato tuber life-cycle. Potato Res 43:297–312
Banerjee AK, Chatterjee M, Yu Y, Suh S-G, Miller WA, Hannapel DJ (2006) Dynamics of a mobile RNA of potato involved in a long-distance signaling pathway. Plant Cell 18:3443–3457
Bologa KL, Fernie AR, Leisse A, Loureiro ME, Geigenberger P (2003) A bypass of sucrose synthase leads to low internal oxygen and impaired metabolic performance in growing potato tubers. Plant Physiol 132:2058–2072
Brown PH, Yang S (2003) Diurnal cycles of stolon and tuber expansion during potato tuberisation. Acta Hortic 619:345–352
Carrera E, Jackson SD, Prat S (1999) Feedback control and diurnal regulation of gibberellin 20-oxidase transcript levels in potato. Plant Physiol 119:765–773
Chaumont F, Barrieu F, Herman EM, Chrispeels MJ (1998) Characterization of a maize tonoplast aquaporin expressed in zones of cell division and elongation. Plant Physiol 117:1143–1152
Chen H, Rosin FM, Prat S, Hannapel DJ (2003) Interacting transcription factors from the TALE superclass regulate tuber formation. Plant Physiol 132:1391–1404
Chen H, Banerjee AK, Hannapel DJ (2004) The tandem complex of BEL and KNOX partners is required for transcriptional repression of ga20ox1. Plant J 38:276–284
Ehlenfeldt MK, Hanneman RE Jr (1984) The use of endosperm balance number and 2n gametes to transfer exotic germplasm in potato. Theor Appl Genet 68:155–161
Ewing EE, Simko I, Omer EA, Davies PJ (2004) Polygene mapping as a tool to study the physiology of potato tuberization and dormancy. Am J Potato Res 81:281–289
Faivre-Rampant O, Cardle L, Marshall D, Viola R, Taylor MA (2004) Changes in gene expression during meristem activation processes in Solanum tuberosum with a focus on the regulation of an auxin response factor gene. J Exp Bot 55:613–622
Fraysse L, Wells B, McCann MC, Kjellbom P (2005) Specific plasma membrane aquaporins of the PIP1 subfamily are expressed in sieve elements and guard cells. Biol Cell 97:519–534
Grant JJ, Yun BW, Loake GJ (2000) Oxidative burst and cognate redox signaling reported by luciferase imaging: identification of a signal network that functions independently of ethylene, SA and Me-JA but is dependent on MAPKK activity. Plant J 24:569–582
Hamberg M (2000) New cyclopentenone fatty acids formed from linoleic and linilenic acids in potato. Lipids 35:353–363
Hannapel DJ, Chen H, Rosin FM, Banerjee AK, Davies PJ (2004) Molecular controls of tuberization. Am J Potato Res 81:263–274
Horváth BM, Magyar Z, Zhang Y, Hamburger AW, Bakó L, Visser RGF, Bachem CWB, Bögre L (2006) EBP1 regulates organ size through cell growth and proliferation in plants. EMBO J 25:4909–4920
Jackson SD (1999) Multiple signaling pathways control tuber induction in potato. Plant Physiol 119:1–8
Jackson SD, Willmitzer L (1994) Jasmonic acid spraying does not induce tuberization in short-day requiring potato species kept in non-inducing conditions. Planta 194:155–159
Jena PK, Reddy ASN, Poovaiah BW (1989) Molecular cloning and sequencing of a cDNA for plant calmodulin: signal-induced changes in the expression of calmodulin. Proc Natl Acad Sci USA 86:3644–3648
Kang SG, Hannapel DJ (1995) Nucleotide sequences of novel potato MADS-box cDNAs and their expression in vegetative organs. Gene 166:329–330
Kang SG, Hannapel DJ, Suh SG (2003) Potato MADS-box gene POTM1–1 transcripts are temporally and spatially distributed in floral organs and vegetative meristems. Mol Cells 15:48–54
Kim M-S, Kim H-S, Kim Y-S, Baek K-H, Oh H-W, Hahn K-W, Bae R-N, Lee I-J, Joung H, Jeon J-H (2007) Superoxide anion regulates plant growth and tuber development of potato. Plant Cell Rep 26:1717–1725
Kloosterman B, Vorst O, Hall RD, Visser RGF, Bachem CW (2005) Tuber on a chip: differential gene expression during potato tuber development. Plant Biotechnol J 3:505–519
Kobayashi M, Kawakita K, Maeshima M, Doke N, Yoshioka H (2006) Subcellular localization of Strboh proteins and NADPH-dependent O2-generating activity in potato tuber tissues. J Exp Bot 57:1371–1379
Koda Y (1997) Possible involvement of jasmonates in various morphogenic events. Physiol Plant 100:639–646
Kolomiets MV, Hannapel DJ, Chen H, Tymeson M, Gladon RJ (2001) Lipoxygenase is involved in the control of potato tuber development. Plant Cell 13:613–626
Lehesranta SJ, Davies HV, Shepherd LVT, Koistinen, Massat N, Nunan N, McNicol JW, Kärenlampi SO (2006) Proteomic analysis of the potato tuber life cycle. Proteomics 6:6042–6052
Maccarrone M, van Zadelhoff G, Veldink GA, Vliegenthart JFG, Finanzzi-Agro A (2000) Early activation of lipoxygenase in lentil (Lens culinaris) root protoplasts by oxidative stress induces programmed cell death. Eur J Biochem 267:5078–5084
Martinez-Garcia JF, Virgós-Soler A, Prat S (2002) Control of photoperiod-regulated tuberization in potato by the Arabidopsis flowering-time gene CONSTANS. Proc Natl Acad Sci USA 99:15211–15216
Nam K-H, Minami C, Kong F, Matsuura H, Takahashi K, Yoshihara T (2005) Relation between environmental factors and the LOX activities upon potato tuber formation and flower-bud formation in morning glory. Plant Growth Regul 46:253–260
Nam K-H, Kong F, Matsuura H, Takahashi K, Nabeta K, Yoshihara T (2007) Temperature regulates tuber-inducing lipoxygenase-derived metabolites in potato (Solanum tuberosum). J Plant Physiol. Doi:10.1016/j.jplph.2007.04.003 (in press)
O’Brien M, Bertrand C, Matton DP (2002) Characterization of a fertilization-induced and developmentally regulated plasma-membrane aquaporin expressed in reproductive tissues, in the wild potato Solanum chacoense Bitt. Planta 215:485–493
Pelacho AM, Mingo-Castel AM (1991) Jasmonic acid induces tuberization of potato stolons cultured in vitro. Plant Physiol 97:1253–1255
Purugannan MD (2000) The molecular genetics of regulatory genes. Mol Ecol 9:1451–1462
Raíces M, Chico JM, Téllez-Iñón MT, Ulloa RM (2001) Molecular characterization of StCDPK1, a calcium-dependent protein kinase from Solanum tuberosum that is induced at the onset of tuber development. Plant Mol Biol 46:591–601
Raíces M, Ulloa RM, MacIntosh GC, Crespi M, Téllez-Iñón MT (2003) StCDPK1 is expressed in potato stolon tips and is induced by high sucrose concentration. J Exp Bot 54:2589–2591
Rodriguez-Falcon M, Bou J, Prat S (2006) Seasonal control of tuberization in potato: conserved elements with the flowering response. Annu Rev Plant Biol 57:151–180
Rosin FM, Hart JK, Horner Jr HT, Davies PJ, Hannapel DJ (2003) Overexpression of a knox gene of potato decreases GA accumulation and enhances tuber formation. Plant Physiol 132:106–117
Royo J, Vancanneyt G, Perez AG, Sanz C, Stormann K, Rosahl S, Sanchez-Serrano JJ (1996) Characterization of three potato lipoxygenases with distinct enzymatic activities and different organ-specific and wound-regulated expression patterns. J Biol Chem 271:21012–21019
Sarkar D, Pandey SK, Sharma S (2006) Cytokinins antagonize the jasmonates action on the regulation of potato (Solanum tuberosum L.) tuber formation in vitro. Plant Cell Tissue Organ Cult 87:285–295
Sergeeva LI, de Bruijn SM, Koot-Gronsveld EAM, Navratil O, Vreugdenhil D (2000) Tuber morphology and starch accumulation are independent phenomena: evidence from ipt-transgenic potato lines. Physiol Plant 108:435–443
Smith H (2000) Phytochromes and light signal perception by plants—an emerging synthesis. Nature 407:585–590
Spooner DM, McLean K, Ramsay G, Waugh R, Bryan GJ (2005) A single domestication for potato based on multilocus amplified fragment length polymorphism genotyping. Proc Natl Acad Sci USA 102:14694–14699
Struik PC, Vreugdenhil D, van Eck HJ, Bachem CW, Visser RGF (1999) Physiological and genetic control of tuber formation. Potato Res 42:313–331
Tauberger E, Fernie AR, Emmermann M, Renz A, Kossmann J, Willmitzer L, Trethewey RN (2000) Antisense inhibition of plastidial phosphoglucomutase provides compelling evidence that potato tuber amyloplasts import carbon from the cytosol in the form of glucose-6-phosphate. Plant J 23:43–53
Taylor MA, Mad Arif SA, Kumar A, Davies HV, Scobie LA, Pearce SR, Flawell AJ (1992) Expression and sequence analysis of cDNAs induced during the early stages of tuberization in different organs of the potato plant (Solanum tuberosum L.). Plant Mol Biol 20:641–651
Taylor MA, George LA, Ross HA, Davies HV (1998) cDNA cloning and characterization of an α-glucosidase gene from potato (Solanum tuberosum L.). Plant J 13:419–425
Ting CT, Tsaur SC, Wu ML, Wu CI (1998) A rapidly evolving homeobox at the site of a hybrid sterility gene. Science 282:1501–1504
Uehlein N, Kaldenhoff R (2006) Aquaporins and biological rhythm. Biol Rhythm Res 37:315–322
van den Berg JH, Ewing EE, Plaisted RL, McMurry S, Bonierable MW (1996) QTL analysis of potato tuberization. Theor Appl Genet 93:307–316
Verhees J, van der Krol AR, Vreugdenhil D, van der Plas LHW (2002) Characterization of gene expression during potato tuber development in individuals and populations using the luciferase reporter system. Plant Mol Biol 50:653–665
Viola R, Roberts AG, Haupt S, Gazzani S, Hancock RD, Marmiroli N, Machray GC, Oparka KJ (2001) Tuberization in potato involves a switch from apoplastic to symplastic phloem unloading. Plant Cell 13:385–398
Vreugdenhil D (2004) Comparing potato tuberization and sprouting: opposite phenomena? Am J Potato Res 81:275–280
Vreugdenhil D, Xu X, Jung CS, van Lammeren AAM, Ewing EE (1999) Initial anatomical changes associated with tuber formation on single-node potato (Solanum tuberosum L.) cuttings: a re-evaluation. Ann Bot 84:675–680
Xu X, van Lammeren AAM, Vermeer E, Vreugdenhil D (1998a) The role of gibberellin, abscisic acid, and sucrose in the regulation of potato tuber formation in vitro. Plant Physiol 117:575–584
Xu X, Vreugdenhil D, van Lammeren AAM (1998b) Cell division and cell enlargement during potato tuber formation. J Exp Bot 49:573–582
Yoshihara T, Amanuma M, Tsutsumi T, Okumura Y, Matsuura H, Ichihara A (1996) Metabolism and transport of [2-14C](±)jasmonic acid in the potato plant. Plant Cell Physiol 37:586–590
Acknowledgments
I thank the Alexander von Humboldt (AvH) Foundation for a fellowship at the Universität Tübingen, Tübingen, Germany, where some of this work was done. I also thank Dr. SK Pandey, Director, Central Potato Research Institute, Shimla (India) for providing facilities. Comments and suggestions on the manuscript from the editor and two anonymous reviewers are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. Kumar.
Rights and permissions
About this article
Cite this article
Sarkar, D. The signal transduction pathways controlling in planta tuberization in potato: an emerging synthesis. Plant Cell Rep 27, 1–8 (2008). https://doi.org/10.1007/s00299-007-0457-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-007-0457-x