Abstract
Wolbachia-based biocontrol has recently emerged as a potential method for prevention and control of dengue and other vector-borne diseases. Major vector species, such as Aedes aegypti females, when deliberately infected with Wolbachia become less capable of getting viral infections and transmitting the virus to human hosts. In this paper, we propose an explicit sex-structured population model that describes an interaction of uninfected (wild) male and female mosquitoes and those deliberately infected with wMelPop strain of Wolbachia in the same locality. This particular strain of Wolbachia is regarded as the best blocker of dengue and other arboviral infections. However, wMelPop strain of Wolbachia also causes the loss of individual fitness in Aedes aegypti mosquitoes. Our model allows for natural introduction of the decision (or control) variable, and we apply the optimal control approach to simulate wMelPop Wolbachia infestation of wild Aedes aegypti populations. The control action consists in continuous periodic releases of mosquitoes previously infected with wMelPop strain of Wolbachia in laboratory conditions. The ultimate purpose of control is to find a tradeoff between reaching the population replacement in minimum time and with minimum cost of the control effort. This approach also allows us to estimate the number of Wolbachia-carrying mosquitoes to be released in day-by-day control action. The proposed method of biological control is safe to human health, does not contaminate the environment, does not make harm to non-target species, and preserves their interaction with mosquitoes in the ecosystem.











Similar content being viewed by others
Notes
This result was later confirmed by Hurst et al. (2012) through experiments with six natural predators of Aedes aegypti.
Such proportion is usually referred to as “infection frequency”.
We have not found any reference corroborating that wAlbB, wMel and wMelPop strains may induce the male-killing effect in Aedes aegypti populations.
This period is also referred to as “extrinsic incubation period” and may last from 2 to 33 days for dengue virus (Chan and Johansson 2012).
Generally speaking, this model is also applicable to simulate Wolbachia invasion of Aedes aegypti populations by other strains (such as wMel and wAlbB).
Here we pretend to consider the “worst scenario” by supposing that no other control actions have been carried out before starting the release program.
This quantity basically provides a ratio between the sex-specific birth and death rates of the mosquitoes.
Wolbachia may cause so-called sex ratio distortion in offspring (Kobayashi and Telschow 2010; Yamauchi et al. 2010) in some insect species. However, there is no scientific evidence regarding Aedes aegypti. Therefore, we have supposed that \(\epsilon _n \ne \epsilon _w, \) without loss of generality.
More details and examples regarding the principle of competitive exclusion can be found in, e.g., Brauer and Castillo-Chávez (2012) or similar textbooks.
Rockwood (2015) defines minimum viable population size as the lower bound of population densities that are necessary for survival and/or persistence of biological species.
See the underlying arguments and more details in “Appendix A”.
Strictly speaking, the terminal endpoint condition (27) should be of the form \(F_n \left( T^{*} \right) =0\). However, given the exponential nature of state Eqs. (29), its trajectories may only approach zero asymptotically when \(t \rightarrow \infty \) but they cannot reach this value in finite time.
Although egg diapause is not very common in Aedes aegypti (Denlinger and Armbruster 2014), there is scientific evidence that under untypical climate conditions (lack of water, extremely low or high humidity and/or temperature, high insolation, etc.) the quiescence of Aedes aegypti eggs may extend for 6 months or more (Soares-Pinheiro et al. 2017).
The idea to use different values for constant \(C >0\) stems from the lack of information regarding the monetary costs related to artificial breeding and posterior releases of Wolbachia-carrying mosquitoes. Nonetheless, even without knowing these monetary costs we further propose two options for decision making in Sect. 4 and solve numerically the optimal control problem (28)–(29) with end-point conditions (25) and (27).
In some studies, marginal costs of control actions are supposed independent of the control variable or even constant. However, our definition of the control variable u(t) as a fraction of Wolbachia-infected population present in the target locality at each day t does not allow for such simplifications. Namely, by taking u(t) instead of \( \dfrac{1}{2} u^2(t)\) in the objective functional (28), marginal cost of the control action would be independent of the control variable and equal to \(C \big [ M_w (t) + F_w(t) \big ]\). The latter has nothing to do with releasing Wolbachia-infected mosquitoes and merely stands for the number of Wolbachia-carriers already present in the target locality at the day t (\(M_w (t) + F_w(t)\)), multiplied by \(C>0\).
It is easy to verify that \(\dfrac{ \partial ^2 H}{ \partial u^{2}}= - C (M_w + F_w) \le 0\) for all admissible u.
It is worthwhile to note that the four adjoint equations are linear with respect to adjoint variables \(\lambda _i, i=1,2,3,4\).
For more information regarding GPOPS-II solver please visit http://gpops2.com/.
GPOPS-II scales automatically all input intervals [0, T] to the interval \([-1,1]\) (see more detailed information in “Appendix B”).
The basic reproductive number is usually defined (see Diekmann and Heesterbeek (2000)) as the average number of secondary cases produced by a “typical” infected (assumed infectious) individual during his/her entire life as infectious (infectious period) when introduced in a population of susceptibles.
Matrix \(\mathcal {M}\) expresses the disease transmission part, i.e. the emergence of new infections, while D represents the disease transition.
This set includes one of the endpoints (that is, either \(-1\) or 1) and the roots of \(P_{K-1}(\tau ) + P_K (\tau )\), where \(P_K(\tau )\) denotes the Legendre polynomial of degree K.
References
Ascher UM, Mattheij RMM, Russell RD (1988) Numerical solution of boundary value problems for ordinary differential equations. Prentice Hall series in computational mathematics. Prentice Hall Inc., Englewood Cliffs
Barton N, Turelli M (2011) Spatial waves of advance with bistable dynamics: cytoplasmic and genetic analogues of Allee effects. Am Nat 178(3):E48–E75
Bian G, Xu Y, Lu P, Xie Y, Xi Z (2010) The endosymbiotic bacterium Wolbachia induces resistance to dengue virus in Aedes aegypti. PLoS Pathog 6(4):e1000,833
Blayneh K, Cao Y, Kwon HD (2009) Optimal control of vector-borne diseases: treatment and prevention. Discrete Contin Dyn Syst B 11(3):587–611. https://doi.org/10.3934/dcdsb.2009.11.587
Bliman PA, Aronna MS, da Silva MA, et al (2015) Global stabilizing feedback law for a problem of biological control of mosquito-borne diseases. In: 2015 54th IEEE conference on decision and control (CDC). IEEE, pp 3206–3211
Brauer F, Castillo-Chávez C (2012) Mathematical models in population biology and epidemiology. Texts Appl Math. https://doi.org/10.1007/978-1-4614-1686-9
Brown JE, McBride CS, Johnson P, Ritchie S, Paupy C, Bossin H, Lutomiah J, Fernandez-Salas I, Ponlawat A, Cornel AJ, Black WC, Gorrochotegui-Escalante N, Urdaneta-Marquez L, Sylla M, Slotman M, Murray KO, Walker C, Powell JR (2011) Worldwide patterns of genetic differentiation imply multiple “domestications” of Aedes aegypti, a major vector of human diseases. Proc R Soc B Biol Sci 278(1717):2446–2454
Bryson A, Ho YC (1975) Applied optimal control: optimization, estimation and control. Halsted Press Book, New York
Bull JJ, Turelli M (2013) Wolbachia versus dengue evolutionary forecasts. Evol Med Public Health 2013(1):197–207
Campo-Duarte DE, Cardona-Salgado D, Vasilieva O (2017a) Establishing wMelPop Wolbachia infection among wild Aedes aegypti females by optimal control approach. Appl Math Inf Sci 11(4):1011–1027. https://doi.org/10.18576/amis/110408
Campo-Duarte DE, Vasilieva O, Cardona-Salgado D (2017b) Optimal control for enhancement of Wolbachia frequency among Aedes aegypti females. Int J Pure Appl Math 112(2):219–238. https://doi.org/10.12732/ijpam.v112i2.1
Castillo-Chávez C, Feng Z, Huang W (2002) On the computation of Ro and its role on global stability. In: Castillo-Chávez C, Blower S, Driessche P, Kirschner D, Yakubu A (eds) Mathematical approaches for emerging and reemerging infectious diseases: an introduction, vol 125. Springer, Berlin, pp 229–250
Caswell H, Weeks DE (1986) Two-sex models: chaos, extinction, and other dynamic consequences of sex. Am Nat 128(5):707–735
Chan M, Johansson MA (2012) The incubation periods of dengue viruses. PloS ONE 7(11):e50,972
Coelho FC, Codeço CT, Gomes MGM (2011) A Bayesian framework for parameter estimation in dynamical models. PloS ONE 6(5):e19,616
Denlinger DL, Armbruster PA (2014) Mosquito diapause. Ann Rev Entomol 59:73–93
Diekmann O, Heesterbeek J (2000) Mathematical epidemiology of infectious diseases: model building, analysis and interpretation. Wiley, Hoboken Wiley Series in Mathematical & Computational Biology
Diekmann O, Heesterbeek J, Metz J (1990) On the definition and the computation of the basic reproduction ratio Ro in models for infectious diseases in heterogeneous populations. J Math Biol 28(4):365–382
Domínguez MC, Ludueña-Almeida FF, Almirón WR (2000) Dinámica poblacional de Aedes aegypti (Diptera: Culicidae) en Córdoba capital [Population dynamics of Aedes aegypti (Diptera: Culicidae) in Cordoba capital]. Revista de la Sociedad Entomológica Argentina 59(1–4):41–50
Dutra HLC, Rocha MN, Dias FBS, Mansur SB, Caragata EP, Moreira LA (2016) Wolbachia blocks currently circulating Zika virus isolates in Brazilian Aedes aegypti mosquitoes. Cell Host Microbe 19(6):771–774
Dye C (1984) Models for the population dynamics of the yellow fever mosquito, Aedes aegypti. J Anim Ecol 53(1):247–268
Farkas JZ, Hinow P (2010) Structured and unstructured continuous models for Wolbachia infections. Bull Math Biol 72(8):2067–2088
Farkas JZ, Gourley SA, Liu R, Yakubu AA (2017) Modelling Wolbachia infection in a sex-structured mosquito population carrying West Nile virus. J Math Biol 75(3):621–647. https://doi.org/10.1007/s00285-017-1096-7
Farkas M (2001) Dynamical models in biology. Elsevier, Amsterdam
Ferguson NM, Kien DTH, Clapham H, Aguas R, Trung VT, Chau TNB, Popovici J, Ryan PA, O’Neill SL, McGraw EA, Long VT, Dui LT, Nguyen HL, Vinh Chau NV, Wills B, Simmons CP (2015) Modeling the impact on virus transmission of Wolbachia-mediated blocking of dengue virus infection of Aedes aegypti. Sci Transl Med 7(279):279ra37–279ra37
Fleming W, Rishel R (1975) Deterministic and stochastic optimal control. Springer, New York
Frentiu FD, Walker T, O’Neill SL (2014a) Biological control of dengue and Wolbachia-based strategies. In: Gubler D, Ooi E, Vasudevan S, Farrar J (eds) Dengue and dengue hemorrhagic fever, 2nd edn. CAB Books, CABI
Frentiu FD, Zakir T, Walker T, Popovici J, Pyke AT, van den Hurk A, McGraw EA, O’Neill SL (2014b) Limited dengue virus replication in field-collected Aedes aegypti mosquitoes infected with Wolbachia. PLoS Negl Trop Dis 8(2):e2688
Garg D, Patterson MA, Darby CL, Francolin C, Huntington GT, Hager WW, Rao AV (2009) Direct trajectory optimization and costate estimation of general optimal control problems using a Radau pseudospectral method. In: Proceedings of the AIAA guidance, navigation, and control conference and exhibit
Hancock PA, Godfray HCJ (2012) Modelling the spread of Wolbachia in spatially heterogeneous environments. J R Soc Interface 9(76):3045–3054
Hancock PA, Sinkins SP, Godfray HCJ (2011a) Population dynamic models of the spread of Wolbachia. Am Nat 177(3):323–333
Hancock PA, Sinkins SP, Godfray HCJ (2011b) Strategies for introducing Wolbachia to reduce transmission of mosquito-borne diseases. PLoS Negl Trop Dis 5(4):e1024
Hilgenboecker K, Hammerstein P, Schlattmann P, Telschow A, Werren JH (2008) How many species are infected with Wolbachia?—A statistical analysis of current data. FEMS Microbiol Lett 281(2):215–220
Hoffmann A, Montgomery B, Popovici J, Iturbe-Ormaetxe I, Johnson P, Muzzi F, Greenfield M, Durkan M, Leong Y, Dong Y, Cook H, Axford J, Callahan A, Kenny N, Omodei C, McGraw E, Ryan P, Ritchie S, Turelli M, O’Neill S (2011) Successful establishment of Wolbachia in Aedes populations to suppress dengue transmission. Nature 476(7361):454–457
Hoffmann AA (2014) Facilitating Wolbachia invasions. Austral Entomol 53(2):125–132
Hoffmann AA, Turelli M (2013) Facilitating Wolbachia introductions into mosquito populations through insecticide-resistance selection. Proc R Soc Lond B Biol Sci 280(1760):20130371
Hughes H, Britton NF (2013) Modelling the use of Wolbachia to control dengue fever transmission. Bull Math Biol 75(5):796–818
Hurst TP, Pittman G, O’Neill SL, Ryan PA, Le Nguyen H, Kay BH (2012) Impacts of Wolbachia infection on predator prey relationships: evaluating survival and horizontal transfer between wMelPop infected Aedes aegypti and its predators. J Med Entomol 49(3):624–630
Jansen CC, Beebe NW (2010) The dengue vector Aedes aegypti: what comes next. Microbes Infect 12(4):272–279
Keyfitz N (1972) The mathematics of sex and marriage. In: Proceedings of the sixth Berkeley symposium on mathematical statistics and probability, vol 4. University of California Press, Berkeley, pp 89–108
Kobayashi Y, Telschow A (2010) Cytoplasmic feminizing elements in a two-population model: infection dynamics, gene flow modification, and the spread of autosomal suppressors. J Evolut Biol 23(12):2558–2568
Koiller J, Da Silva M, Souza M, Codeço C, Iggidr A, Sallet G (2014) Aedes, Wolbachia and Dengue. Technical report
Kot M (2001) Elements of mathematical ecology. Cambridge University Press, Cambridge
Kroese DP, Taimre T, Botev ZI (2011) Handbook of Monte Carlo methods, vol 706. Wiley series in probability and statistics. Wiley, Hoboken
Lawson AB (2006) Statistical methods in spatial epidemiology, 2nd edn. Wiley series in probability and statistics. Wiley, Hoboken
Lenhart S, Workman JT (2007) Optimal control applied to biological models. Chapman & Hall/CRC, Boca Raton
Liles JN (1965) Effects of mating or association of the sexes on longevity in Aedes aegypti (l.). Mosquito News 25:434–439
Lindström J, Kokko H (1998) Sexual reproduction and population dynamics: the role of polygyny and demographic sex differences. Proc R Soc Lond B Biol Sci 265(1395):483–488
Manore CA, Hickmann KS, Xu S, Wearing HJ, Hyman JM (2014) Comparing dengue and chikungunya emergence and endemic transmission in A. aegypti and A. albopictus. J Theor Biol 356:174–191
McGraw EA, O’Neill SL (2013) Beyond insecticides: new thinking on an ancient problem. Nat Rev Microbiol 11(3):181–193. https://doi.org/10.1038/nrmicro2968
McMeniman C et al (2009) Stable introduction of a life-shortening Wolbachia infection into the mosquito Aedes aegypti. Science 323(5910):141–144
McMeniman CJ, O’Neill SL (2010) A virulent Wolbachia infection decreases the viability of the dengue vector Aedes aegypti during periods of embryonic quiescence. PLoS Negl Trop Dis 4(7):e748
Moreira LA, Iturbe-Ormaetxe I, Jeffery JA, Lu G, Pyke AT, Hedges LM, Rocha BC, Hall-Mendelin S, Day A, Riegler M et al (2009) A Wolbachia symbiont in Aedes aegypti limits infection with dengue, chikungunya, and plasmodium. Cell 139(7):1268–1278
Moulay D, Aziz-Alaoui MA, Kwon HD (2012) Optimal control of chikungunya disease: larvae reduction, treatment and prevention. Math Biosci Eng 9(2):369–392. https://doi.org/10.3934/mbe.2012.9.369
Ndii MZ, Hickson R, Allingham D, Mercer G (2015) Modelling the transmission dynamics of dengue in the presence of Wolbachia. Math Biosci 262:157–166
Nguyen T, Le Nguyen H, Nguyen T, Vu S, Tran N, Le T, Vien Q, Bui T, Le H, Kutcher S, Hurst T, Duong T, Jeffery J, Darbro J, Kay H, Iturbe-Ormaetxe I, Popovici J, Montgomery B, Turley A, Zigterman F, Cook H, Cook P, Johnson P, Ryan P, Paton C, Ritchie S, Simmons C, O’Neill S, Hoffmann A (2015) Field evaluation of the establishment potential of wMelPop Wolbachia in Australia and Vietnam for dengue control. Parasit Vectors 8(1):1
Okosun KO, Ouifki R, Marcus N (2011) Optimal control analysis of a malaria disease transmission model that includes treatment and vaccination with waning immunity. Biosystems 106(2):136–145. https://doi.org/10.1016/j.biosystems.2011.07.006
Okosun KO, Rachid O, Marcus N (2013) Optimal control strategies and cost-effectiveness analysis of a malaria model. BioSystems 111(2):83–101. https://doi.org/10.1016/j.biosystems.2012.09.008
Patterson MA, Rao AV (2014) GPOPS-II: A MATLAB software for solving multiple-phase optimal control problems using hp-adaptive Gaussian quadrature collocation methods and sparse nonlinear programming. ACM Trans Math Softw (TOMS) 41(1):1
Popovici J, Moreira LA, Poinsignon A, Iturbe-Ormaetxe I, McNaughton D, O’Neill SL (2010) Assessing key safety concerns of a Wolbachia-based strategy to control dengue transmission by Aedes mosquitoes. Memórias do Instituto Oswaldo Cruz 105(8):957–964
Ritchie SA, Montgomery BL, Hoffmann AA (2013) Novel estimates of Aedes aegypti (Diptera: Culicidae) population size and adult survival based on Wolbachia releases. J Med Entomol 50(3):624–631
Ritchie SA, Townsend M, Paton CJ, Callahan AG, Hoffmann AA (2015) Application of wMelPop Wolbachia strain to crash local populations of Aedes aegypti. PLoS Negl Trop Dis 9(7):e0003930
Roberts SM, Shipman JS (1972) Two-point boundary value problems: shooting methods. In: Modern analytic and computational methods in science and mathematics, vol 31. Elsevier, New York
Rockwood LL (2015) Introduction to population ecology, 2nd edn. Wiley, Hoboken
Ross PA, Endersby NM, Yeap HL, Hoffmann AA (2014) Larval competition extends developmental time and decreases adult size of wMelPop Wolbachia-infected Aedes aegypti. Am J Trop Med Hyg 91(1):198–205
Ruang-Areerate T, Kittayapong P (2006) Wolbachia transinfection in Aedes aegypti: a potential gene driver of dengue vectors. Proc Natl Acad Sci 103(33):12534–12539
Schraiber JG, Kaczmarczyk AN, Kwok R, Park M, Silverstein R, Rutaganira FU, Aggarwal T, Schwemmer MA, Hom CL, Grosberg RK et al (2012) Constraints on the use of lifespan-shortening Wolbachia to control dengue fever. J Theor Biol 297:26–32
Sepúlveda LS, Vasilieva O (2016) Optimal control approach to dengue reduction and prevention in Cali, Colombia. Math Methods Appl Sci 39(18):5475–5496
Sepúlveda-Salcedo LS, Vasilieva O, Martínez-Romero HJ, Arias-Castro JH (2015) Ross-Macdonald: Un modelo para la dinámica del dengue en Cali. Colombia. Revista de Salud Pública 17(5):749–761
Sinkins SP (2004) Wolbachia and cytoplasmic incompatibility in mosquitoes. Insect Biochem Mol Biol 34(7):723–729
Sinkins SP (2013) Wolbachia and arbovirus inhibition in mosquitoes. Future Microbiol 8(10):1249–1256
Soares-Pinheiro V, Dasso-Pinheiro W, Trindade-Bezerra J, Tadei W (2017) Eggs viability of Aedes aegypti Linnaeus (Diptera, Culicidae) under different environmental and storage conditions in Manaus, Amazonas. Brazil. Braz J Biol 77(2):396–401
Styer LM, Minnick SL, Sun AK, Scott TW (2007) Mortality and reproductive dynamics of Aedes aegypti (Diptera: Culicidae) fed human blood. Vector Borne Zoonotic Dis 7(1):86–98
Telschow A, Flor M, Kobayashi Y, Hammerstein P, Werren JH (2007) Wolbachia-induced unidirectional cytoplasmic incompatibility and speciation: mainland-island model. PLoS ONE 2(8):e701
Thieme H (2003) Mathematics in population biology. Mathematical biology series. Princeton University Press, Princeton
Turelli M (2010) Cytoplasmic incompatibility in populations with overlapping generations. Evolution 64(1):232–241
Turelli M, Hoffmann AA (1991) Rapid spread of an inherited incompatibility factor in California Drosophila. Nature 353(6343):440–442
Walker T, Johnson P, Moreira L, Iturbe-Ormaetxe I, Frentiu F, McMeniman C, Leong Y, Dong Y, Axford J, Kriesner P, Lloyd A, Ritchie S, O’Neill S, Hoffmann A (2011) The wMel Wolbachia strain blocks dengue and invades caged Aedes aegypti populations. Nature 476(7361):450–453
Williams CR, Johnson P, Ball T, Ritchie S (2013) Productivity and population density estimates of the dengue vector mosquito Aedes aegypti (Stegomyia aegypti) in Australia. Med Vet Entomol 27(3):313–322
Woolfit M, Iturbe-Ormaetxe I, Brownlie JC, Walker T, Riegler M, Seleznev A, Popovici J, Rancès E, Wee BA, Pavlides J et al (2013) Genomic evolution of the pathogenic Wolbachia strain, wMelPop. Genome Biol Evol 5(11):2189–2204
Xi Z, Khoo CC, Dobson SL (2005) Wolbachia establishment and invasion in an Aedes aegypti laboratory population. Science 310(5746):326–328
Yamauchi A, Telschow A, Kobayashi Y (2010) Evolution of cytoplasmic sex ratio distorters: Effect of paternal transmission. J Theoret Biol 266(1):79–87
Yeap HL, Mee P, Walker T, Weeks AR, O’Neill SL, Johnson P, Ritchie SA, Richardson KM, Doig C, Endersby NM, Hoffmann AA (2011) Dynamics of the “popcorn” Wolbachia infection in outbred Aedes aegypti informs prospects for mosquito vector control. Genetics 187(2):583–595
Yeap HL, Axford JK, Popovici J, Endersby NM, Iturbe-Ormaetxe I, Ritchie SA, Hoffmann AA (2014) Assessing quality of life-shortening Wolbachia-infected Aedes aegypti mosquitoes in the field based on capture rates and morphometric assessments. Parasites Vectors 7(58):1–13. https://doi.org/10.1186/1756-3305-7-58
Acknowledgements
All authors acknowledge the support of COLCIENCIAS (Colombian Ministry of Science, Technology, and Innovation) by way of Research Grant No. 12595-6933846 (2014–2017). Doris E. Campo-Duarte and Olga Vasilieva acknowledge support from COLCIENCIAS and Universidad del Valle (Research Project CI-71089), while Daiver Cardona-Salgado also appreciates the endorsement obtained from the STIC AmSud Program for regional cooperation (16-STIC-02 MOSTICAW Project).
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A: Meaning of \(\mathcal {R}_0\) in Proposition 1
In order to reveal the meaning of threshold \(\mathcal {R}_0\) given by formula (18) in Proposition 1, we propose to follow the next generation operator approach attributed to Diekmann et al. (1990), which was further presented by Castillo-Chávez et al. (2002) in a form of comprehensive tutorial.
In fact, since Wolbachia symbiont is maternally transmitted from a female mosquito to her offspring, the next generation produced by this female will be infected with Wolbachia. Thus, Wolbachia infection can be viewed as a maternally transmitted disease among the mosquito population. The steady state \(E_n^{\sharp } = \big ( M_n^{\sharp }, F_n^{\sharp }, 0, 0 \big ) \) can be regarded as a disease-free equilibrium in epidemiological terminology. The threshold value \(\mathcal {R}_0\) that determines whether the epidemics vanishes or spreads is usually referred to as basic reproductive number.Footnote 21 Diekmann et al. (1990) defined \(\mathcal {R}_0\) as the spectral radius of the “next generation operator”, while Castillo-Chávez et al. (2002) provide a method to calculate this spectral radius in case of discrete heterogeneity, i. e. when the population of individuals is subdivided into several groups with fixed characteristics. According to this approach, the ODE system (10)–(11) can be re-written in the following form:
where \(\varvec{Y}= \big ( M_n, F_n \big ) \in \mathbb {R}^2_+\) couples the uninfected individuals, while \(\varvec{Z}= \big ( M_w, F_w \big )\in \mathbb {R}^2_+\) engages Wolbachia-infected individuals. Since \(E_n^{\sharp } = \big ( M_n^{\sharp }, F_n^{\sharp }, 0, 0 \big )= \big ( \varvec{Y}^{\sharp }, \mathbf{0})\) stands for infection-free equilibrium, we have \(\varvec{f}(\varvec{Y}^{\sharp },\mathbf{0}) = \varvec{g}(\varvec{Y}^{\sharp },\mathbf{0}) = \mathbf{0}.\) Let
and further assume that A can be written in the form \(A = \mathcal {M} - D\), with \(\mathcal {M} \ge 0\) (that is, \(m_{ij} \ge 0\)) and \(D > 0\), a diagonal matrix.Footnote 22 Under this assumption
Then, according to Castillo-Chávez et al. (2002), \(\mathcal {R}_0\) can be defined as the spectral radius of
Namely,
is the dominant eigenvalue of \(\mathcal {M} D^{-1}\). By replacing the values of \(M_n^{\sharp }\) and \(F_n^{\sharp }\) from (14a), we obtain that
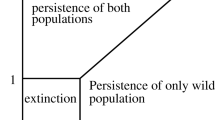
only depends on female-related parameters. The latter is quite logical since all Wolbachia-infected females are “spreaders” of the disease. In view of conditions (12), we have \(\mathcal {R}_0 < 1\) (cf. (18)), that is, the “birth-death ratio” of Wolbachia-infected females (numerator in the right-hand side of (18)) is less than the “birth-death ratio” of uninfected females (denominator in the right-hand side of (18)). In other words, Wolbachia-infected females exhibit reduced individual fitness in comparison to wild females. If we suppose that both infected and uninfected females have the same chances for successful mating that results in viable offspring (i.e., uninfected males and females have sufficiently high frequencies), then at each next generation of mosquitoes there will be a lesser fraction of Wolbachia-infected individuals and a greater fraction of uninfected individuals. Under this scenario, all Wolbachia-carriers will be eventually driven towards extinction, and the disease-free equilibrium \(E_n^{\sharp } = \big ( M_n^{\sharp }, F_n^{\sharp }, 0, 0 \big )\) will be reached.
On the other hand, reduced individual fitness of Wolbachia-infected females is recompensed, at high infection frequencies, by their reproductive advantage derived from CI-phenotype. The latter is explained by Proposition 1.
Hypothetically speaking, if we disregard the conditions (12) and suppose that Wolbachia does not alter neither fecundity nor longevity of the host (that is, \(\epsilon _n=\epsilon _w, \rho _n=\rho _w, \mu _n = \mu _w, \delta _n=\delta _w \) so that \(\mathcal {R}_0 = 1\)), then Wolbachia invasion could be induced by a single release of very small number of Wolbachia-carriers, and this “imaginary” scenario also agrees with our model (10)–(11), as well as with the principle of competitive exclusion.
Appendix B: Technical note on GPOPS-II solver for MATLAB platform
Generally speaking, there are two groups of collocation techniques — the direct collocation and the orthogonal collocation. Under the direct collocation approach, the state and adjoint trajectories of the optimality system are discretized at a set of appropriately chosen grid of nodes (that is, collocation points) in the fixed time interval \([t_0, t_f]\). Then the state and adjoint trajectories are iteratively approximated using the same fixed-degree polynomials (usually, cubic splines) at all subinterval of the grid in order to satisfy the differential constraints (i.e., the discretized ODE system), while the boundary constraints are taken into account at each iteration. Convergence of all methods based on direct collocation is usually achieved by increasing the number of collocation points.
On the other hand, the orthogonal collocation is performed over entire time interval \([t_0, t_f]\) where the collocation points are usually associated with Gaussian quadrature, i.e. they are roots of some orthogonal polynomial, or a linear combination of orthogonal polynomials and its derivatives. Under this approach, the state and adjoint variables of the continuous-time optimality system (as well as their derivatives) are approximated by using Lagrange interpolating polynomials over \([t_0, t_f]\) supported by the collocation points. Convergence of this method can be achieved by increasing the degree of the polynomial approximation.
The GPOPS-II solver implements an adaptive combination of two collocation techniques described above which is also known as Radau pseudospectral method. According to Garg et al. (2009), the Radau pseudospectral method is capable of dealing with free initial \(t_0\) or final \(t_f\) time since the input time interval \(t \in [t_0, t_f]\) (which is in our case is \(t \in [0,T]\) with T left free) should be transformed into \(\tau \in [-1, 1]\) (with fixed endpoints!) using the affine transformation
In particular, this method uses the Legendre-Gauss-Radau setFootnote 23 of orthogonal collocation points what basically explains its name.
Another advantage of Radau pseudospectral method, besides its adaptiveness, is that this method (in contrast to other pseudospectral methods) allows to use the exact formulas for first- and second-order partial derivatives of problem entries and, thus, to solve more accurately the nonlinear programming problems resulting from discretization.
The main limitation of GPOPS-II package is that it requires the continuity of the first- and second-order derivatives of the Hamiltonian and endpoint constraints with respect to all variables. However, the models considered in this paper meet this condition.
Further and more detailed information regarding the GPOPS-II package, as well as some comprehensive examples, can be consulted in the paper by Patterson and Rao (2014).
Rights and permissions
About this article
Cite this article
Campo-Duarte, D.E., Vasilieva, O., Cardona-Salgado, D. et al. Optimal control approach for establishing wMelPop Wolbachia infection among wild Aedes aegypti populations. J. Math. Biol. 76, 1907–1950 (2018). https://doi.org/10.1007/s00285-018-1213-2
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-018-1213-2
Keywords
- Wolbachia-based biocontrol
- wMelPop strain
- Aedes aegypti
- Sex-structured model
- Optimal control
- Optimal release policies