Abstract
Background
Cancer of the thyroid gland is rare in children and adolescents. A history of neck irradiation is a well-established risk factor for tumor development, and most previous reports focused on cases that were induced by radiation exposure. We present here a retrospective review of the clinical features, treatment, and long-term outcome of children and adolescents with papillary thyroid cancer (PTC) without a history of radiation exposure who were treated at our institution over a period of ~50 years.
Methods
We retrospectively investigated 142 PTC patients without an irradiation history who were younger than 20 years of age when treated from 1961 to 2005 (17 males and 125 females; mean age = 16.3 ± 2.7 years; follow-up = 21.8 ± 12.0 years). The clinicopathological results were evaluated using the medical records. Disease-free survival (DFS) and cause-specific survival (CSS) were assessed with the Kaplan-Meier method and compared with the log-rank test. Parametric analyses were performed using Student’s t test and nonparametric analyses were performed using the Mann–Whitney U test.
Results
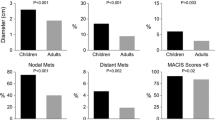
At diagnosis, three patients had distant lung metastasis and 33 had gross neck lymph node (LN) metastasis. All patients were treated with surgery (hemi/partial thyroidectomy in 45 patients, subtotal thyroidectomy in 85, total thyroidectomy in 12, no LN dissection in 50, central compartment dissection in 20, and modified radical neck dissection in 72), and postoperative external beam radiation therapy was administered to 59. Postoperative ablative therapy using I131 was not performed in this series. Recurrence was found for regional LN (n = 25), lung (n = 9), remnant thyroid (n = 5), and others (n = 4). DFS and CSS at 40 years were 74.1 and 97.5%, respectively. DFS was significantly worse in patients aged <16 years with a family history of thyroid cancer, preoperative neck gross LN metastasis, maximum tumor diameter, and extrathyroidal invasion. Preoperative gross neck LN metastasis and distant metastasis at diagnosis were significant factors for CSS. No other factors contributed to DFS and CSS. When the clinical features of children and adolescents were compared, the incidence of preoperative gross neck LN metastasis and distant metastasis at diagnosis and tumors with a maximum diameter >10 mm were significantly higher in the children group than in the adolescent group. DFS was significantly shorter in the children group than in the adolescent group, but no significant difference was found in CSS between these two groups.
Conclusions
The prognosis of PTC in children and adolescents is excellent, regardless of the extent of thyroidectomy and LN dissection. We recommend that only children or adolescents with preoperative gross neck LN metastasis and distant metastasis at diagnosis should be subjected to postoperative ablative therapy.




Similar content being viewed by others
References
Ceccarelli C, Pacini F, Lippi F et al (1988) Thyroid cancer in children and adolescents. Surgery 104:1143–1148
Fassina AS, Rupolo M, Pelizzo MR et al (1994) Thyroid cancer in children and adolescents. Tumori 80:257–262
Danese D, Gardini A, Farsetti A et al (1997) Thyroid carcinoma in children and adolescents. Eur J Pediatr 156:190–194
Segal K, Shvero J, Stern Y et al (1998) Surgery of thyroid cancer in children and adolescents. Head Neck 20:293–297
Dottorini ME, Vignati A, Mazzucchelli L et al (1997) Differentiated thyroid carcinoma in children and adolescents: a 37-year experience in 85 patients. J Nucl Med 38:669–675
Zimmerman D, Hay ID, Gough IR et al (1988) Papillary thyroid carcinoma in children and adults: long-term follow-up of 1039 patients conservatively treated at one institution during three decades. Surgery 104:1157–1166
Pacini F, Vorontsova T, Demidchik EP et al (1997) Post-Chernobyl thyroid carcinoma in Belarus children and adolescents: comparison with naturally occurring thyroid carcinoma in Italy and France. J Clin Endocrinol Metab 82:3563–3569
Demidchik YE, Demidchik EP, Reiners C et al (2006) Comprehensive clinical assessment of 740 cases of surgically treated thyroid cancer in children of Belarus. Ann Surg 243:525–532
Baverstock KF (1993) Thyroid cancer in children in Belarus after Chernobyl. World Health Stat Q 46:204–208
Nikiforov Y, Gnepp DR, Fagin JA (1996) Thyroid lesions in children and adolescents after the Chernobyl disaster: implications for the study of radiation tumorigenesis. J Clin Endocrinol Metab 81:9–14
Tronko MD, Bogdanova TI, Komissarenko IV (1999) Thyroid carcinoma in children and adolescents in Ukraine after the Chernobyl nuclear accident: statistical data and clinicomorphologic characteristics. Cancer 86:149–156
Antonelli A, Miccoli P, Derzhitski VE et al (1996) Epidemiologic and clinical evaluation of thyroid cancer in children from the Gomel region (Belarus). World J Surg 20:867–871. doi:10.1007/s002689900132
La Quaglia MP, Corbally MT, Heller G et al (1988) Recurrence and morbidity in differentiated thyroid carcinoma in children. Surgery 104:1149–1156
Welch Dinauer CA, Tuttle RM, Robie DK et al (1998) Clinical features associated with metastasis and recurrence of differentiated thyroid cancer in children, adolescents and young adults. Clin Endocrinol (Oxf) 49:619–628
Newman KD, Black T, Heller G et al (1998) Differentiated thyroid cancer: determinants of disease progression in patients <21 years of age at diagnosis: a report from the Surgical Discipline Committee of the Children’s Cancer Group. Ann Surg 227:533–541
Landau D, Vini L, A’Hern R et al (2000) Thyroid cancer in children: the Royal Marsden Hospital experience. Eur J Cancer 36:214–220
Jarzab B, Handkiewicz Junak D, Włoch J et al (2000) Multivariate analysis of prognostic factors for differentiated thyroid carcinoma in children. Eur J Nucl Med 27:833–841
Hay ID, Gonzalez-Losada T, Reinalda MS et al (2010) Long-term outcome in 215 children and adolescents with papillary thyroid cancer treated during 1940 through 2008. World J Surg 34:1192–1202. doi:10.1007/s00268-009-0364-0
Brink JS, van Heerden JA, McIver B et al (2000) Papillary thyroid cancer with pulmonary metastases in children: long-term prognosis. Surgery 128:881–886
Noguchi S, Murakami N, Kawamoto H (1994) Classification of papillary cancer of the thyroid based on prognosis. World J Surg 18:552–557. doi:10.1007/BF00353763
Mazzaferri EL, Jhiang SM (1994) Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer. Am J Med 97:418–428
American Joint Committee on Cancer (2002) Chapter 8: thyroid. In: AJCC cancer staging handbook, 6th ed. Springer, New York, pp 89-98
Cady B (1998) Papillary carcinoma of the thyroid gland: treatment based on risk group definition. Surg Oncol Clin N Am 7:633–644
Schlumberger MJ, Filetti S, Hay ID (2002) Nontoxic goiter and thyroid neoplasia. In: Larsen RP, Kronenberg HM, Melmed S, Polonsky KS (eds) Williams textbook of endocrinology, 10th edn. Saunders, Philadelphia, pp 457–490
Hay ID, Bergstralh EJ, Goellner JR et al (1993) Predicting outcome in papillary thyroid carcinoma: development of a reliable prognostic scoring system in a cohort of 1779 patients surgically treated at one institution during 1940 through 1989. Surgery 114:1050–1057
Mazzaferri EL, Kloos RT (2001) Clinical review 128: current approaches to primary therapy for papillary and follicular thyroid cancer. J Clin Endocrinol Metab 86:1447–1463
Yamashita S, Saenko V (2007) Mechanisms of disease: molecular genetics of childhood thyroid cancers. Nat Clin Pract Endocrinol Metab 3:422–429
Lima J, Trovisco V, Soares P et al (2004) BRAF mutations are not a major event in post-Chernobyl childhood thyroid carcinomas. J Clin Endocrinol Metab 89:4267–4271
Penko K, Livezey J, Fenton C et al (2005) BRAF mutations are uncommon in papillary thyroid cancer of young patients. Thyroid 15:320–325
Robinson DW, Orr TG (1955) Carcinoma of the thyroid and other diseases of the thyroid in identical twins. AMA Arch Surg 70:923–928
Ron E, Kleinerman RA, Boice JD Jr et al (1987) Population-based case-control study of thyroid cancer. J Natl Cancer Inst 79:1–12
Hemminki K, Vaittinen P (1999) Familial cancers in a nationwide family cancer database: age distribution and prevalence. Eur J Cancer 35:1109–1117
Goldgar DE, Easton DF, Cannon-Albright LA et al (1994) Systematic population-based assessment of cancer risk in first-degree relatives of cancer probands. J Natl Cancer Inst 86:1600–1608
Stoffer SS, Van Dyke DL, Bach JV et al (1986) Familial papillary carcinoma of the thyroid. Am J Med Genet 25:775–782
Kraimps JL, Bouin-Pineau MH, Amati P et al (1997) Familial papillary carcinoma of the thyroid. Surgery 121:715–718
Lupoli G, Vitale G, Caraglia M et al (1999) Familial papillary thyroid microcarcinoma: a new clinical entity. Lancet 353:637–639
Hrafnkelsson J, Tulinius H, Jonasson JG et al (1989) Papillary thyroid carcinoma in Iceland. A study of the occurrence in families and the coexistence of other primary tumours. Acta Oncol 28:785–788
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Enomoto, Y., Enomoto, K., Uchino, S. et al. Clinical Features, Treatment, and Long-Term Outcome of Papillary Thyroid Cancer in Children and Adolescents Without Radiation Exposure. World J Surg 36, 1241–1246 (2012). https://doi.org/10.1007/s00268-012-1558-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-012-1558-4