Abstract
Aim
To report the multicenter retrospective experience on combination of balloon-occluded MWA(b-MWA) followed by balloon-occluded TACE(b-TACE) in patients with liver malignancies > 3 cm, focusing on appearance and volume of necrotic area, safety profile and oncological results.
Materials and Methods
Twenty-three patients with liver primary malignancies (hepatocellular carcinoma,HCC = 18; intrahepatic cholangiocarcinoma,iCC = 2) and metastasis (colorectal cancer metastasis = 1;sarcoma metastasis = 1;breast metastasis = 1) were treated. Maximum mean diameter of lesions was 4.4 cm (± 1 cm). Treatments were performed using a single-step approach:b-MWA was performed after balloon-microcatheter inflation, followed by b-TACE (with epirubicin or irinotecan). Necrotic area shape and discrepancy with the expected volume of necrosis suggested by vendor’s ablation chart were assessed at post-procedural CT. Complications were categorized according to CIRSE classification. Oncological results at 1 and 3–6 months were evaluated using m-RECIST(HCC) and RECISTv1.1(metastasis/iCC).
Results
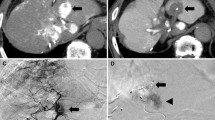
Mean volume of necrotic area was 75 cm3 (± 36). Discrepancy with vendor chart consisted in a medium percentage of volumetric incrementation of necrotic area of 103.2% (± 99.8). Non-spherical shape was observed in 22/23 patients (95.7%). No complications occurred; Post-embolization syndrome occurred in 12/23patients. Complete response and partial response were, respectively, 91, 3% (21/23) and 8.7% (2/23) at 1 month, 85.7% (18/21) and 9.5% (2/21) at 3–6 months. Progression of disease was 4.7% (1/21) at 3–6 months for extra-hepatic progression. Among partial responders, average percentage of tumor volume debulking was 78.8% (± 9.8%).
Conclusion
b-MWA followed by b-TACE in a single-step procedure led to larger necrotic areas than the proposed by vendors ablation chart, non-spherical in shape and corresponded to the vascular segment occluded during ablation. This permitted to safely achieve promising oncological results in patients with > 3 cm tumors.



Similar content being viewed by others
References
Crocetti L, de Baére T, Pereira PL, Tarantino FP. CIRSE standards of practice on thermal ablation of liver tumours. Cardiovasc Intervent Radiol. 2020;43(7):951–62. https://doi.org/10.1007/s00270-020-02471-z.
Galle PR, Forner A, Llovet JM, et al. Clinical practice guidelines of hepatology EASL clinical practice guidelines: management of hepatocellular carcinoma q. J Hepatol. 2018;69(1):182–236. https://doi.org/10.1016/j.jhep.2018.03.019.
Phelip JM, Tougeron D, Léonard D, et al. Metastatic colorectal cancer (mCRC): French intergroup clinical practice guidelines for diagnosis, treatments and follow-up (SNFGE, FFCD, GERCOR, UNICANCER, SFCD, SFED, SFRO, SFR). Dig Liver Dis. 2019;51(10):1357–63. https://doi.org/10.1016/j.dld.2019.05.035.
Han K, Ko HK, Kim KW, Won HJ, Shin YM, Kim PN. Radiofrequency ablation in the treatment of unresectable intrahepatic cholangiocarcinoma: systematic review and meta-analysis. J Vasc Interv Radiol. 2015;26(7):943–8. https://doi.org/10.1016/j.jvir.2015.02.024.
Leung U, Kuk D, D’Angelica MI, et al. Long-term outcomes following microwave ablation for liver malignancies. Br J Surg. 2015;102(1):85–91. https://doi.org/10.1002/bjs.9649.
Doyle A, Gorgen A, Muaddi H, et al. Outcomes of radiofrequency ablation as first-line therapy for hepatocellular carcinoma less than 3 cm in potentially transplantable patients. J Hepatol. 2019;70(5):866–73. https://doi.org/10.1016/j.jhep.2018.12.027.
Kapoor H, Nisiewicz MJ, Jayavarapu R, Gedaly R, Raissi D. Early outcomes with single-antenna high-powered percutaneous microwave ablation for primary and secondary hepatic malignancies: safety, effectiveness, and predictors of ablative failure. J Clin Imaging Sci. 2020;10:10. https://doi.org/10.25259/JCIS_173_2019.
Liu C, Li T, He JT, Shao H. Tace combined with microwave ablation therapy vs. Tace alone for treatment of early-and intermediate-stage hepatocellular carcinomas larger than 5 cm: a meta-analysis. Diagn Interv Radiol. 2020;26(6):575–83. https://doi.org/10.5152/dir.2020.19615.
Lucatelli P, Ginnani Corradini L, De Rubeis G, et al. Balloon-occluded transcatheter arterial chemoembolization (b-TACE) for hepatocellular carcinoma performed with polyethylene-glycol epirubicin-loaded drug-eluting embolics: safety and preliminary results. Cardiovasc Intervent Radiol. 2019. https://doi.org/10.1007/s00270-019-02192-y.
Lucatelli P, De Rubeis G, Rocco B, et al. Balloon occluded TACE (B-TACE) vs DEM-TACE for HCC: a single center retrospective case control study. BMC Gastroenterol. 2021;21(1). https://doi.org/10.1186/s12876-021-01631-w.
Golfieri R, Bezzi M, Verset G, et al. Retrospective European multicentric evaluation of selective transarterial chemoembolisation with and without balloon-occlusion in patients with hepatocellular carcinoma: a propensity score matched analysis. Cardiovasc Intervent Radiol. 2021;44(7):1048–59. https://doi.org/10.1007/s00270-021-02805-5.
Iezzi R, Posa A, Tanzilli A, Carchesio F, Pompili M, Manfredi R. Balloon-occluded MWA (b-MWA) followed by balloon-occluded TACE (b-TACE): Technical note on a new combined single-step therapy for single large HCC. Cardiovasc Intervent Radiol. 2020;43(11):1702–7. https://doi.org/10.1007/s00270-020-02583-6.
Crocetti L, Iezzi R, Goldberg SN, et al. The ten commandments of liver ablation: expert discussion and report from mediterranean interventional oncology (MIOLive) congress 2017. Eur Rev Med Pharmacol Sci. 2018;22(12):3896–904. https://doi.org/10.26355/eurrev_201806_15274.
Chernyak V, Fowler KJ, Kamaya A, et al. Liver imaging reporting and data system (LI-RADS) version 2018: imaging of hepatocellular carcinoma in at-risk patients. Radiology. 2018;289(3):816–30. https://doi.org/10.1148/radiol.2018181494.
Filippiadis DK, Binkert C, Pellerin O, Hoffmann RT, Krajina A, Pereira PL. Cirse quality assurance document and standards for classification of complications: the cirse classification system. Cardiovasc Intervent Radiol. 2017;40(8):1141–6. https://doi.org/10.1007/s00270-017-1703-4.
Lencioni R, Llovet JM. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis. 2010;30(212):52–60. https://doi.org/10.1055/s-0030-1247132.
Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline. Eur J Cancer. 2009;45(2):228–47. https://doi.org/10.1016/j.ejca.2008.10.026.
Puijk RS, Ahmed M, Goldberg SN, Meijerink MR. Consensus guidelines for the definition of time-to-event end points in image-guided tumor ablation: results of the sio and datecan initiative. Radiology. 2021;301(3):533–40. https://doi.org/10.1148/radiol.2021203715.
Urbonas T, Anderson EM, Gordon-Weeks AN, et al. Factors predicting ablation site recurrence following percutaneous microwave ablation of colorectal hepatic metastases. HPB. 2019;21(9):1175–84. https://doi.org/10.1016/j.hpb.2019.01.007.
Irie T, Kuramochi M, Kamoshida T, Takahashi N. Selective balloon-occluded transarterial chemoembolization for patients with one or two hepatocellular carcinoma nodules: retrospective comparison with conventional super-selective TACE. Hepatol Res. 2016;46(2):209–14. https://doi.org/10.1111/hepr.12564.
Sugimoto K, Saguchi T, Saito K, Imai Y, Moriyasu F. Hemodynamic changes during balloon-occluded transarterial chemoembolization (B-TACE) of hepatocellular carcinoma observed by contrast-enhanced ultrasound. J Med Ultrason. 2014;41(2):209–15. https://doi.org/10.1007/s10396-013-0487-7.
Cazzato RL, De Marini P, Leclerc L, et al. Large nearly spherical ablation zones are achieved with simultaneous multi-antenna microwave ablation applied to treat liver tumours. Eur Radiol. 2020;30(2):971–5. https://doi.org/10.1007/s00330-019-06431-1.
Garnon J, Delmas L, De Marini P, et al. Triple-antenna microwave ablation with repositioning for the creation of a reliable 6-cm ablation zone in the liver. Cardiovasc Intervent Radiol. 2021;44(8):1291–5. https://doi.org/10.1007/s00270-021-02854-w.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent for the procedure for anonymized publication of this series of patients was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lucatelli, P., Argirò, R., Crocetti, L. et al. Percutaneous Thermal Segmentectomy: Proof of Concept. Cardiovasc Intervent Radiol 45, 665–676 (2022). https://doi.org/10.1007/s00270-022-03117-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-022-03117-y