Abstract
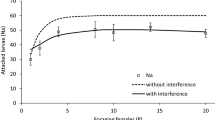
Gregariousness in insects has been shown to improve performance, feeding, and defense against predation. Nevertheless, an increased risk of antagonistic interactions such as competition and predation occurs in group living. We examined the effects of both group size and larval age of aggregations of the butterfly Ascia monuste orseis (Godart, 1819) on larval permanency and attack by natural enemies. Group behavior was also investigated with respect to predation risk, as well as larval responses after enemy attack. One larva and groups of 10 and 50 larvae were assigned on host plants and monitored daily to evaluate larval permanency. At each instar, a subset of groups was observed for a 4-h period to identify natural enemies and to record both group behaviors performed at the moment of attack and responses to the attacks. Permanency on the host was higher in aggregations than in solitary larvae. Larger aggregations conferred greater protection to A. monuste orseis larvae against natural enemies than did smaller aggregations, through lower per capita predation. Parasitoidism was more intense in small-sized larvae, whereas late instars were more susceptible to predators. Similarly to solitary species, larvae were most susceptible to attacks when performing searching and feeding compared to resting and moving. Moving away and headrearing were observed for all instars and groups after enemy attacks and rarely protected larvae against natural enemies. Our results show that behaviors that involve head movements posed more attack risks than those that involve moving the whole body or absence of movements, and that larval aggregation in A. monuste orseis rarely exhibits defensive behaviors against natural enemies. Larval defense is provided through dilution effect.
Significance statement
Natural enemies can use several cues to find their prey, as for example, prey morphology, chemistry, and behavior. Nevertheless, group behavior has been poorly investigated with respect to mortality risks in phytophagous insects. This study investigated whether certain group sizes and behaviors are more risky than others in the gregarious Ascia monuste larvae. Larvae were susceptible to both parasitoids and predators, and larger groups conferred more protection against natural enemies than smaller ones. Similarly to species with a solitary habit, behaviors that involved head movements as searching and feeding posed more attack risks than moving and resting. Defense behaviors were not very effective against natural enemies. Our study reveals that both solitary and gregarious species may share more similarities in terms of susceptibility to natural enemies than previously thought.



Similar content being viewed by others
References
Altmann J (1984) Observational sampling methods for insect behavioural ecology. Fla Entomol 67(1):50–56. https://doi.org/10.2307/3494104
Barros-Bellanda HCH, Zucoloto FS (2001) Influence of chorion ingestion on the performance of Ascia monuste and its association with cannibalism. Ecol Entomol 26(6):557–561. https://doi.org/10.1046/j.1365-2311.2001.00377.x
Barros-Bellanda HCH, Zucoloto FS (2003) Importance of larval migration (dispersal) for the survival of Ascia monuste (Godart) (Lepidoptera: Nymphalidae). Neotrop Entomol 32(1):11–17. https://doi.org/10.1590/S1519-566X2003000100002
Barros-Bellanda HCH, Zucoloto FS (2005) Egg cannibalism in Ascia monuste in the field; opportunistic, preferential and very frequent. J Ethol 23(2):133–138. https://doi.org/10.1007/s10164-004-0138-y
Beccaloni GW, Viloria AL, Hall SK, Robinson GS (2008) Catalogue of the hostplants of the Neotropical butterflies, vol 8. Monografías Tercer Milenio, Saragossa
Bergelson JM, Lawton JH (1988) Does foliage influence predation on the insect herbivores of birch? Ecology 69(2):434–445. https://doi.org/10.2307/1940442
Bernays EA (1997) Feeding by lepidopteran larvae is dangerous. Ecol Entomol 22(1):121–123. https://doi.org/10.1046/j.1365-2311.1997.00042.x
Bernays EA, Singer MS, Rodrigues D (2004) Foraging in nature: foraging efficiency and attentiveness in caterpillars with different diet breadths. Ecol Entomol 29(4):389–397. https://doi.org/10.1111/j.0307-6946.2004.00615.x
Burger J, Gochfeld M (2001) Smooth-billed ani (Crotophaga ani) predation on butterflies in Mato Grosso, Brazil: risk decreases with increased group size. Behav Ecol Sociobiol 49(6):482–492. https://doi.org/10.1007/s002650100327
Clark BR, Faeth SH (1997) The consequences of larval aggregation in the butterfly Chlosyne lacina. Ecol Entomol 22(4):408–415. https://doi.org/10.1046/j.1365-2311.1997.00091.x
Cornell JC, Stamp NE, Bowers MD (1987) Developmental change in aggregation, defense and escape behavior of buckmoth caterpillars, Hemileuca lucina (Saturniidae). Behav Ecol Sociobiol 20(6):383–388. https://doi.org/10.1007/BF00302980
Costa JT (2006) The other insect societies. Belknap Press, Cambridge
Crawley MJ (2012) The R book. Wiley, Chichester. https://doi.org/10.1002/9781118448908
Daly D, Higginson AD, Chen D, Ruxton GD, Speed MP (2012) Density-dependent investment in costly anti-predator defences: an explanation for the weak survival benefit of group living. Ecol Lett 15(6):576–583. https://doi.org/10.1111/j.1461-0248.2012.01770.x
Denno RF, Benrey B (1997) Aggregation facilitates larval growth in the neotropical nymphalid butterfly Chlosyne janais. Ecol Entomol 22(2):133–141. https://doi.org/10.1046/j.1365-2311.1997.t01-1-00063.x
Despland E, Hamzeh S (2004) Ontogenetic changes in social behaviour in the forest tent caterpillar, Malacosoma disstria. Behav Ecol Sociobiol 56(2):177–184. https://doi.org/10.1007/s00265-004-0767-8
Fitzgerald TD (1993) Sociality in caterpillars. In: Stamp NE, Casey TM (eds) Caterpillars: ecological and evolutionary constraints on foraging. Chapman and Hall, New York, pp 372–403
Fitzgerald TD, Vissher CR (1996) Foraging behavior and growth of isolated larvae of a social caterpillar, Malacosoma americanum. Entomol Exp Appl 81(3):293–299. https://doi.org/10.1046/j.1570-7458.1996.00099.x
Fordyce JA (2003) Aggregative feeding of pipevine swallowtail larvae enhances hostplant suitability. Oecologia 135(2):250–257. https://doi.org/10.1007/s00442-003-1177-8
Gobbi N, Machado VLL (1986) Material capturado e utilizado na alimentação de Polybia (Trichothorax) ignobilis (Haliday, 1836) (Hymenoptera, Vespidae). An Soc Entomol Brasil 15:117–124
Heinrich B, Collins SL (1983) Caterpillar leaf damage, and the game of hide-and-seek with birds. Ecology 64(3):592–602. https://doi.org/10.2307/1939978
Higginson AD, Delf J, Ruxton GD, Speed MP (2011) Growth and reproductive costs of larval defence in the aposematic lepidopteran Pieris brassicae. J Anim Ecol 80(2):384–392. https://doi.org/10.1111/j.1365-2656.2010.01786.x
Hochberg ME (1991) Viruses as costs to gregarious feeding behaviour in the Lepidoptera. Oikos 61(3):291–296. https://doi.org/10.2307/3545236
Hunter AF (2000) Gregariousness and repellent defenses in the survival of phytophagous insects. Oikos 91(2):213–224. https://doi.org/10.1034/j.1600-0706.2000.910202.x
Kawasaki N, Miyashita T, Kato Y (2009) Leaf toughness changes the effectiveness of larval aggregation in the butterfly Byasa alcinous bradanus (Lepidoptera: Papilionidae). Entomol Sci 12(2):135–140. https://doi.org/10.1111/j.1479-8298.2009.00314.x
Lamas G (2004) Pieridae. In: Lamas G (ed) Atlas of Neotropical Lepidoptera. Vol 5. Checklist: part 4A. Hesperioidea-Papilionoidea. Scientific Publishers, Gainesville, pp 99–117
Lindstedt C, Huttunen H, Kakko M, Mappers J (2011) Disentangling the evolution of weak warning signals: high detection risk and low production cost of chemical defenses in gregarious pine sawfly larvae. Evol Ecol 25(5):1029–1046. https://doi.org/10.1007/s10682-010-9456-4
McClure M, Despland E (2010) Collective foraging patterns of field colonies of Malacosoma disstria caterpillars. Can Entomol 142(05):473–480. https://doi.org/10.4039/n10-001
McClure M, Despland E (2011) Defensive responses by a social caterpillar are tailored to different predators and change with larval instar and group size. Naturwissenchaften 98(5):425–434. https://doi.org/10.1007/s00114-011-0788-x
Montllor CB, Bernays EA (1993) Invertebrate predators and caterpillar foraging. In: Stamp NE, Casey TM (eds) Caterpillars: ecological and evolutionary constraints on foraging. Chapman and Hall, New York, pp 170–202
Penteado-Dias AM (1986) Parasitismo de Ascia monuste orseis (Latreille, 1819) (Lepidoptera: Pieridae) por Cotesia glomerata (L., 1758) (Hymenoptera: Braconidae: Microgastrinae). Rev Bras Entomol 30:257–259
Pough FH, Brower LP (1977) Predation by birds on great southern white butterflies (Ascia monuste) as a function of palatability, sex, and habitat. Am Midl Nat 98(1):50–58. https://doi.org/10.2307/2424714
Reader T, Hochuli DF (2003) Understanding gregariousness in a larval Lepidopteran: the roles of host plant, predation, and microclimate. Ecol Entomol 28(6):729–737. https://doi.org/10.1111/j.1365-2311.2003.00560.x
Riipi M, Alatalo RV, Lindstrom L, Mappes J (2001) Multiple benefits of gregariousness cover detectability costs in aposematic aggregations. Nature 413(6855):512–514. https://doi.org/10.1038/35097061
Ronnäs C, Larsson S, Pitacco A, Battisti A (2010) Effects of colony size on larval performance in a processionary moth. Ecol Entomol 35:436–445
Santana AFK, Zucoloto FS (2016) Benefits of egg clusters in the evolution of larval aggregation in the neotropical butterfly Ascia monuste orseis: reduction of egg failure and enhanced larval hatching. J Lep Soc 70:72–74
Sokal RR, Rohlf FJ (1995) Biometry. Freeman and Company, New York
Stamp NE (1981) Effect of group size on parasitism in a natural population of the Baltimore checkerspot Euphydryas phaeton. Oecologia 49(2):201–206. https://doi.org/10.1007/BF00349188
Stamp NE, Bowers MD (1988) Direct and indirect effects of predatory wasps (Polistes sp.: Vespidae) on gregarious caterpillars (Hemileuca lucina: Saturniidae). Oecologia 75(4):619–624. https://doi.org/10.1007/BF00776428
Turchin P, Kareiva P (1989) Aggregation in Aphis varians: an effective strategy for reducing predation risk. Ecology 70(4):1008–1016. https://doi.org/10.2307/1941369
Weed AS (2010) Benefits of larval group feeding by Chrysolina aurichalcea asclepiadis on Vincetoxicum: improved host location or feeding facilitation? Entomol Exp Appl 137(3):220–228. https://doi.org/10.1111/j.1570-7458.2010.01057.x
Wertheim B, Marchais J, Vet LEM, Dicke M (2002) Allee effect in larval resource exploitation in Drosophila: an interaction among density of adults, larvae, and micro-organisms. Ecol Entomol 27(5):608–617. https://doi.org/10.1046/j.1365-2311.2002.00449.x
Weseloh RM (1993) Potential effects of parasitoids on the evolution of caterpillar foraging behavior. In: Stamp NE, Casey TM (eds) Caterpillars: ecological and evolutionary constraints on foraging. Chapman and Hall, New York, pp 203–223
Wilson K, Knell R, Boots M, Koch-Osborne J (2003) Group living and investment in immune defence: an interspecific analysis. J Anim Ecol 72(1):133–143. https://doi.org/10.1046/j.1365-2656.2003.00680.x
Winter B (2013) Linear models and linear mixed effects models in R with linguistic applications arXiv: 1308.5499. http://arxiv.org/pdf/1308.5499.pdf
Wrona FJ, Dixon RWJ (1991) Group size and predation risk: a field analysis of encounter and dilution effects. Am Nat 137(2):186–201. https://doi.org/10.1086/285153
Acknowledgements
We thank Laércio Massocato for field assistance. Pedro A. P. Rodrigues provided information about ant ecology and Rodrigo Feitosa identified them. Sidnei Mateus and Renner L.C. Baptista identified the predatory wasps and the spider genus, respectively. José R. Trigo and Adriano Mariscal shared information on Ascia monuste orseis and their predatory wasps. Bruna Ramos greatly helped us with statistical analyses and Felipe Pinheiro Machado exchanged ideas about response behaviors. We are very grateful to Melanie McClure, Janet Reid, and three anonymous reviewers for critically reviewing the manuscript.
Funding
We thank the Fundação de Amparo à Pesquisa do Estado de São Paulo (grants 2010/50816-9 and 2007/07802-4) and the Conselho Nacional de Pesquisa (302503/2010-2) for grants to FSZ, DR, and AKFS, respectively.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J. C. Choe
Electronic supplementary material
ESM 1
(DOCX 15 kb)
Rights and permissions
About this article
Cite this article
Santana, A.F.K., Rodrigues, D. & Zucoloto, F.S. Larval aggregation in a Neotropical butterfly: risky behaviors, per capita risk, and larval responses in Ascia monuste orseis . Behav Ecol Sociobiol 71, 174 (2017). https://doi.org/10.1007/s00265-017-2403-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-017-2403-4