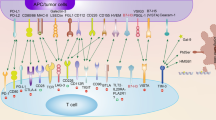
Abstract
The modulation and suppression of anti-tumor immune responses is a characteristic feature of tumor cells to escape immune surveillance. Members of the B7 family are involved in this process, since the level of activation of the anti-tumor immune response depends on the balance between co-stimulatory and co-inhibitory signals. Some molecules are often overexpressed in tumors, which has been associated with the pathogenesis and progression of malignancies as well as their immunological and non-immunological functions. The B7 homologs play a key role in the maintenance of self-tolerance and the regulation of both innate and adaptive immunity in tumor-bearing hosts. Furthermore, the blockade of negative signals mediated by the interaction of co-inhibitory ligands and counter-receptors of the B7 family is currently being studied as a potential immunotherapeutic strategy for the treatment of cancer in humans.


Similar content being viewed by others
Abbreviations
- ALL:
-
Acute lymphoblastic leukemia
- BTLA:
-
B and T lymphocyte attenuator
- CTL:
-
Cytotoxic T lymphocyte
- CTLA4:
-
Cytotoxic T lymphocyte-associated antigen 4
- CR:
-
Complete remission/response
- CTL:
-
Cytotoxic T lymphocyte
- HLA:
-
Human leukocyte antigen
- ICOS:
-
Inducible co-stimulatory molecule
- IFN:
-
Interferon
- JAK:
-
Janus kinase
- mAb:
-
Monoclonal antibody
- MAPK:
-
Mitogen-activated protein kinase
- MDS:
-
Myelodysplastic syndrome
- MDSC:
-
Myeloid-derived suppressor cell
- MHC:
-
Major histocompatibility complex
- MZL:
-
Marginal zone lymphoma
- NK:
-
Natural killer cell
- NSCLC:
-
Non-small cell lung cancer
- OR:
-
Objective response
- PR:
-
Partial response
- PTEN:
-
Phosphatase and tensin homolog
- RCC:
-
Renal cell carcinoma
- STAT:
-
Signal transducer and activator of transcription
- T-ALL:
-
T cell lymphoblastic leukemia
- TAM:
-
Tumor-associated macrophage
- TCR:
-
T cell receptor
- TLR:
-
Toll-like receptor
- TNF:
-
Tumor necrosis factor
- Treg:
-
Regulatory T cell
- SD:
-
Stable disease
References
Carreno BM, Collins M (2002) The B7 family of ligands and its receptors: new pathways for costimulation and inhibition of immune responses. Annu Rev Immunol 20:29–53
Yoshinaga SK, Whoriskey JS, Khare SD, Sarmiento U, Guo J, Horan T, Shih G, Zhang M, Coccia MA, Kohno T, Tafuri-Bladt A, Brankow D, Campbell P, Chang D, Chiu L, Dai T, Duncan G, Elliott GS, Hui A, McCabe SM, Scully S, Shahinian A, Shaklee CL, Van G, Mak TW, Senaldi G (1999) T-cell co-stimulation through B7RP-1 and ICOS. Nature 402(6763):827–832
Dong H, Zhu G, Tamada K, Chen L (1999) B7–H1, a third member of the B7 family, co-stimulates T-cell proliferation and interleukin-10 secretion. Nat Med 5(12):1365–1369
Latchman Y, Wood CR, Chernova T, Chaudhary D, Borde M, Chernova I, Iwai Y, Long AJ, Brown JA, Nunes R, Greenfield EA, Bourque K, Boussiotis VA, Carter LL, Carreno BM, Malenkovich N, Nishimura H, Okazaki T, Honjo T, Sharpe AH, Freeman GJ (2001) PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat Immunol 2(3):261–268
Kaifu T, Escaliere B, Gastinel LN, Vivier E, Baratin M (2011) B7-H6/NKp30 interaction: a mechanism of alerting NK cells against tumors. Cell Mol Life Sci 68(21):3531–3539
Brandt CS, Baratin M, Yi EC, Kennedy J, Gao Z, Fox B, Haldeman B, Ostrander CD, Kaifu T, Chabannon C, Moretta A, West R, Xu W, Vivier E, Levin SD (2009) The B7 family member B7-H6 is a tumor cell ligand for the activating natural killer cell receptor NKp30 in humans. J Exp Med 206(7):1495–1503
Chapoval AI, Ni J, Lau JS, Wilcox RA, Flies DB, Liu D, Dong H, Sica GL, Zhu G, Tamada K, Chen L (2001) B7-H3: a costimulatory molecule for T cell activation and IFN-gamma production. Nat Immunol 2(3):269–274
Sica GL, Choi IH, Zhu G, Tamada K, Wang SD, Tamura H, Chapoval AI, Flies DB, Bajorath J, Chen L (2003) B7–H4, a molecule of the B7 family, negatively regulates T cell immunity. Immunity 18(6):849–861
Kroczek RA, Mages HW, Hutloff A (2004) Emerging paradigms of T-cell co-stimulation. Curr Opin Immunol 16(3):321–327
Jenkins MK, Johnson JG (1993) Molecules involved in T-cell costimulation. Curr Opin Immunol 5(3):361–367
Frauwirth KA, Thompson CB (2002) Activation and inhibition of lymphocytes by costimulation. J Clin Invest 109(3):295–299
Hutloff A, Dittrich AM, Beier KC, Eljaschewitsch B, Kraft R, Anagnostopoulos I, Kroczek RA (1999) ICOS is an inducible T-cell co-stimulator structurally and functionally related to CD28. Nature 397(6716):263–266
Coyle AJ, Lehar S, Lloyd C, Tian J, Delaney T, Manning S, Nguyen T, Burwell T, Schneider H, Gonzalo JA, Gosselin M, Owen LR, Rudd CE, Gutierrez-Ramos JC (2000) The CD28-related molecule ICOS is required for effective T cell-dependent immune responses. Immunity 13(1):95–105
Yao S, Zhu Y, Zhu G, Augustine M, Zheng L, Goode DJ, Broadwater M, Ruff W, Flies S, Xu H, Flies D, Luo L, Wang S, Chen L (2011) B7-h2 is a costimulatory ligand for CD28 in human. Immunity 34(5):729–740
Pulko V, Harris KJ, Liu X, Gibbons RM, Harrington SM, Krco CJ, Kwon ED, Dong H (2011) B7-H1 expressed by activated CD8 T cells is essential for their survival. J Immunol 187(11):5606–5614
Dong H, Strome SE, Salomao DR, Tamura H, Hirano F, Flies DB, Roche PC, Lu J, Zhu G, Tamada K, Lennon VA, Celis E, Chen L (2002) Tumor-associated B7–H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med 8(8):793–800
Suh WK, Gajewska BU, Okada H, Gronski MA, Bertram EM, Dawicki W, Duncan GS, Bukczynski J, Plyte S, Elia A, Wakeham A, Itie A, Chung S, Da Costa J, Arya S, Horan T, Campbell P, Gaida K, Ohashi PS, Watts TH, Yoshinaga SK, Bray MR, Jordana M, Mak TW (2003) The B7 family member B7–H3 preferentially down-regulates T helper type 1-mediated immune responses. Nat Immunol 4(9):899–906
Leitner J, Klauser C, Pickl WF, Stockl J, Majdic O, Bardet AF, Kreil DP, Dong C, Yamazaki T, Zlabinger G, Pfistershammer K, Steinberger P (2009) B7–H3 is a potent inhibitor of human T-cell activation: no evidence for B7–H3 and TREML2 interaction. Eur J Immunol 39(7):1754–1764
Choi IH, Zhu G, Sica GL, Strome SE, Cheville JC, Lau JS, Zhu Y, Flies DB, Tamada K, Chen L (2003) Genomic organization and expression analysis of B7–H4, an immune inhibitory molecule of the B7 family. J Immunol 171(9):4650–4654
Kryczek I, Zou L, Rodriguez P, Zhu G, Wei S, Mottram P, Brumlik M, Cheng P, Curiel T, Myers L, Lackner A, Alvarez X, Ochoa A, Chen L, Zou W (2006) B7–H4 expression identifies a novel suppressive macrophage population in human ovarian carcinoma. J Exp Med 203(4):871–881
Cheng C, Qu QX, Shen Y, Lv YT, Zhu YB, Zhang XG, Huang JA (2011) Overexpression of B7–H4 in tumor infiltrated dendritic cells. J Immunoassay Immunochem 32(4):353–364
Watanabe N, Gavrieli M, Sedy JR, Yang J, Fallarino F, Loftin SK, Hurchla MA, Zimmerman N, Sim J, Zang X, Murphy TL, Russell JH, Allison JP, Murphy KM (2003) BTLA is a lymphocyte inhibitory receptor with similarities to CTLA-4 and PD-1. Nat Immunol 4(7):670–679
Compaan DM, Gonzalez LC, Tom I, Loyet KM, Eaton D, Hymowitz SG (2005) Attenuating lymphocyte activity: the crystal structure of the BTLA–HVEM complex. J Biol Chem 280(47):39553–39561
Frigola X, Inman BA, Krco CJ, Liu X, Harrington SM, Bulur PA, Dietz AB, Dong H, Kwon ED (2012) Soluble B7–H1: differences in production between dendritic cells and T cells. Immunol Lett 142(1–2):78–82
Her M, Kim D, Oh M, Jeong H, Choi I (2009) Increased expression of soluble inducible costimulator ligand (ICOSL) in patients with systemic lupus erythematosus. Lupus 18(6):501–507
Luan Y, Ju J, Luo L, Zhang Z, Wang J, Zhu DM, Cheng L, Zhang SY, Chen L, Wang FS, Wang S (2012) Potential role of soluble B7–H3 in liver immunopathogenesis during chronic HBV infection. J Viral Hepat 19(1):23–31
Zhang G, Hou J, Shi J, Yu G, Lu B, Zhang X (2008) Soluble CD276 (B7–H3) is released from monocytes, dendritic cells and activated T cells and is detectable in normal human serum. Immunology 123(4):538–546
Azuma T, Zhu G, Xu H, Rietz AC, Drake CG, Matteson EL, Chen L (2009) Potential role of decoy B7–H4 in the pathogenesis of rheumatoid arthritis: a mouse model informed by clinical data. PLoS Med 6(10):e1000166
Zou W, Chen L (2008) Inhibitory B7-family molecules in the tumour microenvironment. Nat Rev Immunol 8(6):467–477
Ghebeh H, Tulbah A, Mohammed S, Elkum N, Bin Amer SM, Al-Tweigeri T, Dermime S (2007) Expression of B7–H1 in breast cancer patients is strongly associated with high proliferative Ki-67-expressing tumor cells. Int J Cancer 121(4):751–758
Nakanishi J, Wada Y, Matsumoto K, Azuma M, Kikuchi K, Ueda S (2007) Overexpression of B7–H1 (PD-L1) significantly associates with tumor grade and postoperative prognosis in human urothelial cancers. Cancer Immunol Immunother 56(8):1173–1182
Konishi J, Yamazaki K, Azuma M, Kinoshita I, Dosaka-Akita H, Nishimura M (2004) B7–H1 expression on non-small cell lung cancer cells and its relationship with tumor-infiltrating lymphocytes and their PD-1 expression. Clin Cancer Res 10(15):5094–5100
Geng L, Huang D, Liu J, Qian Y, Deng J, Li D, Hu Z, Zhang J, Jiang G, Zheng S (2008) B7–H1 up-regulated expression in human pancreatic carcinoma tissue associates with tumor progression. J Cancer Res Clin Oncol 134(9):1021–1027
Wu C, Zhu Y, Jiang J, Zhao J, Zhang XG, Xu N (2006) Immunohistochemical localization of programmed death-1 ligand-1 (PD-L1) in gastric carcinoma and its clinical significance. Acta Histochem 108(1):19–24
Hino R, Kabashima K, Kato Y, Yagi H, Nakamura M, Honjo T, Okazaki T, Tokura Y (2010) Tumor cell expression of programmed cell death-1 ligand 1 is a prognostic factor for malignant melanoma. Cancer 116(7):1757–1766
Ohigashi Y, Sho M, Yamada Y, Tsurui Y, Hamada K, Ikeda N, Mizuno T, Yoriki R, Kashizuka H, Yane K, Tsushima F, Otsuki N, Yagita H, Azuma M, Nakajima Y (2005) Clinical significance of programmed death-1 ligand-1 and programmed death-1 ligand-2 expression in human esophageal cancer. Clin Cancer Res 11(8):2947–2953
Geng L, Deng J, Jiang G, Song P, Wang Z, Jiang Z, Zhang M, Zheng S (2011) B7-H1 up-regulated expression in human hepatocellular carcinoma tissue: correlation with tumor interleukin-10 levels. Hepatogastroenterology 58(107–108):960–964
Hamanishi J, Mandai M, Iwasaki M, Okazaki T, Tanaka Y, Yamaguchi K, Higuchi T, Yagi H, Takakura K, Minato N, Honjo T, Fujii S (2007) Programmed cell death 1 ligand 1 and tumor-infiltrating CD8 + T lymphocytes are prognostic factors of human ovarian cancer. Proc Natl Acad Sci USA 104(9):3360–3365
Parsa AT, Waldron JS, Panner A, Crane CA, Parney IF, Barry JJ, Cachola KE, Murray JC, Tihan T, Jensen MC, Mischel PS, Stokoe D, Pieper RO (2007) Loss of tumor suppressor PTEN function increases B7–H1 expression and immunoresistance in glioma. Nat Med 13(1):84–88
Yao Y, Tao R, Wang X, Wang Y, Mao Y, Zhou LF (2009) B7–H1 is correlated with malignancy-grade gliomas but is not expressed exclusively on tumor stem-like cells. Neuro Oncol 11(6):757–766
Thompson RH, Kuntz SM, Leibovich BC, Dong H, Lohse CM, Webster WS, Sengupta S, Frank I, Parker AS, Zincke H, Blute ML, Sebo TJ, Cheville JC, Kwon ED (2006) Tumor B7-H1 is associated with poor prognosis in renal cell carcinoma patients with long-term follow-up. Cancer Res 66(7):3381–3385
Yamamoto R, Nishikori M, Tashima M, Sakai T, Ichinohe T, Takaori-Kondo A, Ohmori K, Uchiyama T (2009) B7–H1 expression is regulated by MEK/ERK signaling pathway in anaplastic large cell lymphoma and Hodgkin lymphoma. Cancer Sci 100(11):2093–2100
Berthon C, Driss V, Liu J, Kuranda K, Leleu X, Jouy N, Hetuin D, Quesnel B (2010) In acute myeloid leukemia, B7–H1 (PD-L1) protection of blasts from cytotoxic T cells is induced by TLR ligands and interferon-gamma and can be reversed using MEK inhibitors. Cancer Immunol Immunother 59(12):1839–1849
Kondo A, Yamashita T, Tamura H, Zhao W, Tsuji T, Shimizu M, Shinya E, Takahashi H, Tamada K, Chen L, Dan K, Ogata K (2010) Interferon-gamma and tumor necrosis factor-alpha induce an immunoinhibitory molecule, B7–H1, via nuclear factor-kappaB activation in blasts in myelodysplastic syndromes. Blood 116(7):1124–1131
Tamura H, Dan K, Tamada K, Nakamura K, Shioi Y, Hyodo H, Wang SD, Dong H, Chen L, Ogata K (2005) Expression of functional B7-H2 and B7.2 costimulatory molecules and their prognostic implications in de novo acute myeloid leukemia. Clin Cancer Res 11(16):5708–5717
Loos M, Hedderich DM, Friess H, Kleeff J (2010) B7–h3 and its role in antitumor immunity. Clin Dev Immunol 2010:683875
Crispen PL, Sheinin Y, Roth TJ, Lohse CM, Kuntz SM, Frigola X, Thompson RH, Boorjian SA, Dong H, Leibovich BC, Blute ML, Kwon ED (2008) Tumor cell and tumor vasculature expression of B7-H3 predict survival in clear cell renal cell carcinoma. Clin Cancer Res 14(16):5150–5157
Quandt D, Fiedler E, Boettcher D, Marsch W, Seliger B (2011) B7-H4 expression in human melanoma: its association with patients’ survival and antitumor immune response. Clin Cancer Res 17(10):3100–3111
Sun Y, Wang Y, Zhao J, Gu M, Giscombe R, Lefvert AK, Wang X (2006) B7–H3 and B7–H4 expression in non-small-cell lung cancer. Lung Cancer 53(2):143–151
Zang X, Thompson RH, Al-Ahmadie HA, Serio AM, Reuter VE, Eastham JA, Scardino PT, Sharma P, Allison JP (2007) B7–H3 and B7x are highly expressed in human prostate cancer and associated with disease spread and poor outcome. Proc Natl Acad Sci USA 104(49):19458–19463
Tringler B, Liu W, Corral L, Torkko KC, Enomoto T, Davidson S, Lucia MS, Heinz DE, Papkoff J, Shroyer KR (2006) B7–H4 overexpression in ovarian tumors. Gynecol Oncol 100(1):44–52
Jiang J, Zhu Y, Wu C, Shen Y, Wei W, Chen L, Zheng X, Sun J, Lu B, Zhang X (2010) Tumor expression of B7–H4 predicts poor survival of patients suffering from gastric cancer. Cancer Immunol Immunother 59(11):1707–1714
Awadallah NS, Shroyer KR, Langer DA, Torkko KC, Chen YK, Bentz JS, Papkoff J, Liu W, Nash SR, Shah RJ (2008) Detection of B7–H4 and p53 in pancreatic cancer: potential role as a cytological diagnostic adjunct. Pancreas 36(2):200–206
Tringler B, Zhuo S, Pilkington G, Torkko KC, Singh M, Lucia MS, Heinz DE, Papkoff J, Shroyer KR (2005) B7–h4 is highly expressed in ductal and lobular breast cancer. Clin Cancer Res 11(5):1842–1848
Chen LJ, Sun J, Wu HY, Zhou SM, Tan Y, Tan M, Shan BE, Lu BF, Zhang XG (2011) B7–H4 expression associates with cancer progression and predicts patient’s survival in human esophageal squamous cell carcinoma. Cancer Immunol Immunother 60(7):1047–1055
Krambeck AE, Thompson RH, Dong H, Lohse CM, Park ES, Kuntz SM, Leibovich BC, Blute ML, Cheville JC, Kwon ED (2006) B7–H4 expression in renal cell carcinoma and tumor vasculature: associations with cancer progression and survival. Proc Natl Acad Sci USA 103(27):10391–10396
Yao Y, Wang X, Jin K, Zhu J, Wang Y, Xiong S, Mao Y, Zhou L (2008) B7–H4 is preferentially expressed in non-dividing brain tumor cells and in a subset of brain tumor stem-like cells. J Neurooncol 89(2):121–129
Frigola X, Inman BA, Lohse CM, Krco CJ, Cheville JC, Thompson RH, Leibovich B, Blute ML, Dong H, Kwon ED (2011) Identification of a soluble form of B7–H1 that retains immunosuppressive activity and is associated with aggressive renal cell carcinoma. Clin Cancer Res 17(7):1915–1923
Thompson RH, Zang X, Lohse CM, Leibovich BC, Slovin SF, Reuter VE, Cheville JC, Blute ML, Russo P, Kwon ED, Allison JP (2008) Serum-soluble B7x is elevated in renal cell carcinoma patients and is associated with advanced stage. Cancer Res 68(15):6054–6058
Zhang G, Xu Y, Lu X, Huang H, Zhou Y, Lu B, Zhang X (2009) Diagnosis value of serum B7–H3 expression in non-small cell lung cancer. Lung Cancer 66(2):245–249
Simon I, Katsaros D, Rigault de la Longrais I, Massobrio M, Scorilas A, Kim NW, Sarno MJ, Wolfert RL, Diamandis EP (2007) B7–H4 is over-expressed in early-stage ovarian cancer and is independent of CA125 expression. Gynecol Oncol 106(2):334–341
Fu T, He Q, Sharma P (2011) The ICOS/ICOSL pathway is required for optimal antitumor responses mediated by anti-CTLA-4 therapy. Cancer Res 71(16):5445–5454
Mu CY, Huang JA, Chen Y, Chen C, Zhang XG (2011) High expression of PD-L1 in lung cancer may contribute to poor prognosis and tumor cells immune escape through suppressing tumor infiltrating dendritic cells maturation. Med Oncol 28(3):682–688
Wang L, Ma Q, Chen X, Guo K, Li J, Zhang M (2010) Clinical significance of B7–H1 and B7–1 expressions in pancreatic carcinoma. World J Surg 34(5):1059–1065
Thompson RH, Gillett MD, Cheville JC, Lohse CM, Dong H, Webster WS, Chen L, Zincke H, Blute ML, Leibovich BC, Kwon ED (2005) Costimulatory molecule B7–H1 in primary and metastatic clear cell renal cell carcinoma. Cancer 104(10):2084–2091
Gadiot J, Hooijkaas AI, Kaiser AD, van Tinteren H, van Boven H, Blank C (2011) Overall survival and PD-L1 expression in metastasized malignant melanoma. Cancer 117(10):2192–2201
Inman BA, Sebo TJ, Frigola X, Dong H, Bergstralh EJ, Frank I, Fradet Y, Lacombe L, Kwon ED (2007) PD-L1 (B7–H1) expression by urothelial carcinoma of the bladder and BCG-induced granulomata: associations with localized stage progression. Cancer 109(8):1499–1505
Wu CP, Jiang JT, Tan M, Zhu YB, Ji M, Xu KF, Zhao JM, Zhang GB, Zhang XG (2006) Relationship between co-stimulatory molecule B7–H3 expression and gastric carcinoma histology and prognosis. World J Gastroenterol 12(3):457–459
Loos M, Hedderich DM, Ottenhausen M, Giese NA, Laschinger M, Esposito I, Kleeff J, Friess H (2009) Expression of the costimulatory molecule B7–H3 is associated with prolonged survival in human pancreatic cancer. BMC Cancer 9:463
Roth TJ, Sheinin Y, Lohse CM, Kuntz SM, Frigola X, Inman BA, Krambeck AE, McKenney ME, Karnes RJ, Blute ML, Cheville JC, Sebo TJ, Kwon ED (2007) B7–H3 ligand expression by prostate cancer: a novel marker of prognosis and potential target for therapy. Cancer Res 67(16):7893–7900
Brunner A, Hinterholzer S, Riss P, Heinze G, Brustmann H (2012) Immunoexpression of B7–H3 in endometrial cancer: relation to tumor T-cell infiltration and prognosis. Gynecol Oncol 124(1):105–111
He C, Qiao H, Jiang H, Sun X (2011) The inhibitory role of B7-h4 in antitumor immunity: association with cancer progression and survival. Clin Dev Immunol 2011:695834
Simon I, Zhuo S, Corral L, Diamandis EP, Sarno MJ, Wolfert RL, Kim NW (2006) B7–h4 is a novel membrane-bound protein and a candidate serum and tissue biomarker for ovarian cancer. Cancer Res 66(3):1570–1575
Sun T, Hu Z, Shen H, Lin D (2009) Genetic polymorphisms in cytotoxic T-lymphocyte antigen 4 and cancer: the dialectical nature of subtle human immune dysregulation. Cancer Res 69(15):6011–6014
Karabon L, Pawlak E, Tomkiewicz A, Jedynak A, Passowicz-Muszynska E, Zajda K, Jonkisz A, Jankowska R, Krzakowski M, Frydecka I (2011) CTLA-4, CD28, and ICOS gene polymorphism associations with non-small-cell lung cancer. Hum Immunol 72(10):947–954
Piras G, Monne M, Uras A, Palmas A, Murineddu M, Arru L, Bianchi A, Calvisi A, Curreli L, Gaviano E, Lai P, Murgia A, Latte GC, Noli A, Gabbas A (2005) Genetic analysis of the 2q33 region containing CD28-CTLA4-ICOS genes: association with non-Hodgkin’s lymphoma. Br J Haematol 129(6):784–790
Blank C, Gajewski TF, Mackensen A (2005) Interaction of PD-L1 on tumor cells with PD-1 on tumor-specific T cells as a mechanism of immune evasion: implications for tumor immunotherapy. Cancer Immunol Immunother 54(4):307–314
Han SJ, Ahn BJ, Waldron JS, Yang I, Fang S, Crane CA, Pieper RO, Parsa AT (2009) Gamma interferon-mediated superinduction of B7–H1 in PTEN-deficient glioblastoma: a paradoxical mechanism of immune evasion. NeuroReport 20(18):1597–1602
Crane CA, Panner A, Murray JC, Wilson SP, Xu H, Chen L, Simko JP, Waldman FM, Pieper RO, Parsa AT (2009) PI(3) kinase is associated with a mechanism of immunoresistance in breast and prostate cancer. Oncogene 28(2):306–312
Liu J, Hamrouni A, Wolowiec D, Coiteux V, Kuliczkowski K, Hetuin D, Saudemont A, Quesnel B (2007) Plasma cells from multiple myeloma patients express B7–H1 (PD-L1) and increase expression after stimulation with IFN-{gamma} and TLR ligands via a MyD88-, TRAF6-, and MEK-dependent pathway. Blood 110(1):296–304
Wasik MA, Zhang Q, Marzec M, Kasprzycka M, Wang HY, Liu X (2009) Anaplastic lymphoma kinase (ALK)-induced malignancies: novel mechanisms of cell transformation and potential therapeutic approaches. Semin Oncol 36(2 Suppl 1):S27–S35
Scandiuzzi L, Ghosh K, Zang X (2011) T cell costimulation and coinhibition: genetics and disease. Discov Med 12(63):119–128
Yi KH, Chen L (2009) Fine tuning the immune response through B7–H3 and B7–H4. Immunol Rev 229(1):145–151
Marzec M, Zhang Q, Goradia A, Raghunath PN, Liu X, Paessler M, Wang HY, Wysocka M, Cheng M, Ruggeri BA, Wasik MA (2008) Oncogenic kinase NPM/ALK induces through STAT3 expression of immunosuppressive protein CD274 (PD-L1, B7–H1). Proc Natl Acad Sci USA 105(52):20852–20857
Steidl C, Shah SP, Woolcock BW, Rui L, Kawahara M, Farinha P, Johnson NA, Zhao Y, Telenius A, Neriah SB, McPherson A, Meissner B, Okoye UC, Diepstra A, van den Berg A, Sun M, Leung G, Jones SJ, Connors JM, Huntsman DG, Savage KJ, Rimsza LM, Horsman DE, Staudt LM, Steidl U, Marra MA, Gascoyne RD (2011) MHC class II transactivator CIITA is a recurrent gene fusion partner in lymphoid cancers. Nature 471(7338):377–381
Gong AY, Zhou R, Hu G, Li X, Splinter PL, O’Hara SP, LaRusso NF, Soukup GA, Dong H, Chen XM (2009) MicroRNA-513 regulates B7–H1 translation and is involved in IFN-gamma-induced B7–H1 expression in cholangiocytes. J Immunol 182(3):1325–1333
Xu H, Cheung IY, Guo HF, Cheung NK (2009) MicroRNA miR-29 modulates expression of immunoinhibitory molecule B7–H3: potential implications for immune based therapy of human solid tumors. Cancer Res 69(15):6275–6281
Fridman WH, Pages F, Sautes-Fridman C, Galon J (2012) The immune contexture in human tumours: impact on clinical outcome. Nat Rev Cancer 12(4):298–306
Kaiser AD, Schuster K, Gadiot J, Borkner L, Daebritz H, Schmitt C, Andreesen R, Blank C (2012) Reduced tumor-antigen density leads to PD-1/PD-L1-mediated impairment of partially exhausted CD8(+) T cells. Eur J Immunol 42(3):662–671
Allavena P, Mantovani A (2012) Immunology in the clinic review series; focus on cancer: tumour-associated macrophages: undisputed stars of the inflammatory tumour microenvironment. Clin Exp Immunol 167(2):195–205
Chen J, Li G, Meng H, Fan Y, Song Y, Wang S, Zhu F, Guo C, Zhang L, Shi Y (2012) Upregulation of B7–H1 expression is associated with macrophage infiltration in hepatocellular carcinomas. Cancer Immunol Immunother 61(1):101–108
Filipazzi P, Huber V, Rivoltini L (2012) Phenotype, function and clinical implications of myeloid-derived suppressor cells in cancer patients. Cancer Immunol Immunother 61(2):255–263
Shin T, Yoshimura K, Crafton EB, Tsuchiya H, Housseau F, Koseki H, Schulick RD, Chen L, Pardoll DM (2005) In vivo costimulatory role of B7-DC in tuning T helper cell 1 and cytotoxic T lymphocyte responses. J Exp Med 201(10):1531–1541
Shin T, Kennedy G, Gorski K, Tsuchiya H, Koseki H, Azuma M, Yagita H, Chen L, Powell J, Pardoll D, Housseau F (2003) Cooperative B7–1/2 (CD80/CD86) and B7-DC costimulation of CD4+ T cells independent of the PD-1 receptor. J Exp Med 198(1):31–38
Okudaira K, Hokari R, Tsuzuki Y, Okada Y, Komoto S, Watanabe C, Kurihara C, Kawaguchi A, Nagao S, Azuma M, Yagita H, Miura S (2009) Blockade of B7–H1 or B7-DC induces an anti-tumor effect in a mouse pancreatic cancer model. Int J Oncol 35(4):741–749
Benson DM Jr, Bakan CE, Mishra A, Hofmeister CC, Efebera Y, Becknell B, Baiocchi RA, Zhang J, Yu J, Smith MK, Greenfield CN, Porcu P, Devine SM, Rotem-Yehudar R, Lozanski G, Byrd JC, Caligiuri MA (2010) The PD-1/PD-L1 axis modulates the natural killer cell versus multiple myeloma effect: a therapeutic target for CT-011, a novel monoclonal anti-PD-1 antibody. Blood 116(13):2286–2294
Zang X, Sullivan PS, Soslow RA, Waitz R, Reuter VE, Wilton A, Thaler HT, Arul M, Slovin SF, Wei J, Spriggs DR, Dupont J, Allison JP (2010) Tumor associated endothelial expression of B7–H3 predicts survival in ovarian carcinomas. Mod Pathol 23(8):1104–1112
Kryczek I, Wei S, Zhu G, Myers L, Mottram P, Cheng P, Chen L, Coukos G, Zou W (2007) Relationship between B7–H4, regulatory T cells, and patient outcome in human ovarian carcinoma. Cancer Res 67(18):8900–8905
Cameron F, Whiteside G, Perry C (2011) Ipilimumab: first global approval. Drugs 71(8):1093–1104
Peggs KS, Quezada SA, Allison JP (2008) Cell intrinsic mechanisms of T-cell inhibition and application to cancer therapy. Immunol Rev 224:141–165
Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC, Akerley W, van den Eertwegh AJ, Lutzky J, Lorigan P, Vaubel JM, Linette GP, Hogg D, Ottensmeier CH, Lebbe C, Peschel C, Quirt I, Clark JI, Wolchok JD, Weber JS, Tian J, Yellin MJ, Nichol GM, Hoos A, Urba WJ (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363(8):711–723
Small EJ, Tchekmedyian NS, Rini BI, Fong L, Lowy I, Allison JP (2007) A pilot trial of CTLA-4 blockade with human anti-CTLA-4 in patients with hormone-refractory prostate cancer. Clin Cancer Res 13(6):1810–1815
Yang JC, Beck KE, Blansfield JA, Tran KQ, Lowy I, Rosenberg SA (2005) Tumor regression in patients with metastatic renal cancer treated with a monoclonal antibody to CTLA4 (MDX-010). J Clin Oncol 2005 ASCO Annual meeting proceedings vol 23, no. 16S, Part I of II (June 1 Supplement), 2005: 2501
Brahmer JR, Drake CG, Wollner I, Powderly JD, Picus J, Sharfman WH, Stankevich E, Pons A, Salay TM, McMiller TL, Gilson MM, Wang C, Selby M, Taube JM, Anders R, Chen L, Korman AJ, Pardoll DM, Lowy I, Topalian SL (2010) Phase I study of single-agent anti-programmed death-1 (MDX-1106) in refractory solid tumors: safety, clinical activity, pharmacodynamics, and immunologic correlates. J Clin Oncol 28(19):3167–3175
Sznol M (2011) Toxicity and activity of anti-PD-1 in phase 1 clinical trials. American association for cancer research (AACR) 102nd annual meeting. Orlando, FL; April 2–6
McDermott DF Drake CG, Sznol M, Sosman JA, Smith DC, Powderly JD (2011) A phase I study to evalute safety and antitumor activity of biweekly BMS-936558 (Anti-PD-1, MDX-1106/ONO-4538) in patients with RCC and other advanced refractory malignancies (abstract). American Society for Clinical Oncology (ASCO) February 17–19, Orlando, FL. (J Clin Oncol 29:2011): (sippl 7; abstr. 331)
Berger R, Rotem-Yehudar R, Slama G, Landes S, Kneller A, Leiba M, Koren-Michowitz M, Shimoni A, Nagler A (2008) Phase I safety and pharmacokinetic study of CT-011, a humanized antibody interacting with PD-1, in patients with advanced hematologic malignancies. Clin Cancer Res 14(10):3044–3051
Borkner L, Kaiser A, van de Kasteele W, Andreesen R, Mackensen A, Haanen JB, Schumacher TN, Blank C (2010) RNA interference targeting programmed death receptor-1 improves immune functions of tumor-specific T cells. Cancer Immunol Immunother 59(8):1173–1183
Chinnadurai R, Grakoui A (2010) B7–H4 mediates inhibition of T cell responses by activated murine hepatic stellate cells. Hepatology 52(6):2177–2185
Salceda S, Tang T, Kmet M, Munteanu A, Ghosh M, Macina R, Liu W, Pilkington G, Papkoff J (2005) The immunomodulatory protein B7–H4 is overexpressed in breast and ovarian cancers and promotes epithelial cell transformation. Exp Cell Res 306(1):128–141
Kaufman HL, Deraffele G, Mitcham J, Moroziewicz D, Cohen SM, Hurst-Wicker KS, Cheung K, Lee DS, Divito J, Voulo M, Donovan J, Dolan K, Manson K, Panicali D, Wang E, Horig H, Marincola FM (2005) Targeting the local tumor microenvironment with vaccinia virus expressing B7.1 for the treatment of melanoma. J Clin Invest 115(7):1903–1912
Raez LE, Cassileth PA, Schlesselman JJ, Sridhar K, Padmanabhan S, Fisher EZ, Baldie PA, Podack ER (2004) Allogeneic vaccination with a B7.1 HLA-A gene-modified adenocarcinoma cell line in patients with advanced non-small-cell lung cancer. J Clin Oncol 22(14):2800–2807
Prieto PA, Yang JC, Sherry RM, Hughes MS, Kammula US, White DE, Levy CL, Rosenberg SA, Phan GQ (2012) CTLA-4 blockade with ipilimumab: long-term follow-up of 177 patients with metastatic melanoma. Clin Cancer Res 18(7):2039–2047
Taube JM, Anders RA, Young GD, Xu H, Sharma R, McMiller TL, Chen S, Klein AP, Pardoll DM, Topalian SL, Chen L (2012) Colocalization of inflammatory response with b7-h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci Transl Med 4(127):127ra137
Sun J, Chen LJ, Zhang GB, Jiang JT, Zhu M, Tan Y, Wang HT, Lu BF, Zhang XG (2010) Clinical significance and regulation of the costimulatory molecule B7–H3 in human colorectal carcinoma. Cancer Immunol Immunother 59(8):1163–1171
Ghebeh H, Mohammed S, Al-Omair A, Qattan A, Lehe C, Al-Qudaihi G, Elkum N, Alshabanah M, Bin Amer S, Tulbah A, Ajarim D, Al-Tweigeri T, Dermime S (2006) The B7–H1 (PD-L1) T lymphocyte-inhibitory molecule is expressed in breast cancer patients with infiltrating ductal carcinoma: correlation with important high-risk prognostic factors. Neoplasia 8(3):190–198
Thompson RH, Gillett MD, Cheville JC, Lohse CM, Dong H, Webster WS, Krejci KG, Lobo JR, Sengupta S, Chen L, Zincke H, Blute ML, Strome SE, Leibovich BC, Kwon ED (2004) Costimulatory B7–H1 in renal cell carcinoma patients: indicator of tumor aggressiveness and potential therapeutic target. Proc Natl Acad Sci USA 101(49):17174–17179
Arigami T, Uenosono Y, Hirata M, Yanagita S, Ishigami S, Natsugoe S (2011) B7–H3 expression in gastric cancer: a novel molecular blood marker for detecting circulating tumor cells. Cancer Sci 102(5):1019–1024
Ribas A, Camacho LH, Lopez-Berestein G, Pavlov D, Bulanhagui CA, Millham R, Comin-Anduix B, Reuben JM, Seja E, Parker CA, Sharma A, Glaspy JA, Gomez-Navarro J (2005) Antitumor activity in melanoma and anti-self responses in a phase I trial with the anti-cytotoxic T lymphocyte-associated antigen 4 monoclonal antibody CP-675,206. J Clin Oncol 23(35):8968–8977
Hodi FS, Butler M, Oble DA, Seiden MV, Haluska FG, Kruse A, Macrae S, Nelson M, Canning C, Lowy I, Korman A, Lautz D, Russell S, Jaklitsch MT, Ramaiya N, Chen TC, Neuberg D, Allison JP, Mihm MC, Dranoff G (2008) Immunologic and clinical effects of antibody blockade of cytotoxic T lymphocyte-associated antigen 4 in previously vaccinated cancer patients. Proc Natl Acad Sci USA 105(8):3005–3010
Hodi FS, Mihm MC, Soiffer RJ, Haluska FG, Butler M, Seiden MV, Davis T, Henry-Spires R, MacRae S, Willman A, Padera R, Jaklitsch MT, Shankar S, Chen TC, Korman A, Allison JP, Dranoff G (2003) Biologic activity of cytotoxic T lymphocyte-associated antigen 4 antibody blockade in previously vaccinated metastatic melanoma and ovarian carcinoma patients. Proc Natl Acad Sci USA 100(8):4712–4717
Acknowledgments
We would like to thank Sylvi Magdeburg for secretarial help and acknowledge the Wilhelm Roux Program of the Martin Luther University Halle-Wittenberg (D.Q.) and a grant from the Mildred Scheel Foundation, Bonn (B.S., D.Q.).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper is a Focussed Research Review based on a presentation given at the Eleventh International Conference on Progress in Vaccination against Cancer (PIVAC 11), held in Copenhagen, Denmark, 10–13 October 2011. It is part of a CII series of Focussed Research Reviews and meeting report.
Rights and permissions
About this article
Cite this article
Seliger, B., Quandt, D. The expression, function, and clinical relevance of B7 family members in cancer. Cancer Immunol Immunother 61, 1327–1341 (2012). https://doi.org/10.1007/s00262-012-1293-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-012-1293-6