Abstract
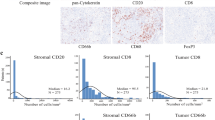
The prognostic value of the local immune phenotype in patients with colorectal cancer has been extensively studied. Neoadjuvant radiotherapy and/or chemotherapy may potentially influence these immune responses. In this study, we examined the prognostic role of indoleamine-2,3-Dioxygenase (IDO1) and infiltrating cytotoxic T lymphocytes (CD8+) in locally advanced rectal carcinomas after neoadjuvant treatment. Expression of IDO1 and CD8 was evaluated by immunohistochemistry in 106 archival tumour tissue samples from patients following neoadjuvant chemoradiation and radical resection. The average infiltration of IDO1+ and CD8+ cells was calculated along the tumour invasive front, in the tumour centre and within the neoplastic cells and expressed as total scores. Of the tumour specimens evaluable for immunohistochemistry, 100% showed CD8+ lymphocyte infiltration and 93.4% stained positive for IDO1. Total IDO1 score positively correlated with total CD8 score for all three subsites (p = 0.002, Kendall-tau-b 0.357). A high total CD8 score was positively correlated with lower ypUICC-stages (p = 0.047) and lower ypT-categories (p = 0.032). Total IDO1 expression showed a clear trend towards a lower risk of recurrence (p = 0.078). A high total IDO1 score was an independent prognostic marker for prolonged disease-free survival (HR 0.38, p = 0.046) and a high total CD8 score for favourable overall survival (HR 0.16, p = 0.029). Analysis of the local CD8 and IDO1 expression profile may be a helpful tool in predicting prognosis for patients with locally advanced rectal cancer following neoadjuvant chemoradiation.


Similar content being viewed by others
Abbreviations
- AJCC/UICC:
-
American Joint Committee on Cancer/Union Internationale Contre le Cancer
- CD8:
-
Cytotoxic T lymphocyte
- DFS:
-
Disease-free survival
- HR:
-
Hazard ratio
- IDO1:
-
Indoleamine-2,3-Dioxygenase 1
- IE:
-
Intraepithelial
- LARC:
-
Locally advanced rectal cancer
- OS:
-
Overall survival
- ST:
-
Stromal
- TDO:
-
Tryptophan-2,3-dioxygenase
- TME:
-
Total mesorectal excision
- TNM:
-
Tumour-node-metastasis
References
Siegel RL, Miller KD, Jemal A (2017) Cancer Statistics, 2017. CA Cancer J Clin 67(1):7–30. https://doi.org/10.3322/caac.21387
Torre LA, Siegel RL, Ward EM, Jemal A (2016) Global Cancer Incidence and Mortality Rates and Trends–An Update. Cancer Epidemiol Biomarkers Prev 25(1):16–27. https://doi.org/10.1158/1055-9965.EPI-15-0578
Heald RJ, Ryall RD (1986) Recurrence and survival after total mesorectal excision for rectal cancer. Lancet 1(8496):1479–1482
Sauer R, Becker H, Hohenberger W, Rodel C, Wittekind C, Fietkau R, Martus P, Tschmelitsch J, Hager E, Hess CF, Karstens JH, Liersch T, Schmidberger H, Raab R, German Rectal Cancer Study G (2004) Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med 351(17):1731–1740. https://doi.org/10.1056/NEJMoa040694
Kapiteijn E, Marijnen CA, Nagtegaal ID, Putter H, Steup WH, Wiggers T, Rutten HJ, Pahlman L, Glimelius B, van Krieken JH, Leer JW, van de Velde CJ (2001) Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. Dutch Colorectal Cancer N Engl J Med 345(9):638–646. https://doi.org/10.1056/NEJMoa010580
Sauer R, Liersch T, Merkel S, Fietkau R, Hohenberger W, Hess C, Becker H, Raab HR, Villanueva MT, Witzigmann H, Wittekind C, Beissbarth T, Rodel C (2012) Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. J Clin Oncol 30(16):1926–1933. https://doi.org/10.1200/JCO.2011.40.1836
van Gijn W, Marijnen CA, Nagtegaal ID, Kranenbarg EM, Putter H, Wiggers T, Rutten HJ, Pahlman L, Glimelius B, van de Velde CJ, Dutch Colorectal Cancer G (2011) Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicentre, randomised controlled TME trial. Lancet Oncol 12(6):575–582. https://doi.org/10.1016/S1470-2045(11)70097-3
Smith JJ, Garcia-Aguilar J (2015) Advances and challenges in treatment of locally advanced rectal cancer. J Clin Oncol 33(16):1797–1808. https://doi.org/10.1200/JCO.2014.60.1054
Edge SB, Compton CC (2010) The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol 17(6):1471–1474. https://doi.org/10.1245/s10434-010-0985-4
Nagtegaal ID, Quirke P, Schmoll HJ (2011) Has the new TNM classification for colorectal cancer improved care? Nat Rev Clin Oncol 9(2):119–123. https://doi.org/10.1038/nrclinonc.2011.157
Benson AB 3rd, Bekaii-Saab T, Chan E, Chen YJ, Choti MA, Cooper HS, Engstrom PF, Enzinger PC, Fakih MG, Fuchs CS, Grem JL, Hunt S, Leong LA, Lin E, Martin MG, May KS, Mulcahy MF, Murphy K, Rohren E, Ryan DP, Saltz L, Sharma S, Shibata D, Skibber JM, Small W Jr, Sofocleous CT, Venook AP, Willett CG, Freedman-Cass DA, Gregory KM (2012) Rectal cancer. J Natl Compr Canc Netw 10(12):1528–1564
Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pages C, Tosolini M, Camus M, Berger A, Wind P, Zinzindohoue F, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Pages F (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313(5795):1960–1964. https://doi.org/10.1126/science.1129139
Mlecnik B, Tosolini M, Kirilovsky A, Berger A, Bindea G, Meatchi T, Bruneval P, Trajanoski Z, Fridman WH, Pages F, Galon J (2011) Histopathologic-based prognostic factors of colorectal cancers are associated with the state of the local immune reaction. J Clin Oncol 29(6):610–618. https://doi.org/10.1200/JCO.2010.30.5425
Galon J, Pages F, Marincola FM, Angell HK, Thurin M, Lugli A, Zlobec I, Berger A, Bifulco C, Botti G, Tatangelo F, Britten CM, Kreiter S, Chouchane L, Delrio P, Arndt H, Asslaber M, Maio M, Masucci GV, Mihm M, Vidal-Vanaclocha F, Allison JP, Gnjatic S, Hakansson L, Huber C, Singh-Jasuja H, Ottensmeier C, Zwierzina H, Laghi L, Grizzi F, Ohashi PS, Shaw PA, Clarke BA, Wouters BG, Kawakami Y, Hazama S, Okuno K, Wang E, O’Donnell-Tormey J, Lagorce C, Pawelec G, Nishimura MI, Hawkins R, Lapointe R, Lundqvist A, Khleif SN, Ogino S, Gibbs P, Waring P, Sato N, Torigoe T, Itoh K, Patel PS, Shukla SN, Palmqvist R, Nagtegaal ID, Wang Y, D’Arrigo C, Kopetz S, Sinicrope FA, Trinchieri G, Gajewski TF, Ascierto PA, Fox BA (2012) Cancer classification using the Immunoscore: a worldwide task force. J Transl Med 10:205. https://doi.org/10.1186/1479-5876-10-205
Pages F, Mlecnik B, Marliot F, Bindea G, Ou FS, Bifulco C, Lugli A, Zlobec I, Rau TT, Berger MD, Nagtegaal ID, Vink-Borger E, Hartmann A, Geppert C, Kolwelter J, Merkel S, Grutzmann R, Van den Eynde M, Jouret-Mourin A, Kartheuser A, Leonard D, Remue C, Wang JY, Bavi P, Roehrl MHA, Ohashi PS, Nguyen LT, Han S, MacGregor HL, Hafezi-Bakhtiari S, Wouters BG, Masucci GV, Andersson EK, Zavadova E, Vocka M, Spacek J, Petruzelka L, Konopasek B, Dundr P, Skalova H, Nemejcova K, Botti G, Tatangelo F, Delrio P, Ciliberto G, Maio M, Laghi L, Grizzi F, Fredriksen T, Buttard B, Angelova M, Vasaturo A, Maby P, Church SE, Angell HK, Lafontaine L, Bruni D, El Sissy C, Haicheur N, Kirilovsky A, Berger A, Lagorce C, Meyers JP, Paustian C, Feng Z, Ballesteros-Merino C, Dijkstra J, van de Water C, van Lent-van Vliet S, Knijn N, Musina AM, Scripcariu DV, Popivanova B, Xu M, Fujita T, Hazama S, Suzuki N, Nagano H, Okuno K, Torigoe T, Sato N, Furuhata T, Takemasa I, Itoh K, Patel PS, Vora HH, Shah B, Patel JB, Rajvik KN, Pandya SJ, Shukla SN, Wang Y, Zhang G, Kawakami Y, Marincola FM, Ascierto PA, Sargent DJ, Fox BA, Galon J (2018) International validation of the consensus Immunoscore for the classification of colon cancer: a prognostic and accuracy study. Lancet 391(10135):2128–2139. https://doi.org/10.1016/S0140-6736(18)30789-X
Munn DH, Mellor AL (2013) Indoleamine 2,3 dioxygenase and metabolic control of immune responses. Trends Immunol 34(3):137–143. https://doi.org/10.1016/j.it.2012.10.001
Munn DH, Shafizadeh E, Attwood JT, Bondarev I, Pashine A, Mellor AL (1999) Inhibition of T cell proliferation by macrophage tryptophan catabolism. J Exp Med 189(9):1363–1372
Munn DH, Sharma MD, Baban B, Harding HP, Zhang Y, Ron D, Mellor AL (2005) GCN2 kinase in T cells mediates proliferative arrest and anergy induction in response to indoleamine 2,3-dioxygenase. Immunity 22(5):633–642. https://doi.org/10.1016/j.immuni.2005.03.013
Metz R, Rust S, Duhadaway JB, Mautino MR, Munn DH, Vahanian NN, Link CJ, Prendergast GC (2012) IDO inhibits a tryptophan sufficiency signal that stimulates mTOR: a novel IDO effector pathway targeted by D-1-methyl-tryptophan. Oncoimmunology 1(9):1460–1468. https://doi.org/10.4161/onci.21716
Fallarino F, Grohmann U, Vacca C, Bianchi R, Orabona C, Spreca A, Fioretti MC, Puccetti P (2002) T cell apoptosis by tryptophan catabolism. Cell Death Differ 9(10):1069–1077. https://doi.org/10.1038/sj.cdd.4401073
Terness P, Bauer TM, Rose L, Dufter C, Watzlik A, Simon H, Opelz G (2002) Inhibition of allogeneic T cell proliferation by indoleamine 2,3-dioxygenase-expressing dendritic cells: mediation of suppression by tryptophan metabolites. J Exp Med 196(4):447–457
Fallarino F, Grohmann U, You S, McGrath BC, Cavener DR, Vacca C, Orabona C, Bianchi R, Belladonna ML, Volpi C, Santamaria P, Fioretti MC, Puccetti P (2006) The combined effects of tryptophan starvation and tryptophan catabolites down-regulate T cell receptor zeta-chain and induce a regulatory phenotype in naive T cells. J Immunol 176(11):6752–6761
Mezrich JD, Fechner JH, Zhang X, Johnson BP, Burlingham WJ, Bradfield CA (2010) An interaction between kynurenine and the aryl hydrocarbon receptor can generate regulatory T cells. J Immunol 185(6):3190–3198. https://doi.org/10.4049/jimmunol.0903670
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674. https://doi.org/10.1016/j.cell.2011.02.013
van Baren N, Van den Eynde BJ (2015) Tumoral immune resistance mediated by enzymes that degrade tryptophan. Cancer Immunol Res 3(9):978–985. https://doi.org/10.1158/2326-6066.CIR-15-0095
Brandacher G, Perathoner A, Ladurner R, Schneeberger S, Obrist P, Winkler C, Werner ER, Werner-Felmayer G, Weiss HG, Gobel G, Margreiter R, Konigsrainer A, Fuchs D, Amberger A (2006) Prognostic value of indoleamine 2,3-dioxygenase expression in colorectal cancer: effect on tumor-infiltrating T cells. Clin Cancer Res 12(4):1144–1151. https://doi.org/10.1158/1078-0432.CCR-05-1966
Ferdinande L, Decaestecker C, Verset L, Mathieu A, Moles Lopez X, Negulescu AM, Van Maerken T, Salmon I, Cuvelier CA, Demetter P (2012) Clinicopathological significance of indoleamine 2,3-dioxygenase 1 expression in colorectal cancer. Br J Cancer 106(1):141–147. https://doi.org/10.1038/bjc.2011.513
Gao YF, Peng RQ, Li J, Ding Y, Zhang X, Wu XJ, Pan ZZ, Wan DS, Zeng YX, Zhang XS (2009) The paradoxical patterns of expression of indoleamine 2,3-dioxygenase in colon cancer. J Transl Med 7:71. https://doi.org/10.1186/1479-5876-7-71
Godin-Ethier J, Hanafi LA, Piccirillo CA, Lapointe R (2011) Indoleamine 2,3-dioxygenase expression in human cancers: clinical and immunologic perspectives. Clin Cancer Res 17(22):6985–6991. https://doi.org/10.1158/1078-0432.CCR-11-1331
Schmiegel W, Buchberger B, Follmann M, Graeven U, Heinemann V, Langer T, Nothacker M, Porschen R, Rodel C, Rosch T, Schmitt W, Wesselmann S, Pox C (2017) S3-Leitlinie Kolorektales Karzinom. Z Gastroenterol 55(12):1344–1498. https://doi.org/10.1055/s-0043-121106
Theate I, van Baren N, Pilotte L, Moulin P, Larrieu P, Renauld JC, Herve C, Gutierrez-Roelens I, Marbaix E, Sempoux C, Van den Eynde BJ (2015) Extensive profiling of the expression of the indoleamine 2,3-dioxygenase 1 protein in normal and tumoral human tissues. Cancer Immunol Res 3(2):161–172. https://doi.org/10.1158/2326-6066.CIR-14-0137
Dahlin AM, Henriksson ML, Van Guelpen B, Stenling R, Oberg A, Rutegard J, Palmqvist R (2011) Colorectal cancer prognosis depends on T-cell infiltration and molecular characteristics of the tumor. Mod Pathol 24(5):671–682. https://doi.org/10.1038/modpathol.2010.234
Ling A, Edin S, Wikberg ML, Oberg A, Palmqvist R (2014) The intratumoural subsite and relation of CD8(+) and FOXP3(+) T lymphocytes in colorectal cancer provide important prognostic clues. Br J Cancer 110(10):2551–2559. https://doi.org/10.1038/bjc.2014.161
Ogino S, Nosho K, Irahara N, Meyerhardt JA, Baba Y, Shima K, Glickman JN, Ferrone CR, Mino-Kenudson M, Tanaka N, Dranoff G, Giovannucci EL, Fuchs CS (2009) Lymphocytic reaction to colorectal cancer is associated with longer survival, independent of lymph node count, microsatellite instability, and CpG island methylator phenotype. Clin Cancer Res 15(20):6412–6420. https://doi.org/10.1158/1078-0432.CCR-09-1438
Pages F, Berger A, Camus M, Sanchez-Cabo F, Costes A, Molidor R, Mlecnik B, Kirilovsky A, Nilsson M, Damotte D, Meatchi T, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Galon J (2005) Effector memory T cells, early metastasis, and survival in colorectal cancer. N Engl J Med 353(25):2654–2666. https://doi.org/10.1056/NEJMoa051424
Galon J, Mlecnik B, Bindea G, Angell HK, Berger A, Lagorce C, Lugli A, Zlobec I, Hartmann A, Bifulco C, Nagtegaal ID, Palmqvist R, Masucci GV, Botti G, Tatangelo F, Delrio P, Maio M, Laghi L, Grizzi F, Asslaber M, D’Arrigo C, Vidal-Vanaclocha F, Zavadova E, Chouchane L, Ohashi PS, Hafezi-Bakhtiari S, Wouters BG, Roehrl M, Nguyen L, Kawakami Y, Hazama S, Okuno K, Ogino S, Gibbs P, Waring P, Sato N, Torigoe T, Itoh K, Patel PS, Shukla SN, Wang Y, Kopetz S, Sinicrope FA, Scripcariu V, Ascierto PA, Marincola FM, Fox BA, Pages F (2014) Towards the introduction of the ‘Immunoscore’ in the classification of malignant tumours. J Pathol 232(2):199–209. https://doi.org/10.1002/path.4287
Galon J, Pages F, Marincola FM, Thurin M, Trinchieri G, Fox BA, Gajewski TF, Ascierto PA (2012) The immune score as a new possible approach for the classification of cancer. J Transl Med 10:1. https://doi.org/10.1186/1479-5876-10-1
Church SE, Galon J (2017) Regulation of CTL Infiltration Within the Tumor Microenvironment. Adv Exp Med Biol 1036:33–49. https://doi.org/10.1007/978-3-319-67577-0_3
Lob S, Konigsrainer A, Schafer R, Rammensee HG, Opelz G, Terness P (2008) Levo- but not dextro-1-methyl tryptophan abrogates the IDO activity of human dendritic cells. Blood 111(4):2152–2154. https://doi.org/10.1182/blood-2007-10-116111
Lob S, Konigsrainer A, Zieker D, Brucher BL, Rammensee HG, Opelz G, Terness P (2009) IDO1 and IDO2 are expressed in human tumors: levo- but not dextro-1-methyl tryptophan inhibits tryptophan catabolism. Cancer Immunol Immunother 58(1):153–157. https://doi.org/10.1007/s00262-008-0513-6
Naito Y, Saito K, Shiiba K, Ohuchi A, Saigenji K, Nagura H, Ohtani H (1998) CD8 + T cells infiltrated within cancer cell nests as a prognostic factor in human colorectal cancer. Cancer Res 58(16):3491–3494
Coulie PG, Van den Eynde BJ, van der Bruggen P, Boon T (2014) Tumour antigens recognized by T lymphocytes: at the core of cancer immunotherapy. Nat Rev Cancer 14(2):135–146. https://doi.org/10.1038/nrc3670
Galluzzi L, Buque A, Kepp O, Zitvogel L, Kroemer G (2015) Immunological Effects of Conventional Chemotherapy and Targeted Anticancer Agents. Cancer Cell 28(6):690–714. https://doi.org/10.1016/j.ccell.2015.10.012
Frey B, Ruckert M, Deloch L, Ruhle PF, Derer A, Fietkau R, Gaipl US (2017) Immunomodulation by ionizing radiation-impact for design of radio-immunotherapies and for treatment of inflammatory diseases. Immunol Rev 280(1):231–248. https://doi.org/10.1111/imr.12572
Munn DH, Zhou M, Attwood JT, Bondarev I, Conway SJ, Marshall B, Brown C, Mellor AL (1998) Prevention of allogeneic fetal rejection by tryptophan catabolism. Science 281(5380):1191–1193
Ravishankar B, Liu H, Shinde R, Chandler P, Baban B, Tanaka M, Munn DH, Mellor AL, Karlsson MC, McGaha TL (2012) Tolerance to apoptotic cells is regulated by indoleamine 2,3-dioxygenase. Proc Natl Acad Sci U S A 109(10):3909–3914. https://doi.org/10.1073/pnas.1117736109
Uyttenhove C, Pilotte L, Theate I, Stroobant V, Colau D, Parmentier N, Boon T, Van den Eynde BJ (2003) Evidence for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine 2,3-dioxygenase. Nat Med 9(10):1269–1274. https://doi.org/10.1038/nm934
Baban B, Chandler P, McCool D, Marshall B, Munn DH, Mellor AL (2004) Indoleamine 2,3-dioxygenase expression is restricted to fetal trophoblast giant cells during murine gestation and is maternal genome specific. J Reprod Immunol 61(2):67–77. https://doi.org/10.1016/j.jri.2003.11.003
de Faudeur G, de Trez C, Muraille E, Leo O (2008) Normal development and function of dendritic cells in mice lacking IDO-1 expression. Immunol Lett 118(1):21–29. https://doi.org/10.1016/j.imlet.2008.02.006
Lob S, Konigsrainer A, Rammensee HG, Opelz G, Terness P (2009) Inhibitors of indoleamine-2,3-dioxygenase for cancer therapy: can we see the wood for the trees? Nat Rev Cancer 9(6):445–452. https://doi.org/10.1038/nrc2639
Monjazeb AM, Kent MS, Grossenbacher SK, Mall C, Zamora AE, Mirsoian A, Chen M, Kol A, Shiao SL, Reddy A, Perks JR, W TNC, Sparger EE, Canter RJ, Sckisel GD, Murphy WJ (2016) Blocking indolamine-2,3-dioxygenase rebound immune suppression boosts antitumor effects of radio-immunotherapy in murine models and spontaneous canine malignancies. Clin Cancer Res 22(17):4328–4340. https://doi.org/10.1158/1078-0432.CCR-15-3026
Spranger S, Spaapen RM, Zha Y, Williams J, Meng Y, Ha TT, Gajewski TF (2013) Up-regulation of PD-L1, IDO, and T(regs) in the melanoma tumor microenvironment is driven by CD8(+) T cells. Sci Transl Med 5(200):200ra116. https://doi.org/10.1126/scitranslmed.3006504
Gajewski TF, Louahed J, Brichard VG (2010) Gene signature in melanoma associated with clinical activity: a potential clue to unlock cancer immunotherapy. Cancer J 16(4):399–403. https://doi.org/10.1097/PPO.0b013e3181eacbd8
Netter J, Douard R, Durdux C, Landi B, Berger A, Taieb J (2016) Advances in management of adjuvant chemotherapy in rectal cancer: Consequences for clinical practice. Clin Res Hepatol Gastroenterol 40(5):546–552. https://doi.org/10.1016/j.clinre.2016.03.004
Sorensen RB, Berge-Hansen L, Junker N, Hansen CA, Hadrup SR, Schumacher TN, Svane IM, Becker JC, thor Straten P, Andersen MH (2009) The immune system strikes back: cellular immune responses against indoleamine 2,3-dioxygenase. PLoS One 4(9):e6910. https://doi.org/10.1371/journal.pone.0006910
Sorensen RB, Hadrup SR, Svane IM, Hjortso MC, Thor Straten P, Andersen MH (2011) Indoleamine 2,3-dioxygenase specific, cytotoxic T cells as immune regulators. Blood 117(7):2200–2210. https://doi.org/10.1182/blood-2010-06-288498
Andersen MH, Svane IM (2015) Indoleamine 2,3-dioxygenase vaccination. Oncoimmunology 4(1):e983770. https://doi.org/10.4161/2162402X.2014.983770
Iversen TZ, Engell-Noerregaard L, Ellebaek E, Andersen R, Larsen SK, Bjoern J, Zeyher C, Gouttefangeas C, Thomsen BM, Holm B, Thor Straten P, Mellemgaard A, Andersen MH, Svane IM (2014) Long-lasting disease stabilization in the absence of toxicity in metastatic lung cancer patients vaccinated with an epitope derived from indoleamine 2,3 dioxygenase. Clin Cancer Res 20(1):221–232. https://doi.org/10.1158/1078-0432.CCR-13-1560
Vacchelli E, Aranda F, Eggermont A, Sautes-Fridman C, Tartour E, Kennedy EP, Platten M, Zitvogel L, Kroemer G, Galluzzi L (2014) Trial watch: IDO inhibitors in cancer therapy. Oncoimmunology 3(10):e957994. https://doi.org/10.4161/21624011.2014.957994
Prendergast GC, Mondal A, Dey S, Laury-Kleintop LD, Muller AJ (2018) Inflammatory Reprogramming with IDO1 Inhibitors: Turning Immunologically Unresponsive ‘Cold’ Tumors ‘Hot’. Trends Cancer 4(1):38–58. https://doi.org/10.1016/j.trecan.2017.11.005
Muller AJ, Manfredi MG, Zakharia Y, Prendergast GC (2018) Inhibiting IDO pathways to treat cancer: lessons from the ECHO-301 trial and beyond. Semin Immunopathol. https://doi.org/10.1007/s00281-018-0702-0
Companies Scaling Back IDO1 Inhibitor Trials (2018) Cancer Discov 8(7):OF5. https://doi.org/10.1158/2159-8290.CD-ND2018-007
Acknowledgements
The authors would like to thank Sabine Roth for her tireless efforts and qualified support in the set-up of IDO1 immunohistochemistry. We thank Mohammed Hankir for proofreading the manuscript.
Funding
No relevant funding.
Author information
Authors and Affiliations
Contributions
Julia Schollbach: performed immunohistochemistry, manuscript writing. Stefan Kircher: performed immunohistochemistry. Armin Wiegering: study design, manuscript writing. Florian Seyfried: study design, manuscript writing. Ingo Klein: study design. Andreas Rosenwald: study design. Christoph-Thomas Germer: manuscript writing. Stefan Löb: study design, manuscript writing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Patient and treatment characteristics were retrospectively collected from patient records and the Würzburg comprehensive cancer registry. All procedures performed were in accordance with the standards of the institutional ethical committee and with the 1964 Helsinki declaration and its later amendments. Approval of the study was obtained from the Ethics Committee, Medical Faculty, University of Würzburg, Germany (reference number 34/16). All patients provided informed written consent to the use of their anonymized data in scientific studies.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Schollbach, J., Kircher, S., Wiegering, A. et al. Prognostic value of tumour-infiltrating CD8+ lymphocytes in rectal cancer after neoadjuvant chemoradiation: is indoleamine-2,3-dioxygenase (IDO1) a friend or foe?. Cancer Immunol Immunother 68, 563–575 (2019). https://doi.org/10.1007/s00262-019-02306-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-019-02306-y