Abstract
We and others have shown that cytogenetically normal (CN)-AML patients with biallelic CEBPA gene mutations (biCEBPA) represent a molecularly distinct group with a favorable prognosis. Patients carrying a monoallelic CEBPA mutation (moCEBPA), however, show no different outcome compared to patients with wildtype CEBPA, and these mutations are frequently associated with mutated NPM1 or FLT3-ITD. So far, no molecular or clinical hallmark has been identified to prognostically distinguish moCEBPA patients from patients with wildtype CEBPA. Therefore, we used the data of 663 CN-AML patients treated within the AMLCG 1999 trial to explore the prognostic value of moCEBPA in the context of concomitant clinical and molecular markers (mutated NPM1, FLT3-ITD). Multiple Cox regression in 515 patients adjusting for all available potential confounders revealed that the NPM1 mutation modified the prognostic value of moCEBPA with respect to overall survival (OS, p = 0.017) and event-free survival (EFS, p = 0.011). MoCEBPA was beneficial in NPM1 mutated patients: adjusted OS–hazard ratio (HR) 0.09, 95% confidence interval (CI) 0.01–0.63, p = 0.016; EFS–HR (95% CI) 0.16 (0.04–0.65), p = 0.010. In contrast, moCEBPA had no prognostic impact in patients with wildtype NPM1: OS–HR (95% CI) 1.08 (0.59–1.97), p = 0.804; EFS–HR (95% CI) 1.12 (0.64–1.96), p = 0.682. We found no prognostic effect modification for moCEBPA by FLT3-ITD. The presence of a moCEBPA mutation was shown to be associated with prolonged survival in NPM1 mutated CN-AML patients. Confirmation of these results in larger studies will clarify whether an additional moCEBPA mutation influences the risk stratification of patients with an NPM1 mutated/FLT3-ITD positive genotype.


Similar content being viewed by others
References
Tenen DG, Hromas R, Licht JD, Zhang DE (1997) Transcription factors, normal myeloid development, and leukemia. Blood 90(2):489–519
Zhang DE, Zhang P, Wang ND, Hetherington CJ, Darlington GJ, Tenen DG (1997) Absence of granulocyte colony-stimulating factor signaling and neutrophil development in CCAAT enhancer binding protein alpha-deficient mice. Proc Natl Acad Sci U S A 94(2):569–574
Zhang P, Iwasaki-Arai J, Iwasaki H, Fenyus ML, Dayaram T, Owens BM, Shigematsu H, Levantini E, Huettner CS, Lekstrom-Himes JA, Akashi K, Tenen DG (2004) Enhancement of hematopoietic stem cell repopulating capacity and self-renewal in the absence of the transcription factor C/EBP alpha. Immunity 21(6):853–863
Bienz M, Ludwig M, Leibundgut EO, Mueller BU, Ratschiller D, Solenthaler M, Fey MF, Pabst T (2005) Risk assessment in patients with acute myeloid leukemia and a normal karyotype. Clin Cancer Res 11(4):1416–1424
Dufour A, Schneider F, Metzeler KH, Hoster E, Schneider S, Zellmeier E, Benthaus T, Sauerland MC, Berdel WE, Buchner T, Wormann B, Braess J, Hiddemann W, Bohlander SK, Spiekermann K (2009) Acute myeloid leukemia with biallelic CEBPA gene mutations and normal karyotype represents a distinct genetic entity associated with a favorable clinical outcome. J Clin Oncol 28(4):570–577
Frohling S, Schlenk RF, Stolze I, Bihlmayr J, Benner A, Kreitmeier S, Tobis K, Dohner H, Dohner K (2004) CEBPA mutations in younger adults with acute myeloid leukemia and normal cytogenetics: prognostic relevance and analysis of cooperating mutations. J Clin Oncol 22(4):624–633
Pabst T, Mueller BU, Zhang P, Radomska HS, Narravula S, Schnittger S, Behre G, Hiddemann W, Tenen DG (2001) Dominant-negative mutations of CEBPA, encoding CCAAT/enhancer binding protein-alpha (C/EBPalpha), in acute myeloid leukemia. Nat Genet 27(3):263–270
Gombart AF, Hofmann WK, Kawano S, Takeuchi S, Krug U, Kwok SH, Larsen RJ, Asou H, Miller CW, Hoelzer D, Koeffler HP (2002) Mutations in the gene encoding the transcription factor CCAAT/enhancer binding protein alpha in myelodysplastic syndromes and acute myeloid leukemias. Blood 99(4):1332–1340
Asou H, Gombart AF, Takeuchi S, Tanaka H, Tanioka M, Matsui H, Kimura A, Inaba T, Koeffler HP (2003) Establishment of the acute myeloid leukemia cell line Kasumi-6 from a patient with a dominant-negative mutation in the DNA-binding region of the C/EBPalpha gene. Genes Chromosomes Cancer 36(2):167–174
Green CL, Koo KK, Hills RK, Burnett AK, Linch DC, Gale RE (2010) Prognostic significance of CEBPA mutations in a large cohort of younger adult patients with acute myeloid leukemia: impact of double CEBPA mutations and the interaction with FLT3 and NPM1 mutations. J Clin Oncol 28(16):2739–2747
Pabst T, Eyholzer M, Fos J, Mueller BU (2009) Heterogeneity within AML with CEBPA mutations; only CEBPA double mutations, but not single CEBPA mutations are associated with favourable prognosis. Br J Cancer 100(8):1343–1346
Taskesen E, Bullinger L, Corbacioglu A, Sanders M, Erpelinck CA, Wouters BJ, van der Poel-vandeLuytgaarde S, Damm F, Krauter J, Ganser A, Schlenk RF, Lowenberg B, Delwel R, Dohner H, Valk PJ, Dohner K (2011) Prognostic impact, concurrent genetic mutations and gene expression features of AML with CEBPA mutations in a cohort of 1182 cytogenetically normal AML: further evidence for CEBPA double mutant AML as a distinctive disease entity. Blood 117(8):2469–2475
Wouters BJ, Lowenberg B, Erpelinck-Verschueren CA, van Putten WL, Valk PJ, Delwel R (2009) Double CEBPA mutations, but not single CEBPA mutations, define a subgroup of acute myeloid leukemia with a distinctive gene expression profile that is uniquely associated with a favorable outcome. Blood 113(13):3088–3091
van Waalwijk B, van Doorn-Khosrovani S, Erpelinck C, Meijer J, van Oosterhoud S, van Putten WL, Valk PJ, Berna Beverloo H, Tenen DG, Lowenberg B, Delwel R (2003) Biallelic mutations in the CEBPA gene and low CEBPA expression levels as prognostic markers in intermediate-risk AML. Hematol J 4(1):31–40
Lin LI, Chen CY, Lin DT, Tsay W, Tang JL, Yeh YC, Shen HL, Su FH, Yao M, Huang SY, Tien HF (2005) Characterization of CEBPA mutations in acute myeloid leukemia: most patients with CEBPA mutations have biallelic mutations and show a distinct immunophenotype of the leukemic cells. Clin Cancer Res 11(4):1372–1379
Bereshchenko O, Mancini E, Moore S, Bilbao D, Mansson R, Luc S, Grover A, Jacobsen SE, Bryder D, Nerlov C (2009) Hematopoietic stem cell expansion precedes the generation of committed myeloid leukemia-initiating cells in C/EBPalpha mutant AML. Cancer Cell 16(5):390–400
Kato N, Kitaura J, Doki N, Komeno Y, Watanabe-Okochi N, Togami K, Nakahara F, Oki T, Enomoto Y, Fukuchi Y, Nakajima H, Harada Y, Harada H, Kitamura T (2011) Two types of C/EBP{alpha} mutations play distinct but collaborative roles in leukemogenesis: lessons from clinical data and BMT models. Blood 117(1):221–233
Kirstetter P, Schuster MB, Bereshchenko O, Moore S, Dvinge H, Kurz E, Theilgaard-Monch K, Mansson R, Pedersen TA, Pabst T, Schrock E, Porse BT, Jacobsen SE, Bertone P, Tenen DG, Nerlov C (2008) Modeling of C/EBPalpha mutant acute myeloid leukemia reveals a common expression signature of committed myeloid leukemia-initiating cells. Cancer Cell 13(4):299–310
Somervaille TC, Cleary ML (2009) Preview. Mutant CEBPA: priming stem cells for myeloid leukemogenesis. Cell Stem Cell 5(5):453–454
Dohner K, Schlenk RF, Habdank M, Scholl C, Rucker FG, Corbacioglu A, Bullinger L, Frohling S, Dohner H (2005) Mutant nucleophosmin (NPM1) predicts favorable prognosis in younger adults with acute myeloid leukemia and normal cytogenetics: interaction with other gene mutations. Blood 106(12):3740–3746
Falini B, Mecucci C, Tiacci E, Alcalay M, Rosati R, Pasqualucci L, La Starza R, Diverio D, Colombo E, Santucci A, Bigerna B, Pacini R, Pucciarini A, Liso A, Vignetti M, Fazi P, Meani N, Pettirossi V, Saglio G, Mandelli F, Lo-Coco F, Pelicci PG, Martelli MF (2005) Cytoplasmic nucleophosmin in acute myelogenous leukemia with a normal karyotype. N Engl J Med 352(3):254–266
Schnittger S, Schoch C, Kern W, Mecucci C, Tschulik C, Martelli MF, Haferlach T, Hiddemann W, Falini B (2005) Nucleophosmin gene mutations are predictors of favorable prognosis in acute myelogenous leukemia with a normal karyotype. Blood 106(12):3733–3739
Schlenk RF, Dohner K, Krauter J, Frohling S, Corbacioglu A, Bullinger L, Habdank M, Spath D, Morgan M, Benner A, Schlegelberger B, Heil G, Ganser A, Dohner H (2008) Mutations and treatment outcome in cytogenetically normal acute myeloid leukemia. N Engl J Med 358(18):1909–1918
Verhaak RG, Goudswaard CS, van Putten W, Bijl MA, Sanders MA, Hugens W, Uitterlinden AG, Erpelinck CA, Delwel R, Lowenberg B, Valk PJ (2005) Mutations in nucleophosmin (NPM1) in acute myeloid leukemia (AML): association with other gene abnormalities and previously established gene expression signatures and their favorable prognostic significance. Blood 106(12):3747–3754
Ley TJ, Ding L, Walter MJ, McLellan MD, Lamprecht T, Larson DE, Kandoth C, Payton JE, Baty J, Welch J, Harris CC, Lichti CF, Townsend RR, Fulton RS, Dooling DJ, Koboldt DC, Schmidt H, Zhang Q, Osborne JR, Lin L, O'Laughlin M, McMichael JF, Delehaunty KD, McGrath SD, Fulton LA, Magrini VJ, Vickery TL, Hundal J, Cook LL, Conyers JJ, Swift GW, Reed JP, Alldredge PA, Wylie T, Walker J, Kalicki J, Watson MA, Heath S, Shannon WD, Varghese N, Nagarajan R, Westervelt P, Tomasson MH, Link DC, Graubert TA, DiPersio JF, Mardis ER, Wilson RK (2010) DNMT3A mutations in acute myeloid leukemia. N Engl J Med 363 (25):2424-2433
Mardis ER, Ding L, Dooling DJ, Larson DE, McLellan MD, Chen K, Koboldt DC, Fulton RS, Delehaunty KD, McGrath SD, Fulton LA, Locke DP, Magrini VJ, Abbott RM, Vickery TL, Reed JS, Robinson JS, Wylie T, Smith SM, Carmichael L, Eldred JM, Harris CC, Walker J, Peck JB, Du F, Dukes AF, Sanderson GE, Brummett AM, Clark E, McMichael JF, Meyer RJ, Schindler JK, Pohl CS, Wallis JW, Shi X, Lin L, Schmidt H, Tang Y, Haipek C, Wiechert ME, Ivy JV, Kalicki J, Elliott G, Ries RE, Payton JE, Westervelt P, Tomasson MH, Watson MA, Baty J, Heath S, Shannon WD, Nagarajan R, Link DC, Walter MJ, Graubert TA, DiPersio JF, Wilson RK, Ley TJ (2009) Recurring mutations found by sequencing an acute myeloid leukemia genome. N Engl J Med 361(11):1058–1066
Yan XJ, Xu J, Gu ZH, Pan CM, Lu G, Shen Y, Shi JY, Zhu YM, Tang L, Zhang XW, Liang WX, Mi JQ, Song HD, Li KQ, Chen Z, Chen SJ (2011) Exome sequencing identifies somatic mutations of DNA methyltransferase gene DNMT3A in acute monocytic leukemia. Nat Genet 43 (4):309-315
Abbas S, Lugthart S, Kavelaars FG, Schelen A, Koenders JE, Zeilemaker A, van Putten WJ, Rijneveld AW, Lowenberg B, Valk PJ (2010) Acquired mutations in the genes encoding IDH1 and IDH2 both are recurrent aberrations in acute myeloid leukemia: prevalence and prognostic value. Blood 116 (12):2122-2126
Green CL, Evans CM, Hills RK, Burnett AK, Linch DC, Gale RE (2010) The prognostic significance of IDH1 mutations in younger adult patients with acute myeloid leukemia is dependent on FLT3/ITD status. Blood 116 (15):2779-2782
Marcucci G, Maharry K, Wu YZ, Radmacher MD, Mrozek K, Margeson D, Holland KB, Whitman SP, Becker H, Schwind S, Metzeler KH, Powell BL, Carter TH, Kolitz JE, Wetzler M, Carroll AJ, Baer MR, Caligiuri MA, Larson RA, Bloomfield CD (2010) IDH1 and IDH2 gene mutations identify novel molecular subsets within de novo cytogenetically normal acute myeloid leukemia: a Cancer and Leukemia Group B study. J Clin Oncol 28 (14):2348-2355
Paschka P, Schlenk RF, Gaidzik VI, Habdank M, Kronke J, Bullinger L, Spath D, Kayser S, Zucknick M, Gotze K, Horst HA, Germing U, Dohner H, Dohner K (2010) IDH1 and IDH2 mutations are frequent genetic alterations in acute myeloid leukemia and confer adverse prognosis in cytogenetically normal acute myeloid leukemia with NPM1 mutation without FLT3 internal tandem duplication. J Clin Oncol 28 (22):3636-3643
Thol F, Damm F, Ludeking A, Winschel C, Wagner K, Morgan M, Yun H, Gohring G, Schlegelberger B, Hoelzer D, Lubbert M, Kanz L, Fiedler W, Kirchner H, Heil G, Krauter J, Ganser A, Heuser M (2010) Incidence and prognostic influence of DNMT3A mutations in acute myeloid leukemia. J Clin Oncol 29 (21):2889-2896
Verhaak RG, Wouters BJ, Erpelinck CA, Abbas S, Beverloo HB, Lugthart S, Lowenberg B, Delwel R, Valk PJ (2009) Prediction of molecular subtypes in acute myeloid leukemia based on gene expression profiling. Haematologica 94(1):131–134
Buchner T, Berdel WE, Schoch C, Haferlach T, Serve HL, Kienast J, Schnittger S, Kern W, Tchinda J, Reichle A, Lengfelder E, Staib P, Ludwig WD, Aul C, Eimermacher H, Balleisen L, Sauerland MC, Heinecke A, Wormann B, Hiddemann W (2006) Double induction containing either two courses or one course of high-dose cytarabine plus mitoxantrone and postremission therapy by either autologous stem-cell transplantation or by prolonged maintenance for acute myeloid leukemia. J Clin Oncol 24(16):2480–2489
Benthaus T, Schneider F, Mellert G, Zellmeier E, Schneider S, Kakadia PM, Hiddemann W, Bohlander SK, Feuring-Buske M, Braess J, Spiekermann K, Dufour A (2008) Rapid and sensitive screening for CEBPA mutations in acute myeloid leukaemia. Br J Haematol 143(2):230–239
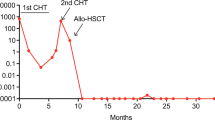
Papadaki C, Dufour A, Seibl M, Schneider S, Bohlander SK, Zellmeier E, Mellert G, Hiddemann W, Spiekermann K (2009) Monitoring minimal residual disease in acute myeloid leukaemia with NPM1 mutations by quantitative PCR: clonal evolution is a limiting factor. Br J Haematol 144(4):517–523
Schnittger S, Schoch C, Dugas M, Kern W, Staib P, Wuchter C, Loffler H, Sauerland CM, Serve H, Buchner T, Haferlach T, Hiddemann W (2002) Analysis of FLT3 length mutations in 1003 patients with acute myeloid leukemia: correlation to cytogenetics, FAB subtype, and prognosis in the AMLCG study and usefulness as a marker for the detection of minimal residual disease. Blood 100(1):59–66
Bacher U, Haferlach C, Kern W, Haferlach T, Schnittger S (2008) Prognostic relevance of FLT3-TKD mutations in AML: the combination matters–an analysis of 3082 patients. Blood 111(5):2527–2537
Schnittger S, Kinkelin U, Schoch C, Heinecke A, Haase D, Haferlach T, Buchner T, Wormann B, Hiddemann W, Griesinger F (2000) Screening for MLL tandem duplication in 387 unselected patients with AML identify a prognostically unfavorable subset of AML. Leukemia 14(5):796–804
Felsberg J, Wolter M, Seul H, Friedensdorf B, Goppert M, Sabel MC, Reifenberger G (2010) Rapid and sensitive assessment of the IDH1 and IDH2 mutation status in cerebral gliomas based on DNA pyrosequencing. Acta Neuropathol 119 (4):501-507
Markova J, Michkova P, Burckova K, Brezinova J, Michalova K, Dohnalova A, Maaloufova JS, Soukup P, Vitek A, Cetkovsky P, Schwarz J (2010) Prognostic impact of DNMT3A mutations in patients with intermediate cytogenetic risk profile acute myeloid leukemia. Eur J Haematol 88 (2):128-135
Thiede C, Koch S, Creutzig E, Steudel C, Illmer T, Schaich M, Ehninger G (2006) Prevalence and prognostic impact of NPM1 mutations in 1485 adult patients with acute myeloid leukemia (AML). Blood 107(10):4011–4020
Dohner H, Estey EH, Amadori S, Appelbaum FR, Buchner T, Burnett AK, Dombret H, Fenaux P, Grimwade D, Larson RA, Lo-Coco F, Naoe T, Niederwieser D, Ossenkoppele GJ, Sanz MA, Sierra J, Tallman MS, Lowenberg B, Bloomfield CD (2010) Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood 115 (3):453-474
Gale RE, Green C, Allen C, Mead AJ, Burnett AK, Hills RK, Linch DC (2008) The impact of FLT3 internal tandem duplication mutant level, number, size, and interaction with NPM1 mutations in a large cohort of young adult patients with acute myeloid leukemia. Blood 111(5):2776–2784
Kottaridis PD, Gale RE, Frew ME, Harrison G, Langabeer SE, Belton AA, Walker H, Wheatley K, Bowen DT, Burnett AK, Goldstone AH, Linch DC (2001) The presence of a FLT3 internal tandem duplication in patients with acute myeloid leukemia (AML) adds important prognostic information to cytogenetic risk group and response to the first cycle of chemotherapy: analysis of 854 patients from the United Kingdom Medical Research Council AML 10 and 12 trials. Blood 98(6):1752–1759
Shih LY, Huang CF, Wu JH, Lin TL, Dunn P, Wang PN, Kuo MC, Lai CL, Hsu HC (2002) Internal tandem duplication of FLT3 in relapsed acute myeloid leukemia: a comparative analysis of bone marrow samples from 108 adult patients at diagnosis and relapse. Blood 100(7):2387–2392
Andersson A, Johansson B, Lassen C, Mitelman F, Billstrom R, Fioretos T (2004) Clinical impact of internal tandem duplications and activating point mutations in FLT3 in acute myeloid leukemia in elderly patients. Eur J Haematol 72(5):307–313
Acknowledgements
The authors would like to thank Evelyn Zellmeier and Gudrun Mellert for excellent technical assistance.
Conflict of interest
The authors indicate no potential conflict of interest.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Annika Dufour and Friederike Schneider contributed equally to this manuscript.
Appendix
Appendix
Selection
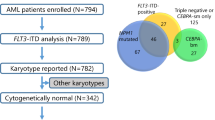
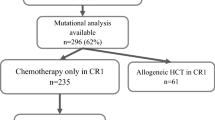
Patients with a complete molecular status of CEBPA, NPM1 and FLT3-ITD were selected out of a total of 802 patients. The selected patients (n = 663) had a significantly higher percentage of bone marrow blasts and leucocytes compared to non-selected patients (n = 139). Other clinical parameters and the frequency of molecular markers were similar in selected versus unselected patients. OS, EFS and RFS did not significantly differ between both tested and untested groups (p = 0.342, p = 0.219, p = 0.600, respectively) (data not shown).
List of participating centers within the AML CG 1999 study
Study coordinators: T. Büchner, W. Hiddemann, W.E. Berdel, B. Wörmann
Statisticians: A. Heinecke, M.C. Sauerland, F. Schneider, E. Hoster, M. Unterhalt
Cytogenetic and molecular genetic review: C. Haferlach, S. Schnittger, P.M. Kakadia, SK. Bohlander, A. Dufour, K. Spiekermann
Cytology review: T. Haferlach, J. Braess, K Spiekermann
Contributing centers and investigators:
University Hospital, Aachen (T.H. Ittel)
Municipal Hospital Augsburg (C. Schmid, D. Oruzio)
Municipal Hospital, Bad Saarow (H. Fu)
Vinzenz-Pallotti-Hospital, Bergisch-Gladbach (S. Korsten, D. Hennesser)
Municipal Hospital Neukölln, Berlin (A. Mayr, A. Grüneisen)
University Hospital Charité Mitte, Berlin (K. Possinger, J. Blau)
University Hospital Charité, Berlin (R. Arnold, B. Dörken, G. Maschmeyer)
St Hedwig Hospital, Berlin (C. Boewer, M. Derwahl, H.J. Englisch)
University Hospital Benjamin Franklin, Berlin (M. Notter, E. Thiel)
University Hospital Robert Rössle, Berlin (W.D. Ludwig, D. Schöndube)
Vivantes Klinikum, Berlin (E. Späth-Schwalbe, S. Hesse)
Vivantes Klinikum Berlin Neukölln (A. Grüneisen)
Ev. Hospital Spandau, Berlin (J. Potenberg)
Municipal Hospital, Bielefeld (A.J. Weh, A. Zumsprekel)
Knappschaft Hospital, Bochum (C. Teschendorf, M. Stechstor)
Knappschaft Hospital, Bottrop (G. Trenn)
Municipal Medical Center, Braunschweig (B. Wörmann)
Municipal Hospital, St Jürgenstrasse, Bremen (B. Hertenstein, H. Thomssen, A. Peyn)
University Hospital, Cologne (M. Hallek, P. Staib, KA Kreuzer)
Municipal Hospital, Dortmund (M. Heike, A. Niederste-Hollenberg)
St Johannes Hospital, Dortmund (H. Pielken, H. Hindahl)
University Hospital, Düsseldorf (A. Wehmeyer, A. Heyll)
St Johannes Hospital, Duisburg (C. Aul, C. Giagounidis)
Johanniter Hospital Rheinhausen, Duisburg (W. Lange, S.E. Kuhlemann)
Municipal Hospital, Düren (M. Flasshove, J. Karow)
St Antonius Hospital, Eschweiler (R. Fuchs, F. Schlegel)
University Hospital, Frankfurt/Oder (M. Kiehl)
St Joseph-Hospital, Gelsenkirchen (G. Meckenstock, G. Giagounidis)
University Hospital, Göttingen (D. Haase, L. Trümper, F. Griesinger)
Municipal Hospital, Gütersloh (C. Gropp, R. Depenbusch)
Municipal Hospital, Hagen (H. Eimermacher, Lindemann)
Municipal Hospital Martha-Maria, Halle (W. Schütte, U. Haak)
General Hospital Altona, Hamburg (D. Braumann)
Protestant Hospital, Hamm (L. Balleisen)
District Hospital, Herford (J.G. Lange, U. Schmitz-Hubner)
Municipal Hospital, Idar-Oberstein (A. Fauser)
Municipal Hospital, Kassel (M. Wolf, B. Ritter)
Municipal Hospital, Kaiserslautern (H. Link)
University Hospital I, Kiel (U.R. Fölsch)
University Hospital II, Kiel (M. Kneba)
Municipal Hospital, Köln (A. Dormann)
Municipal Hospital, Krefeld (Th. Frieling, M. Planker)
Hospital Lippe-Lemgo, Lemgo (F. Hartmann, H. Middeke, C.Gründgens,C.Constantin)
Trinity Hospital, Lippstadt (K.-A. Jost)
University Hospital, Lübeck (Th. Wagner)
Municipal Hospital South Lübeck, Lübeck (S. Fetscher, J. Schmielau)
Municipal Hospital, Ludwigshafen (M. Uppenkamp, M. Hoffmann)
University Hospital, Mannheim (R. Hehlmann, E. Lengfelder)
St Walburga Hospital, Meschede (M. Schwonzen, H. Spangenberg)
Maria-Hilf-Hospital, Mönchengladbach (D. Graeven, D. Kohl, T. Heuer)
University Hospital Muenster (WE. Berdel)
University Hospital Innenstadt, Munich (B. Emmerich, R. Dengler, B. Schlag)
University Hospital Grosshadern, Munich (W. Hiddemann, J. Braess, K. Spiekermann, S.K. Bohlander, C. Buske)
Municipal Hospital Neuperlach, Munich (K. Nibler, D. Fleckenstein)
Municipal Hospital Harlaching, Munich (M. Hentrich, X. Schiel)
Rights and permissions
About this article
Cite this article
Dufour, A., Schneider, F., Hoster, E. et al. Monoallelic CEBPA mutations in normal karyotype acute myeloid leukemia: independent favorable prognostic factor within NPM1 mutated patients. Ann Hematol 91, 1051–1063 (2012). https://doi.org/10.1007/s00277-012-1423-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-012-1423-4