Abstract
Background
Injection-related pain of dermal fillers is a consistent and bothersome problem for patients undergoing soft tissue augmentation. Reducing the pain could improve overall patient satisfaction.
Objective
The purpose of this study was to compare the pain relief, efficacy, and safety of HA IDF plus containing lidocaine with HA IDF without lidocaine during correction of nasolabial folds (NLFs).
Methods
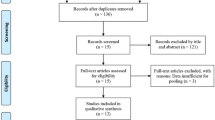
Sixty-two subjects were enrolled in a randomized, multicenter, double-blind, split-face study of HA IDF plus and HA IDF for NLF correction. For split-face study, HA IDF plus was injected to one side of NLF, and HA IDF was injected to the other side. The first evaluation variable was the injection site pain measured using a 100-mm visual analogue scale (VAS). The second evaluation variables included the global aesthetic improvement scale, wrinkle severity rating scale, and adverse events.
Results
Immediately after injection, 91.94% of subjects experienced at least 10 mm decrease in VAS scores at the side injected with HA IDF plus compared with HA IDF, and the rate of subjects is statistically significant. The two fillers were not significantly different in safety profile or wrinkle correction during the follow-up visit.
Conclusions
HA IDF plus significantly reduced the injection-related pain during NLFs correction compared with HA IDF without altering clinical outcomes or safety. Both HA IDF plus and HA IDF were considerably tolerated and most adverse reactions were mild and transient.
Level of evidence I
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.


Similar content being viewed by others
References
Flament F, Bazin R, Laquieze S, Rubert V, Simonpietri E, Piot B (2013) Effect of the sun on visible clinical signs of aging in Caucasian skin. Clin Cosmet Investig Dermatol 6:221–232
Kahn DM, Shaw RB (2010) Overview of current thoughts on facial volume and aging. Fac Plast Surg 26(5):350–355
Farage MA, Miller KW, Elsner P, Maibach HI (2008) Intrinsic and extrinsic factors in skin ageing: a review. Int J Cosmet Sci 30(2):87–95
Levy PM, De Boulle K, Raspaldo H (2009) A split-face comparison of a new hyaluronic acid facial filler containing pre-incorporated lidocaine versus a standard hyaluronic acid facial filler in the treatment of naso-labial folds. J Cosmet Laser Ther 11(3):169–173
Puizina-Ivić N (2008) Skin aging. Acta Dermatovenerol Alp Pannonica Adriat 17(2):47–54
Choi WJ, Han SW, Kim JE, Kim HW, Kim MB, Kang H (2015) The efficacy and safety of lidocaine-containing hyaluronic acid dermal filler for treatment of nasolabial folds: a multicenter, randomized clinical study. Aesthet Plast Surg 39(6):953–962
2015 Plastic surgery statistics report: American society of plastic surgeons. https://d2wirczt3b6wjm.cloudfront.net/News/Statistics/2015/plastic-surgery-statistics-full-report-2015.pdf
Edwards PC, Fantasia JE (2007) Review of long-term adverse effects associated with the use of chemically-modified animal and nonanimal source hyaluronic acid dermal fillers. Clin Interv Aging 2(4):509–519
Tierney EP, Hanke CW (2009) Recent trends in cosmetic and surgical procedure volumes in dermatologic surgery. Dermatol Surg 35(9):1324–1333
Kim JE, Sykes JM (2011) Hyaluronic acid fillers: history and overview. Fac Plast Surg 27(6):523–528
Jeon JH, Kim JH, Lee JW, Park KY, Seo SJ, Song HJ (2013) Split-face comparison study of new hyaluronic acid filler for efficacy and safety of HA-IDF versus Restylane® for correction of nasolabial folds. Korean Dermatological Association, Seoul
Monheit GD, Campbell RM, Neugent H, Nelson CP, Prather CL, Bachtell N et al (2010) Reduced pain with use of proprietary hyaluronic acid with lidocaine for correction of nasolabial folds: a patient-blinded, prospective, randomized controlled trial. Dermatol Surg 36(1):94–101
Brandt F, Bank D, Cross SL, Weiss R (2010) A lidocaine-containing formulation of large-gel particle hyaluronic acid alleviates pain. Dermatol Surg 36(Suppl 3):1876–1885
Brandt FS, Cazzaniga A (2008) Hyaluronic acid gel fillers in the management of facial aging. Clin Interv Aging 3(1):153–159
Matarasso SL, Carruthers JD, Jewell ML (2006) Consensus recommendations for soft tissue augmentation with nonanimal stabilized hyaluronic acid (Restylane). Plast Reconstr Surg 117(3):3S–34S discussion (5S–43S)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to disclose in relation to the content of this article.
Additional information
The correct spelling of the first author’s name is Jong-Hun Lee. His correct affiliation is Department of Plastic and Reconstructive Surgery, Nowon Eulji Medical Center, School of Medicine, Eulji University, 68 Hangeulbiseok-gil, Nowon-gu, Seoul 01830, Republic of Korea. The correct affiliation of the corresponding author is Department of Plastic and Reconstructive Surgery, Soonchunhyang University Bucheon Hospital, 170 Jomaru-ro, Wonmi-gu, Bucheon-si, Gyeonggi-do 14584, Republic of Korea.
An erratum to this article is available at http://dx.doi.org/10.1007/s00266-017-0782-6.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, JH., Kim, SH. & Park, ES. The Efficacy and Safety of HA IDF Plus (with Lidocaine) Versus HA IDF (Without Lidocaine) in Nasolabial Folds Injection: A Randomized, Multicenter, Double-Blind, Split-Face Study. Aesth Plast Surg 41, 422–428 (2017). https://doi.org/10.1007/s00266-016-0769-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-016-0769-8