Abstract
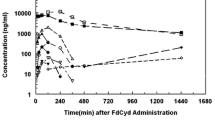
Purpose: Cladribine (2-chlorodeoxyadenosine, 2-CDA) is effective in the treatment of various lymphoproliferative disorders. In the standard protocol the compound is administered by continuous intravenous (i.v.) infusion. In order to allow outpatient therapy alternative modes of administration such as subcutaneous (s.c.) injection would be desirable. The aim of the present study was to compare the pharmacokinetics of 2-CDA after i.v. and s.c. administration. Patients and methods: Nine patients received 0.1 mg/kg 2-CDA per 24 h on one occasion by continuous i.v. infusion and on another occasion as a bolus subcutaneously. The concentrations of 2-CDA in the plasma and urine were determined by HPLC. Results: During i.v. infusion the concentration of 2-CDA in the plasma reached a plateau after 4–8 h, whereas with s.c. administration almost ten times higher peak concentrations were reached within 20 to 60 min. A two-compartment model was fitted to the data points whereby the goodness-of-fit statistics showed R 2 values of >0.98. The calculated rate of elimination, kelim, averaged 0.336 h−1 with s.c. and 0.397 h−1 with i.v. administration. The estimated volumes of distribution were 1.67 and 1.58 l/kg. The areas under the concentration time curves (608 ± 65 pmol · h/ml after s.c. administration vs 571 ± 50 pmol · h/ml during i.v. infusion) and the urinary excretion of 2-CDA in 24 h (4.75 ± 0.95 vs 3.55 ± 0.53 μmol/24 h) were similar in both groups, indicating identical bioavailability. Conclusions: Although the pharmacokinetic profile of 2-CDA administered s.c. differs substantially from the profile of a continuous i.v. infusion the areas under the plasma concentration time curves, the urinary excretion of unchanged drug and the estimated pharmacokinetic variables were similar with both modes of administration, indicating that the different time-courses of the plasma concentration did not influence the fraction metabolized or eliminated.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 3 November 1999 / Accepted: 3 March 2000
Rights and permissions
About this article
Cite this article
Sonderegger, T., Betticher, D., Cerny, T. et al. Pharmacokinetics of 2-chloro-2′-deoxyadenosine administered subcutaneously or by continuous intravenous infusion. Cancer Chemother Pharmacol 46, 40–42 (2000). https://doi.org/10.1007/s002800000129
Issue Date:
DOI: https://doi.org/10.1007/s002800000129