Abstract
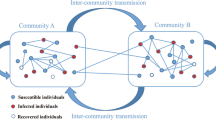
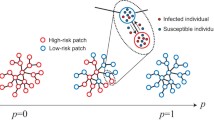
As there are no targeted medicines or vaccines for newly emerging infectious diseases, isolation among communities (villages, cities, or countries) is one of the most effective intervention measures. As such, the number of intercommunity edges (\({\textit{NIE}}\)) becomes one of the most important factor in isolating a place since it is closely related to normal life. Unfortunately, how \({\textit{NIE}}\) affects epidemic spread is still poorly understood. In this paper, we quantitatively analyzed the impact of \({\textit{NIE}}\) on infectious disease transmission by establishing a four-dimensional \({\textit{SIR}}\) edge-based compartmental model with two communities. The basic reproduction number \(R_{0}(\langle l\rangle )\) is explicitly obtained subject to \({\textit{NIE}}\) \(\langle l\rangle \). Furthermore, according to \(R_{0}(0)\) with zero \({\textit{NIE}}\), epidemics spread could be classified into two cases. When \(R_{0}(0)>1\) for the case 2, epidemics occur with at least one of the reproduction numbers within communities greater than one, and otherwise when \(R_0(0)<1\) for case 1, both reproduction numbers within communities are less than one. Remarkably, in case 1, whether epidemics break out strongly depends on intercommunity edges. Then, the outbreak threshold in regard to \({\textit{NIE}}\) is also explicitly obtained, below which epidemics vanish, and otherwise break out. The above two cases form a severity-based hierarchical intervention scheme for epidemics. It is then applied to the SARS outbreak in Singapore, verifying the validity of our scheme. In addition, the final size of the system is gained by demonstrating the existence of positive equilibrium in a four-dimensional coupled system. Theoretical results are also validated through numerical simulation in networks with the Poisson and Power law distributions, respectively. Our results provide a new insight into controlling epidemics.








Similar content being viewed by others
References
Allen L (2010) An introduction to stochastic processes with applications to biology, 2nd edn. Chapman and Hall/CRC. https://doi.org/10.1201/b12537
Arino J, Bajeux N, Kirkland S (2019) Number of source patches required for population persistence in a source-sink metapopulation with explicit movement. Bull Math Biol 81:1916. https://doi.org/10.1007/s11538-019-00593-1
Arino J, Boëlle PY, Milliken E, Portet S (2021) Risk of COVID-19 variant importation? How useful are travel control measures? Infect Dis Model 6:875. https://doi.org/10.1016/j.idm.2021.06.006
Asamoah JKK, Okyere E, Abidemi A, Moore SE, Sun GQ, Jin Z, Acheampong E, Gordon JF (2022) Optimal control and comprehensive cost-effectiveness analysis for COVID-19. Results Phys 33:105177. https://doi.org/10.1016/j.rinp.2022.105177
Bajiya VP, Tripathi JP, Kakkar V, Wang J, Sun G (2021) Global dynamics of a multi-group SEIR epidemic model with infection age. Chin Ann Math Ser B 42(6):833. https://doi.org/10.1007/s11401-021-0294-1
Ball F, Neal P (2008) Network epidemic models with two levels of mixing. Math Biosci 212(1):69. https://doi.org/10.1016/j.mbs.2008.01.001
Bavel JJV, Baicker K, Boggio PS, Capraro V, Cichocka A, Cikara M, Crockett MJ, Crum AJ, Douglas KM, Druckman JN et al (2020) Using social and behavioural science to support COVID-19 pandemic response. Nat Hum Behav 4(5):460. https://doi.org/10.1038/s41562-020-0884-z
Bender EA, Canfield ER (1978) The asymptotic number of labeled graphs with given degree sequences. J Combin Theory Ser A 24(3):296. https://doi.org/10.1016/0097-3165(78)90059-6
Boily MC, Lowndes C, Alary M (2002) The impact of HIV epidemic phases on the effectiveness of core group interventions: insights from mathematical models. Sex Transm Infect 78(suppl 1):i78. https://doi.org/10.1136/sti.78.suppl_1.i78
Buonomo B, Lacitignola D, de León CV (2014) Qualitative analysis and optimal control of an epidemic model with vaccination and treatment. Math Comput Simul 100:88. https://doi.org/10.1016/j.matcom.2013.11.005
Butler D (2014) Ebola experts seek to expand testing. Nature 516(7530):154. https://doi.org/10.1038/516154a
C for Disease Control P (CDC) (2003) Severe acute respiratory syndrome? Singapore. MMWR Morb Mortal Wkly Rep 52:405
Cao X (2020) COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol 20(5):269. https://doi.org/10.1038/s41577-020-0308-3
Catanzaro M, Boguná M, Pastor-Satorras R (2005) Generation of uncorrelated random scale-free networks. Phys Rev E 71(2):027103. https://doi.org/10.1103/PhysRevE.71.027103
Chen L, Sun J (2014) Global stability and optimal control of an SIRS epidemic model on heterogeneous networks. Phys A Stat Mech Appl 410:196. https://doi.org/10.1016/j.physa.2014.05.034
Chen L, Sun J (2014) Optimal vaccination and treatment of an epidemic network model. Phys Lett A 378:3028. https://doi.org/10.1016/j.physleta.2014.09.002
Chowdhury R, Heng K, Shawon M, Goh G, Franco OH (2020) Dynamic interventions to control COVID-19 pandemic: a multivariate prediction modelling study comparing 16 worldwide countries. Eur J Epidemiol 35(1):1. https://doi.org/10.1007/s10654-020-00649-w
Chowell G, Fenimore P, Castillo-Garsow M, Castillo-Chavez C (2003) SARS outbreaks in Ontario, Hong Kong and Singapore: the role of diagnosis and isolation as a control mechanism. J Theor Biol 224(1):1. https://doi.org/10.1016/S0022-5193(03)00228-5
Clara S, Remco VDH, Johan SHVL (2016) Epidemic spreading on complex networks with community structures. Sci Rep 6(1):1. https://doi.org/10.1038/srep29748
de Jesus Esquivel-Gómez J, Barajas-Ramírez JG (2018) Efficiency of quarantine and self-protection processes in epidemic spreading control on scale-free networks. Chaos 28(1):013119. https://doi.org/10.1063/1.5001176
Feng S, Luo X, Pei X, Jin Z, Lewis M, Wang H (2022) Modeling the early transmission of COVID-19 in New York and San Francisco using a pairwise network model. Infect Dis Model 7(1):19. https://doi.org/10.1016/j.idm.2021.12.009
Forster GA, Gilligan CA (2007) Optimizing the control of disease infestations at the landscape scale. Proc Natl Acad Sci 104:4984. https://doi.org/10.1073/pnas.0607900104
Fraser C, Riley S, Anderson RM, Ferguson NM (2004) Factors that make an infectious disease outbreak controllable. Proc Natl Acad Sci 101(16):6146. https://doi.org/10.1073/PNAS.0307506101
Fu X, Small M, Walker DM, Zhang H (2008) Epidemic dynamics on scale-free networks with piecewise linear infectivity and immunization. Phys Rev E 77:036113. https://doi.org/10.1103/PhysRevE.77.036113
Guo J, Huang F, Liu J, Chen Y, Wang W, Cao B, Zou Z, Liu S, Pan J, Bao C et al (2015) The serum profile of hypercytokinemia factors identified in H7N9-infected patients can predict fatal outcomes. Sci Rep 5(1):1. https://doi.org/10.1038/srep21230
Halloran ME, Jr I, Nizam A, Yang Y (2002) Containing bioterrorist smallpox. Science 298(5597):1428. https://doi.org/10.1126/science.1074674
Hammarlund E, Lewis MW, Hansen SG, Strelow LI, Nelson JA, Sexton GJ, Hanifin JM, Slifka MK (2003) Duration of antiviral immunity after smallpox vaccination. Nat Med 9:1131. https://doi.org/10.1038/nm917
Haynes BF, Pantaleo G, Fauci AS (1996) Toward an understading of the correlates of protective immunity to HIV infection. Science 271(5247):324. https://doi.org/10.1126/science.271.5247.324
Horby P (2013) H7N9 is a virus worth worrying about. Nature 496(7446):399. https://doi.org/10.1038/496399a
Huang W, Li C (2007) Epidemic spreading in scale-free networks with community structure. J Stat Mech Theory Exp 2007(01):P01014. https://doi.org/10.1088/1742-5468/2007/01/P01014
Huang W, Li C (2007) Epidemic spreading in scale-free networks with community structure. J Stat Mech Theory Exp 2007:P01014. https://doi.org/10.1088/1742-5468/2007/01/P01014
Iacoviello D, Stasio N (2013) Optimal control for SIRC epidemic outbreak. Comput Methods Programs Biomed 110:333. https://doi.org/10.1016/j.cmpb.2013.01.006
Jin EM, Girvan M, Newman M (2001) The structure of growing social networks. Work Pap 64(4 Pt 2):046132. https://doi.org/10.1103/PhysRevE.64.046132
Jin Z, Zhang J, Song LP, Sun GQ, Kan J, Zhu H (2011) Modelling and analysis of influenza A (H1N1) on networks. BMC Public Health 11(1):1. https://doi.org/10.1186/1471-2458-11-S1-S9
Kandhway K, Kuri J (2014) How to run a campaign: optimal control of SIS and SIR information epidemics. Appl Math Comput 231:79. https://doi.org/10.1016/j.amc.2013.12.164
Kandhway K, Kuri J (2014) Optimal control of information epidemics modeled as Maki Thompson rumors. Commun Nonlinear Sci Numer Simul 19(12):4135. https://doi.org/10.1016/j.cnsns.2014.04.022
Kang H, Liu K, Fu X (2017) Dynamics of an epidemic model with quarantine on scale-free networks. Phys Lett A 381(47):3945. https://doi.org/10.1016/j.physleta.2017.09.040
Keeling MJ, Gilligan CA (2000) Metapopulation dynamics of bubonic plague. Nature 407(6806):903. https://doi.org/10.1038/35038073
Koch D, Illner R, Ma J (2013) Edge removal in random contact networks and the basic reproduction number. J Math Biol 67(2):217. https://doi.org/10.1007/s00285-012-0545-6
Kostyuchenko VA, Lim EXY, Zhang S, Fibriansah G, Ng TS, Ooi JSG, Shi J, Lok SM (2016) Structure of the thermally stable Zika virus. Nature 533(7603):425. https://doi.org/10.1038/nature17994
Larocca RA, Abbink P, Peron JPS, de Zanotto PM, Iampietro MJ, Badamchi-Zadeh A, Boyd M, Ng’ang’a D, Kirilova M, Nityanandam R et al (2016) Vaccine protection against Zika virus from Brazil. Nature 536(7617):474. https://doi.org/10.1038/nature18952
Li J, Wang J, Jin Z (2018) SIR dynamics in random networks with communities. J Math Biol 77(4):1117. https://doi.org/10.1007/s00285-018-1247-5
Liu Z, Hu B (2005) Epidemic spreading in community networks. Europhys Lett 72(2):315. https://doi.org/10.1209/epl/i2004-10550-5
Lloyd-Smith JO, George D, Pepin KM, Pitzer VE, Pulliam J, Dobson AP, Hudson PJ, Grenfell BT (2009) Epidemic dynamics at the human–animal interface. Science 326:1362. https://doi.org/10.1126/science.1177345
Lu L, Jia M, Ma Y, Yang L, Chen Z, Ho DD, Jiang Y, Zhang L (2008) The changing face of HIV in China. Nature 455(7213):609. https://doi.org/10.1038/455609a
Luo X, Jin Z (2020) A new insight into isolating the high-degree nodes in network to control infectious diseases. Commun Nonlinear Sci Numer Simul 91:105363. https://doi.org/10.1016/j.cnsns.2020.105363
Ma X, Luo XF, Li L, Li Y, Sun GQ (2022) The influence of mask use on the spread of COVID-19 during pandemic in New York City. Results Phys 34:105224. https://doi.org/10.1016/j.rinp.2022.105224
Madar N, Kalisky T, Cohen R, ben Avraham D, Havlin S (2004) Immunization and epidemic dynamics in complex networks. Eur Phys J B 38:269. https://doi.org/10.1140/epjb/e2004-00119-8
Magal P, Seydi O, Webb G (2016) Final size of an epidemic for a two-group SIR model. SIAM J Appl Math 76(5):2042. https://doi.org/10.1137/16M1065392
Meyers LA, Pourbohloul B, Newman M, Skowronski DM, Brunham RC (2005) Network theory and SARS: predicting outbreak diversity. J Theor Biol 232(1):71. https://doi.org/10.1016/j.jtbi.2004.07.026
Miller JC (2011) A note on a paper by Erik Volz: SIR dynamics in random networks. J Math Biol 62(3):349. https://doi.org/10.1007/s00285-010-0337-9
Miller JC, Volz EM (2013) Incorporating disease and population structure into models of SIR disease in contact networks. PLoS ONE 8(8):e69162. https://doi.org/10.1371/journal.pone.0069162
Miller JC, Slim AC, Volz EM (2012) Edge-based compartmental modelling for infectious disease spread. J R Soc Interface 9(70):890. https://doi.org/10.1098/rsif.2011.0403
Momoh AA, Fügenschuh A (2018) Optimal control of intervention strategies and cost effectiveness analysis for a Zika virus model. Oper Res Health Care 18:99. https://doi.org/10.1016/j.orhc.2017.08.004
Mt V, Bickmann J, Wittkowski R (2020) Effects of social distancing and isolation on epidemic spreading: a dynamical density functional theory model. arXiv preprint arXiv:2003.13967
Newman M (2002) The spread of epidemic disease on networks. Phys Rev E Stat Nonlinear Soft Matter Phys 66(1 Pt 2):016128. https://doi.org/10.1103/PhysRevE.66.016128
Newman ME, Strogatz SH, Watts DJ (2001) Random graphs with arbitrary degree distributions and their applications. Phys Rev E 64(2):026118. https://doi.org/10.1103/PhysRevE.64.026118
Nowzari C, Preciado VM, Pappas GJ (2016) Analysis and control of epidemics: a survey of spreading processes on complex networks. IEEE Control Syst Mag 36(1):26. https://doi.org/10.1109/MCS.2015.2495000
Orman GK, Labatut V, Cherifi H (2012) An empirical study of the relation between community structure and transitivity. Workshop Complex Netw. https://doi.org/10.1007/978-3-642-30287-9_11
Pandey A, Atkins KE, Medlock J, Wenzel N, Townsend JP, Childs JE, Nyenswah TG, Ndeffo-Mbah ML, Galvani AP (2014) Strategies for containing Ebola in West Africa. Science 346(6212):991. https://doi.org/10.1126/science.1260612
Pastor-Satorras R, Vespignani A (2002) Immunization of complex networks. Phys Rev E 65:036104. https://doi.org/10.1103/PhysRevE.65.036104
Peng L, Yang W, Zhang D, Zhuge C, Hong L (2020) Epidemic analysis of COVID-19 in China by dynamical modeling. arXiv preprint arXiv:2002.06563
Rattana P, Miller JC, Kiss IZ (2014) Pairwise and edge-based models of epidemic dynamics on correlated weighted networks. Math Model Nat Phenom 9(2):58. https://doi.org/10.1051/mmnp/20149204
Rowthorn RE, Laxminarayan R, Gilligan CA (2009) Optimal control of epidemics in metapopulations. J R Soc Interface. https://doi.org/10.1098/rsif.2008.0402
Salathé M, Jones JH (2010) Dynamics and control of diseases in networks with community structure. PLoS Comput Biol. https://doi.org/10.1371/journal.pcbi.1000736
Salathé M, Jones JH (2010) Dynamics and control of diseases in networks with community structure. PLoS Comput Biol 6(4):e1000736. https://doi.org/10.1371/journal.pcbi.1000736
Singh RK, Dhama K, Khandia R, Munjal A, Karthik K, Tiwari R, Chakraborty S, Malik YS, Bueno-Marí R (2018) Prevention and control strategies to counter Zika virus, a special focus on intervention approaches against vector mosquitoes current updates. Front Microbiol 9:87. https://doi.org/10.3389/fmicb.2018.00087
Starnini M, Pastor-Satorras R (2014) Temporal percolation in activity-driven networks. Phys Rev E 89(3):032807. https://doi.org/10.1103/PhysRevE.89.032807
Strona G, Castellano C (2018) Rapid decay in the relative efficiency of quarantine to halt epidemics in networks. Phys Rev E 97:022308. https://doi.org/10.1103/PhysRevE.97.022308
Volz E (2008) SIR dynamics in random networks with heterogeneous connectivity. J Math Biol 56(3):293. https://doi.org/10.1007/s00285-007-0116-4
Xu Z, Li K, Sun M, Fu X (2019) Interaction between epidemic spread and collective behavior in scale-free networks with community structure. J Theor Biol 462:122. https://doi.org/10.1016/j.jtbi.2018.11.003
Yan X, Zou Y (2008) Optimal and sub-optimal quarantine and isolation control in SARS epidemics. Math Comput Model 47(1):235. https://doi.org/10.1016/j.mcm.2007.04.003
Yuan P, Tan Y, Yang L, Aruffo E, Ogden NH, Belair J, Arino J, Heffernan JM, Watmough J, Carabin H et al (2022) Modelling vaccination and control strategies of outbreaks of monkeypox at gatherings. Front Public Health. https://doi.org/10.3389/fpubh.2022.1026489
Zheng B, Liang L, Zhang C (2010) Extended jury criterion. Science China Math 53(4):1133. https://doi.org/10.1007/s11425-009-0208-2
Zuzek LG, Stanley HE, Braunstein LA (2015) Epidemic model with isolation in multilayer networks. Sci Rep 5(1):1. https://doi.org/10.1038/srep12151
Acknowledgements
Thank Zhenzhen Zhang, Xia Ma, Juan Zhang and Wei Zhang for their help. This research is supported by the National Natural Science Foundation of China under Grants 12101573, 12022113 and 12126416, and by Fundamental Research Program of Shanxi Province under Grants 20210302124381.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
1.1 Appendix A
In this subsection, we apply the linear system at disease-free equilibrium of Eq. (19) to verify the basic reproduction number in (25). The Jacobian matrix at disease-free equilibrium \(E_{0}=(1, 1, 1, 1)\) of model Eq. (19) is
where \(J_{11} = -(\beta _{11}+\gamma _{1})+\beta _{11}G_{11}^{'}(1)\), \(J_{44} = -(\beta _{22}+\gamma _{2})+\beta _{22}G_{22}^{'}(1)\).
Its characteristic equation is
where
As the complexity of the fourth order Eq. (30), the stability of disease-free equilibrium is hard to verify by Routh–Hurwitz criterion. However, for the type of edge-based equations, reference Rattana et al. (2014) has indicated that the case of \(a_{4}=0\) in fourth order equation (30) is equivalent to \(R_{0}=1\). Therefore, when \(a_{4}=0\) in (30), we obtain
It indirectly verifies the validity of \(R_{0}\) in (25), although the analysis lacks strict proof.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, R., Luo, X., Asamoah, J.K.K. et al. A hierarchical intervention scheme based on epidemic severity in a community network. J. Math. Biol. 87, 29 (2023). https://doi.org/10.1007/s00285-023-01964-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00285-023-01964-y