Abstract
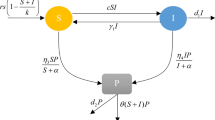
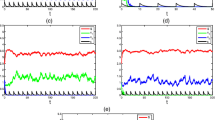
Recent research indicates that viruses are much more prevalent in aquatic environments than previously imagined. We derive a model of competition between two populations of bacteria for a single limiting nutrient in a chemostat where a virus is present. It is assumed that the virus can only infect one of the populations, the population that would be a more efficient consumer of the resource in a virus free environment, in order to determine whether introduction of a virus can result in coexistence of the competing populations. We also analyze the subsystem that results when the resistant competitor is absent. The model takes the form of an SIS epidemic model. Criteria for the global stability of the disease free and endemic steady states are obtained for both the subsystem as well as for the full competition model. However, for certain parameter ranges, bi-stability, and/or multiple periodic orbits is possible and both disease induced oscillations and competition induced oscillations are possible. It is proved that persistence of the vulnerable and resistant populations can occur, but only when the disease is endemic in the population. It is also shown that it is possible to have multiple attracting endemic steady states, oscillatory behavior involving Hopf, saddle-node, and homoclinic bifurcations, and a hysteresis effect. An explicit expression for the basic reproduction number for the epidemic is given in terms of biologically meaningful parameters. Mathematical tools that are used include Lyapunov functions, persistence theory, and bifurcation analysis.
Similar content being viewed by others
References
Aris R, Humphrey AE (1977) Dynamics of a chemostat in which two organisms compete for a common substrate. Biotechnol Bioeng 19: 1375–1386
Barbălat I (1957) Systemes d’equations differentielle d’oscillations nonlineaires. Rom Math Pures Appl 4: 267–270
Beretta E, Kuang Y (2001) Modeling and analysis of a marine bacteriophage infection with latency period. Nonlinear Anal Real World Appl 2: 35–74
Beretta E, Carletti M, Solimano F (2000) On the effects of environmental fluctuations in a simple model of bacteria-bacteriophage infection. Can Appl Math Q 8: 321–366
Bergh O, Borsheim KY, Bratbak G, Heldal M (1989) High abundance of viruses found in aquatic environments. Nature 340: 467–468
Bratbak G, Heldal M (2000) Viruses rule the waves—the smallest and most abundant members of marine ecosystems. Microbiol Today 27: 171–173
Brussaard CPD (2003) Role of viruses in controlling phytoplankton blooms. In: Ecology of marine viruses. CIEMS workshop monographs, vol 21. CIEMS
Butler GJ, Wolkowicz GSK (1985) A mathematical model of the chemostat with a general class of functions describing nutrient uptake. SIAM J Appl Math 45: 138–151
Campbell A (1961) Conditions for the existence of bacteriophage. Evolution 15: 153–165
Canchaya C, Proux C, Fournous G, Bruttin A, Brüssow H (2003) Prophage genomics. Microbiol Mol Biol Rev 67: 238–276
Cavanaugh CM, McKiness ZP, Newton ILG, Stewart FJ (2006) Marine chemosynthetic symbioses. In: Dworkin M, Falkow S, Rosenberg E, Schleifer KH, Stackebrandt E (eds) The prokaryotes, 3rd edn. A handbook on the biology of bacteria: symbiotic associations, biotechnology, applied microbiology. Springer, New York, pp 475–507
Daoussis SP (1992) Predator mediated competition: predator feeding on two different trophic levels. Master’s thesis, McMaster University, Hamilton, ON, January 1992
Ermentrout B (2002) Simulating, analyzing, and animating dynamical systems: a guide to XPPAUT for researchers and students. SIAM, Philadelphia
Hsu S-B (1978) Limiting behavior for competing species. SIAM J Appl Math 34: 760–763
Hsu SB, Hubbell S, Waltman P (1977) A mathematical theory for single-nutrient competition in continuous cultures of micro-organisms. SIAM J Appl Math 32: 366–383
Hurwitz A (1895) On the conditions under which an equation has only roots with negative real parts. Math Ann 46: 273–284
Imran M, Smith HL (2007) A mathematical model of gene transfer in a biofilm. In: Mathematics for ecology and environmental sciences. Biological and medical physics, biomedical engineering, vol 1. Springer, Berlin, pp 93–123
Larsen A, Castberg T, Sandaa RA, Brussaard CPD, Egge J, Heldal M, Paulino A, Thyrhaug R, van Hannen EJ, Bratbak G (2001) Population dynamics and diversity of phytoplankton, bacteria and viruses in a seawater enclosure. Mar Ecol Prog Ser 221: 47–57
Li B (1999) Global asymptotic behavior of the chemostat: general response functions and differential death rates. SIAM J Appl Math 59: 411–422
Lwoff A (1953) Lysogeny. Bacteriol Rev 17: 269–337
Mestivier D, Pakdaman K, Boelle PY, Nicolas JC, Lebaron P (2003) Viral regulation of bacterial biodiversity. In: Ecology of marine viruses. CIEMS workshop monographs, vol 21
Proctor LM, Fuhrman JA (1990) Viral mortality of marine bacteria and cyanobacteria. Nature 343: 60–62
Qiu Z (2007) The analysis and regulation for the dynamics of a temperate bacteriophage model. Math Biosci 209: 417–450
Routh EJ (1877) A treatise on the stability of motion. Macmillan, New York
Shabir AD, Alfons J, Kuenen JG, Gerard M (2007) Co-existence of physiologically similar sulfate-reducing bacteria in a full-scale sulfidogenic bioreactor fed with a single organic electron donor. Appl Microbiol Biotechnol 75: 1463–1472
Smith HL (2008) Models of virulent phage growth with application to phage therapy. SIAM J Appl Math 68: 1717–1737
Smith HL, Waltman P (1995) The theory of the chemostat. Cambridge University Press, New York
Smith HL, Waltman P (1999) Perturbation of a globally stable steady state. Proc Am Math Soc 127: 447–453
Stewart MF, Levin BR (1977) The population biology of bacterial plasmids: a priori conditions for the existence of conjugationally transmitted factors. Genetics 87: 209–228
Suttle CA, Chan AM, Cottrell MT (1990) Infection of phytoplankton by viruses and reduction of primary productivity. Nature 347: 467–469
Tapper MA, Hicks RE (1998) Temperate viruses and lysogeny in lake superior bacterioplankton. Limnol Oceanogr 43: 95–103
The Mathworks Inc. (2010) Matlab 2010
Thieme HR (1993) Persistence under relaxed point-dissipativity. SIAM J Math Anal 24: 407–435
van den Driessche P, Zeeman ML (2004) Disease induced oscillations between two competing species. SIAM J Appl Math 3: 601–619
Weitz JS, Hartman H, Levin SA (2005) Coevolutionary arms races between bacteria and bacteriophage. Proc Natl Acad Sci USA 102: 9535–9540
Westwater C, Kasman LM, Schofield DA, Werner PA, Dolan JW, Schmidt MG, Norris JS (2003) Use of genetically engineered phage to deliver antimicrobial agents to bacteria: an alternative therapy for treatment of bacterial infections. Antimicrob Agents Chemother 47: 1301–1307
Wolkowicz GSK, Lu Z (1992) Global dynamics of a mathematical model of competition in the chemostat: general response functions and differential death rates. SIAM J Appl Math 52: 222–233
Wolkowicz GSK, Ballyk MM, Daoussis SP (1995) Interaction in a chemostat: introduction of a competitor can promote greater diversity. Rocky Mt J Math 25: 515–543
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Northcott, K., Imran, M. & Wolkowicz, G.S.K. Competition in the presence of a virus in an aquatic system: an SIS model in the chemostat. J. Math. Biol. 64, 1043–1086 (2012). https://doi.org/10.1007/s00285-011-0439-z
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-011-0439-z
Keywords
- SIS epidemic model
- Lytic and lysogenic virus
- Hopf, homoclinic, and saddle-node bifurcations
- Bi-stability and hysteresis effect
- Multiple limit cycles
- Biodiversity