Abstract
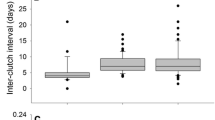
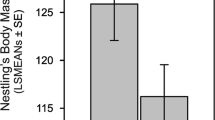
Males can through their behavior (e.g., courtship feeding) exert an indirect effect on their partner’s reproductive traits, such as the seasonal timing and size of her clutch. Evidence for such indirect (male) effect on reproduction is starting to accumulate. We quantify female and male effects on reproduction in the tawny owl Strix aluco using a hierarchical mixed model on data collected in 1978–2013. We find that differences between males explain 7 % of the phenotypic variance in laying date (females 5 %). In contrast, females have a clear (11 %) effect on clutch size, whereas males have no effect. Based on multivariate hierarchical modeling, we find an individual-level correlation between the male-specific effect on laying date and his body mass (but not his plumage color or wing length). Heavy males may be able to affect their partner’s seasonal timing of laying because of an advantage in providing courtship feeding prior to reproduction. Our findings illustrate that males can be an important determinant of variation in reproduction and that multivariate mixed models present a general approach to pinpoint which individual characteristics could be associated with such indirect effects.

Similar content being viewed by others
References
Ah-King M, Ahnesjö I (2013) The “sex role” concept: an overview and evaluation. Evol Biol 40:461–470
Ahola K, Niiranen S (1986) Lehtopöllön iän määrittäminen (age determination of the tawny owl). Lintumies 21:81–85
Auld JR, Perrins CM, Charmantier A (2013) Who wears the pants in a mute swan pair? Deciphering the effects of male and female age and identity on breeding success. J Anim Ecol 82:826–835
Bentler PM (1980) Multivariate analysis with latent variables. Causal modelling. Annu Rev Psychol 31:419–456
Bergsma R, Kanis E, Knol EF, Bijma P (2008) The contribution of social effects to heritable variation in finishing traits of domestic pigs (Sus scrofa). Genetics 178:1559–1570
Bijma P (2014) The quantitative genetics of indirect genetic effects: a selective review of modelling issues. Heredity 112:61–69
Bijma P, Wade MJ (2008) The joint effects of kin, multilevel selection and indirect genetic effects on response to genetic selection. J Evol Biol 21:1175–1188
Bijma P, Muir WM, Ellen ED, Wolf JB, Van Arendonk JAM (2007a) Multilevel selection 2: estimating the genetic parameters determining inheritance and response to selection. Genetics 175:289–299
Bijma P, Muir WM, van Arendonk JAM (2007b) Multilevel selection 1: quantitative genetics of inheritance and response to selection. Genetics 175:277–288
Bleakley BH, Brodie ED III (2009) Indirect genetic effects influence antipredator behavior in guppies: estimates of the coefficient of interaction psi and the inheritance of reciprocity. Evolution 63:1796–1806
Brommer JE (2013) On between-individual and residual (co)variances in the study of animal personality: are you willing to make the individual gambit? Behav Ecol Sociobiol 67:1027–1032
Brommer JE, Rattiste K (2008) ‘Hidden’ reproductive costs between mates in a wild bird population. Evolution 62:2326–2333
Brommer JE, Ahola K, Karstinen T (2005) The colour of fitness: plumage coloration and lifetime reproductive success in the tawny owl. Proc R Soc Lond B 272:935–940
Brommer JE, Kontiainen P, Pietiäinen H (2012) Selection on plasticity of seasonal life-history traits using random regression mixed model analysis. Ecol Evol 2:695–704
Brommer JE, Karell P, Ahola K, Karstinen T (2014) Residual correlations, and not intrinsic properties of the individuals, determine a nest defense boldness syndrome in tawny owls. Behav Ecol 25:802–812
Browne WJ, McCleery RH, Sheldon BC, Pettifor RA (2007) Using cross-classified multivariate mixed response models with application to life history traits in great tits (Parus major). Stat Model 7:217–238
Butler DG, Cullis BR, Gilmour AR, Gogel BJ (2009) ASReml-R reference manual version 3. Department of Primary Industries and Fisheries, State of Queensland, Australia
Chapman T, Arnqvist G, Bangham J, Rowe L (2003) Sexual conflict. Trends Ecol Evol 18:41–47
Charmantier A, Perrins C, McCleery RH, Sheldon BC (2006) Evolutionary response to selection in clutch size in a long-term study of the mute swan. Am Nat 167:453–465
Da Silva A, van den Brink V, Emaresi G, Luzio E, Bize P, Dreiss AN, Roulin A (2013) Melanin-based colour polymorphism signals aggressive personality in nest and territory defence in the tawny owl (Strix aluco). Behav Ecol Sociobiol 67:1041–1052
Dingemanse NJ, Dochtermann NA, Nakagawa S (2012) Defining behavioural syndromes and the role of ‘syndrome deviation’ in understanding their evolution. Behav Ecol Sociobiol 66:1543–1548
Ducrest AL, Keller L, Roulin A (2008) Pleiotropy in the melanocortin system, coloration and behavioural syndromes. Trends Ecol Evol 23:502–510
Edward DA, Poissant J, Wilson AJ, Chapman T (2014) Sexual conflict and interacting phenotypes: a quantitative genetic analysis of fecundity and copula duration in Drosophila melanogaster. Evolution 68:1651–1660
Ellen ED, Visscher J, van Arendonk JAM, Bijma P (2008) Survival of laying hens: genetic parameters for direct and associative effects in three purebred layer lines. Poult Sci 87:233–239
Forstmeier W (2004) Female resistance to male seduction in zebra finches. Anim Behav 68:1005–1015
Gasparini J, Bize P, Piault R, Wakamatsu K, Blount JD, Ducrest AL, Roulin A (2009) Strength and cost of an induced immune response are associated with a heritable melanin‐based colour trait in female tawny owls. J Anim Ecol 78:608–616
Gienapp P, Postma E, Visser ME (2006) Why breeding time has not responded to selection for earlier breeding in a songbird population. Evolution 60:2381–2388
Griffing B (1967) Selection in reference to biological groups. I. Individual and group selection applied to populations of unordered groups. Aust J Biol Sci 20:127–139
Griffing B (1981) A theory of natural selection incorporating interaction among individuals. I. The modeling process. J Theor Biol 89:635–658
Gwynne DT (2008) Sexual conflict over nuptial gifts in insects. Annu Rev Entomol 53:83–101
Hadfield JD, Wilson AJ (2007) Multilevel selection 3: modeling the effects of interacting individuals as a function of group size. Genetics 17:667–668
Hakkarainen H, Huhta E, Lahti K, Lundvall P, Mappes T, Tolonen P, Wiehn J (1996) A test of male mating and hunting success in the kestrel: the advantages of smallness? Behav Ecol Sociobiol 39:375–380
Hall MD, Lailvaux SP, Brooks RC (2013) Sex-specific evolutionary potential of pre- and postcopulatory reproductive interactions in the field cricket Teleogryphus commodus. Evolution 67:1831–1837
Hirons GJM (1985) The importance of body reserves for successful reproduction in the tawny owl (Strix aluco). J Zool Lond B 1:1–20
Horvathova T, Nakagawa S, Uller T (2012) Strategic female reproductive investment in response to male attractiveness in birds. Proc R Soc Lond B 279:163–170
Karell P, Kontiainen P, Pietiäinen H, Siitari H, Brommer JE (2008) Maternal effects on offspring Igs and egg size in relation to natural and experimentally improved food supply. Funct Ecol 22:682–690
Karell P, Ahola K, Karstinen T, Zolei A, Brommer JE (2009) Population dynamics in a cyclic environment. Consequences of cyclic food abundance on tawny owl reproduction and survival. J Anim Ecol 78:1050–1062
Karell P, Ahola K, Karstinen T, Brommer JE (2013) Brown tawny owls moult more flight feathers than grey ones. J Avian Biol 44:235–244
Kekkonen J, Kolunen H, Pietiäinen H, Karell P, Brommer JE (2008) Tawny owl reproduction and offspring sex ratios under variable food conditions. J Ornith 149: 59–66
Korpimäki E (1986) Reversed size dimorphism in birds of prey, especially in Tengmalm’s owl Aegolius funereus: a test of the “ starvation hypothesis”. Ornis Scand 17:326–332
Ludwigs JD, Becker PH (2005) What do pairing patterns in common tern, Sterna hirundo, recruits reveal about the significance of sex and breeding experience? Behav Ecol Sociobiol 57:412–421
Lundberg A (1986) Adaptive advantages of reversed sexual size dimorphism in European owls. Ornis Scand 17:133–140
Lynch M, Walsh B (1998) Genetics and analysis of quantitative traits. Sinauer Associates, Sunderland, MA
McCleery RH, Pettifor RA, Armbruster P, Meyer K, Sheldon BC, Perrins CM (2004) Components of variance underlying fitness in a natural population of the great tit Parus major. Am Nat 164:E62–E72
McGlothlin JW, Brodie ED III (2009) How to measure indirect genetic effects: the congruence of trait-based and variance partitioning approaches. Evolution 63:1785–1795
Mikkola H (1983) Owls of Europe. T & AD Poyser, Calton, UK
Moore AJ, Brodie ED III, Wolf JB (1997) Interacting phenotypes and the evolutionary process: I direct and indirect genetic effects of social interactions. Evolution 51:1352–1362
Petty SJ (1992) A guide to age determination of tawny owl Strix aluco. In: Galbraith CA, Taylor IR, Percival S (eds) The ecology and conservation of European owls). Joint Nature Conservation Committee, UK Nature Conservation, Peterborough, pp 89–91
Pietiäinen H, Kolunen H (1993) Female body condition and breeding of the Ural owl Strix uralensis. Funct Ecol 7:726–735
Pinheiro JC, Bates DM (2000) Mixed effects models in S and S-PLUS. Springer, New York
Rice SH (2004) Evolutionary theory. Mathematical and conceptual foundations, Sinauer, Sunderland
Sasvári L, Hegyi Z, Csörgõ T, Hahn I (2000) Age-dependent diet change, parental care and reproductive costin tawny owls Strix aluco. Acta Oecol 21:267–275
Sheldon BC, Kruuk LEB, Merilä J (2003) Natural selection and inheritance of breeding time and clutch size in the collared flycatcher. Evolution 57:406–420
Shuster SS, Wade MJ (2003) Mating systems and strategies. Princeton University Press, Princeton
Teplitsky C, Mills JA, Yarrall JW, Merilä J (2010) Indirect genetic effects in a sex‐limited trait: the case of breeding time in red‐billed gulls. J Evol Biol 23:935–944
van de Pol M, Heg D, Bruinzeel LW, Kuijper B, Verhulst S (2006) Experimental evidence for a causal effect of pair-bond duration on reproductive performance in oystercatchers (Haematopus ostralegus). Behav Ecol 17:982–991
Wilson AJ, Gelin U, Perron M-C, Réale D (2009) Indirect genetic effects and the evolution of aggression in a vertebrate system. Proc R Soc Lond B 276:533–541
Acknowledgments
This work was supported by the Academy of Finland (to PK). All members of Kimpari Bird Projects (KBP) and Veikko Tarsa are thanked for their help in collecting field data on the tawny owls. Reviewers are thanked for their comments which improved the quality of this paper. This is publication nr 17 from KBP.
Ethical standards
The procedures required to collect the data described in this work include capture of individuals, marking individuals by ringing, and measuring their morphology, which all fall under the ringing licenses of the authors, as provided by the relevant local authorities.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. Pilastro
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 96 kb)
Rights and permissions
About this article
Cite this article
Brommer, J.E., Karell, P., Aaltonen, E. et al. Dissecting direct and indirect parental effects on reproduction in a wild bird of prey: dad affects when but not how much. Behav Ecol Sociobiol 69, 293–302 (2015). https://doi.org/10.1007/s00265-014-1842-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-014-1842-4