Abstract
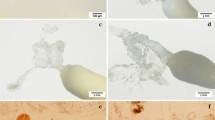
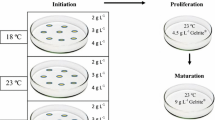
In this study, several improvements and simplifications of SE protocols in Pinus pinaster (Ait.), a species of economic importance in the regions of Western Europe, are described. These improvements pertained to all stages of SE including high initiation frequencies in eight control pollinated seed families, relatively high somatic embryo maturation yield when cells were coated with particles of activated charcoal and a rapid production of plants directly in a shade house. The SE initiation frequency from isolated zygotic embryos was high (up to 100%) and plants were produced from 11 embryogenic lines representing all crosses. Based on these results, the estimated number of somatic embryos required to produce 1,000 plants varied from slightly more than the required number of plants to more than double this number depending on the line. Such an estimate is critical in developing plant production strategy when a number of embryogenic lines are considered for production of clonal plants.

Similar content being viewed by others
References
Bercetche J, Pâques M (1995) Somatic embryogenesis in maritime pine (Pinus pinaster). In: Jain SM, Gupta PK, Newton RJ (eds) Somatic embryogenesis in woody plants. Gymnosperms, vol 3. Kluwer Academic Publishers, Dordrecht, Netherlands, pp 221–242
Baradat P, Pastuszka P (1992) Le pin maritime. In: Gallais A, Bannerot H (eds) Amélioration des espèces végétales cultivées. Institut National de la Recherche Agronomique, Paris, pp 695–709
Breton D, Harvengt L, Trontin J-F, Bouvet A, Favre J-M (2005) High subculture frequency, and maltose-based and hormone-free medium sustained early development of somatic embryos in maritime pine. In Vitro Cell Dev Biol Plant 41:494–504
Brown H, Prescott R (1999) Applied mixed models in medicine. Wiley, Chichester, 408 pp
Charbit E, Legavre T, Lardet L, Bourgeois E, Ferriere N, Carron MP (2004) Identification of differentially expressed cDNA sequences and histological characteristics of Hevea brasiliensis calli in relation to their embryogenic and regenerative capacities. Plant Cell Rep 22:539–548
Cyr DR, Klimaszewska K (2002) Conifer somatic embryogenesis: Applications. Dendrobiology 48:41–49
Ebert A, Taylor F, Blake J (1993) Changes of 6-benzylaminopurine and 2,4-dichlorophenoxyacetic acid concentrations in plant tissue culture media in the presence of activated charcoal. Plant Cell Tissue Organ Cult 33:157–162
Hopkins ER, Butcher TB (1993) Provenance comparisons of Pinus pinaster (Ait.) in Western Australia. CALMScience 1:55–105
Illy G (1966) Recherches sur l’amélioration génétique du pin maritime. Ann Sci Forest 23:757–948
Klimaszewska K, Cyr DR (2002) Conifer somatic embryogenesis: Development. Dendrobiology 48:31–39
Klimaszewska K, Smith DR (1997) Maturation of somatic embryos of Pinus strobus is promoted by a high concentration of gellan gum. Physiol Plant 100:949–957
Klimaszewska K, Bernier-Cardou M, Cyr DR, Sutton BCS (2000) Influence of gelling agents on culture medium gel strength, water availability, tissue water potential, and maturation response in embryogenic cultures of Pinus strobus L. In Vitro Cell Dev Biol-Plant 36:279–286
Klimaszewska K, Park Y-S, Overton C, Maceacheron I, Bonga JM (2001) Optimized somatic embryogenesis in Pinus strobus L. In Vitro Cell Dev Biol-Plant 37:392–399
Lelu MA, Bastien C, Klimaszewska K, Ward C, Charest PJ (1994) An improved method for somatic plantlet production in hybrid larch (Larix x leptoeuropaea). Part 1 Somatic embryo maturation. Plant Cell Tissue Organ Cult 36:107–115
Lelu MA, Bastien C, Drugeault A, Gouez ML, Klimaszewska K (1999) Somatic embryogenesis and plantlet development in Pinus sylvestris and Pinus pinaster on medium with and without growth regulators. Physiol Plant 105:719–728
Litvay JD, Verma DC, Johnson MA (1985) Influence of loblolly pine (Pinus taeda L.) culture medium and its components on growth and somatic embryogenesis of the wild carrot (Daucus carota L.). Plant Cell Rep 4:325–328
McCullagh P, Nelder JA (1989) Generalized linear models. Chapman & Hall, London, 511 pp
Miguel C, Gonçalves S, Tereso S, Marum L, Oliveira MM (2004) Somatic embryogenesis from 20 open-pollinated seed families of Portuguese plus trees of maritime pine. Plant Cell Tissue Org Cult 76:121–130
Niskanen A-M, Lu J, Seitz S, Keinonen K, von Weissenberg K, Pappinen A (2004) Effect of parent genotype on somatic embryogenesis in Scots pine (Pinus sylvestris). Tree Physiol 24:1259–1265
Park YS, Pond SE, Bonga JM (1993) Initiation of somatic embryogenesis in white spruce (Picea glauca): genetic control, culture treatment effects, and implications for tree breeding. Theory Appl Genet 86:427–436
Percy RE, Klimaszewska K, Cyr DR (2000) Evaluation of somatic embryogenesis for clonal propagation of western white pine. Can J For Res 30:1867–1876
Ramarosandratana A, Harvengt L, Bouvet A, Calvayrac R, Pâques M (2001a) Effects of carbohydrate source, polyethylene glycol and gellan gum concentration on embryonal-suspensor mass (ESM) proliferation and maturation of maritime pine somatic embryos. In Vitro Cell Dev Biol-Plant 37:29–34
Ramarosandratana A, Harvengt L, Bouvet A, Calvayrac R, Pâques M (2001b) Influence of the embryonal-suspensor mass (ESM) sampling on development and proliferation of maritime pine somatic embryos. Plant Sci 160:473–479
von Aderkas P, Label P, Lelu MA (2002) Charcoal affects early development and hormonal concentrations of somatic embryos of hybrid larch. Tree Physiol 22:431–434
Acknowledgements
This work was partly funded by EU (GEMINI, QLK5-CT-1999- 00942). The authors thank A. Raffin and P. Patuszka from INRA, Bordeaux for providing the cones from the breeding program. K. Klimaszewska's contribution was possible through the financial support of le Studium, Region Centre, France.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by L. Jouanin
Rights and permissions
About this article
Cite this article
Lelu-Walter, MA., Bernier-Cardou, M. & Klimaszewska, K. Simplified and improved somatic embryogenesis for clonal propagation of Pinus pinaster (Ait.). Plant Cell Rep 25, 767–776 (2006). https://doi.org/10.1007/s00299-006-0115-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-006-0115-8