Abstract
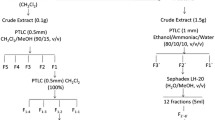
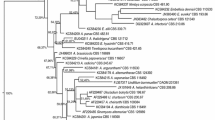
A new antagonistic Burkholderia strain, designated MP-1 and producing antifungal activities against various filamentous plant pathogenic fungi, was isolated from the rhizoshere in the Naju area. Cultural characteristic studies strongly suggested that this strain belongs to the genus Burkholderia. The nucleotide sequence of the 16S rRNA gene (1491 pb) of strain MP-1 exhibited close similarity (99% to 100%) with other Burkholderia 16S rRNA genes. Extraction of fermentation broth of Burkholderia sp. MP-1 and various separations and purification steps led to isolation of four pure active molecules. The chemical structure of these four compounds—named phenylacetic acid, hydrocinnamic acid, 4-hydroxyphenylacetic acid, and 4-hydroxyphenylacetate methyl ester—was established on the basis on their gas chromatography–electron impact–mass spectrometry (GC-EI-MS) and trimethylsilation GC-EI-MS data. The four isolated compounds inhibited filamentous fungal growth on potato dextrose agar medium supplemented with 100 mg/L of phenylacetic acid, hydrocinnamic acid, 4-hydroxyphenylacetic acid and 4-hydroxyphenylacetate methyl ester individually.





Similar content being viewed by others
Literature Cited
Arima K, Imanaka H, Kousaka M, Fukuda A, Tamura G (1965) Studies on pyrrolnitrin, a new antibiotic. I. Isolation and properties of pyrrolnitrin. J Antibiot 18:201–204
Burhead KD, Slininger PJ, Schisler DA (1998) Biological control bacterium Enterobacter cloacae S11:T:07 (NRRL B-21050) produces the antifungal compound phenylacetic acid in sabouraur maltose broth culture. Soil Biol Biochem 30:665–667
Byung KH, Song WL, Beom SK, Jung YL, Surk SM (2001) Isolation and in vitro and in vivo antifungal activity of phenylacetic acid and sodium phenylacetate from Streptomyces humidus. Appl Environ Microbiol 67:3739–3745
Camirand A, Phipps J, Wightman F (1983) Comparative metabolism of L-tyrosine and L-phenylalanine in tobacco plants in relation to the biosynthesis of phenylacetic acids. Can J Bot 61:2302–2308
Cartwright DK, Benson DM (1995) Comparison of Pseudomonas species and application techniques for biocontrol of Rhizoctonia stem rot of poinsettia. Plant Dis 79:309–313
Cartwright DK, Chilton WS, Benson DM (1995) Pyrrolnitrin and phenazine production by Pseudomonas cepacia, strain 5.5B, a biocontrol agent of Rhizoctonia solani. Appl Microbiol Biotechnol 43:211–216
Cho JY, Kim HK, Ma SJ, Moon JH, Park KH (2000) Isolation and identification of azelaic acid and 3,4-dihydroxybenzoic acid from buckwheat hull as antimicrobial substances. Food Sci Biotechnol 9:313–316
Dagley S, Chapman PJ, Gibson DT (1965) The metabolism of β-phenylpropionic acid by an Achromobacter. Biochem J 97:643–650
Di Cello F, Bevivino A, Chiarini L, Fani R, Paffetti D, Tabacchioni S, et al. (1997) Biodiversity of a Burkholderia cepacia population isolated from the maize rhizoshere at different plant growth stages. Appl Environ. Microbiol 63:4485–4493
El-Banna N, Winkelmann G (1998) Pyrrolnitrin from Burkholdera cepacia: Antibiotic activity against fungi and novel activities against streptomycetes. J Appl Microbiol 85:69–78
Fravel DR (1988) Role of antibiosis in the biocontrol of plant disease. Annu Rev Phytopathol 26:75–91
Hirota A, Horikawa T, Fujiwara A (1992) Isolation of phenylacetic acid from a phytopathogenic fungus, Glomerella cingulata. Biosci Biotech Biochem 57:492
Holmes A, Govan J, Goldstein R (1998) Agricultural use of Burkholderia (Pseudomonas) cepacia: A threat to human health? Emerg Infect Dis 4:221–227
Hwang J, Chiton WS, Benson DM (2002) Pyrrolnitrin production by Burkholderia cepacia and biocontrol of Rhizoctonia stem rot of poinsettia. Biol Control 25:56–63
Jae GK, Sun TK, Kyu YK (1999) Production of the antifungal compound phenylacetic acid by antagonistic bacterium Pseudomonas sp. Agric. Chem Biotechnol 42:197–201
Janisiewicz W, Roitman J (1988) Biological control of blue mold and gray mold on apple and pear with Pseudomonas cepacia. Phytopathology 78:1697–1700
Jiao Y, Yoshihara T, Ishikuri S, Uchino H, Ichihara A (1996) Structural identification of cepaciamide A, a novel fungi-toxic compound from Pseudonomas cepacia D-202. Tetrahedron Lett 37:1039–1042
Kawazu K, Zhang H, Kanzaki (1996) Accumulation of benzoic acid in suspension of cultured cells of Pinus thungergii Oarl in response to phenylacetic acid administration. Biosci Biotechnol Biochem 60:1410–1412
King EB, Parke JL (1993) Biocontrol of Aphanomyces root rot and Pythium damping-off by Pseudomonas cepacia AMMD on four pea cultivars. Plant Dis 77:1185–1188
Kuk JH, Ma SJ, Moon JH, Kim KY, Choi SH, Park KH (2002) Antibacterial and antifungal activities of a naphthoquinone derivative isolated from the fruits of Catalpa ovata G. DON. J. Microbiol. Biotechnol. 12:858–863
Lee C, Kim S, Hyun B, Suh J, Yon C, Kim C, et al. (1994) Cepacidine A, a novel antifungal antibiotic produced by Pseudonomas cepacia. I. Taxonomy, production, isolation and biological activity. J Antibiot 47:1402–1418
Lee SJ, Cho JY, Cho JI, Moon JH, Park KD, Lee YJ, et al. (2004) Isolation and characterization of antimicrobial substance macrolactin A produced from Bacillus amyloliquefaciens CHO104 isolated from soil. J Microbiol Biotechnol 14:525–531
Leisinger T, Margraff R (1979) Secondary metabolites of fluorescent Pseudomonas. Microbiol Rev 43:422–442
Meyers E, Bisacchi GS, Dean L (1987) Xylocandin: A new complex of antifungal peptide. I. Taxonomy, isolation and biological activity. J Antibiot 21:1515–1519
Meyer JM, Hohnadel D, Halle F (1989) Cepabactin from Pseudomonas cepacia, a new type of siderophore. J Gen Microbiol 135:1479–1487
Parker WL, Rathnum ML, Seiner V, Trejo WH, Principe PA, Sykes RB (1984) Cepacin A and cepacin B, two antibiotics produced by Pseudonomas cepacia. J Antibiot 37:431–440
Reyes Chilpa R, Quiroz Vázquez RI, Jiménez Estrada M, Navarro-Ocaña A, Cassani Hernández J (1997) Antifungal activity of selected plant secondary metabolites against Coriolus versicolor. J Trop For Prods 3:110–113
Roitman JN, Mahoney NE, Janisiewicz WJ (1990) Production and composition of phenylpyrrole metabolites produced by Pseudomonas cepacia. Appl Microbiol Biotechnol 34:381–386
Weller DM (1988) Biological control of soilborne plant pathogens in the rhizoshere with bacteria. Annu Rev Phytopathol 26:379–407
Wightman F, Lighty DL (1982) Identification of phenylacetic acid as a natural auxin in the shoots of higher plants. Physiol Plant 55:17–24
Yim CK, Moon JH, Park KH (1999) Isolation of 3,4-dihydroxybenzoic acid, which exhibits antimicrobial activity, from fruits of Gardenia jasminoides. Korean J Food Sci Technol 31:1386–1392
Yoon K, Cho JY, Kuk JH, Moon JH, Cho JI, Kim YC, et al. (2004) Identification and antimicrobial activity of phenylacetic acid produced by Bacillus licheniformis isolated from fermented soybean, Chungkook-Jang. Curr Microbiol 48:312–317
Zaki K, Misaghi IJ, Heydari A (1998) Control of cotton seedling damping-off in the field by Burkholderia(Pseudomonas) cepacia. Plant Dis 82:291–293
Acknowledgments
This study was supported by a grant from the BioGreen 21 program, Rural Development Administration, and the Environmental-Friendly Agriculture Research Program (2005), Ministry of Agriculture, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mao, S., Lee, SJ., Hwangbo, H. et al. Isolation and Characterization of Antifungal Substances from Burkholderia sp. Culture Broth. Curr Microbiol 53, 358–364 (2006). https://doi.org/10.1007/s00284-005-0333-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-005-0333-2