Abstract
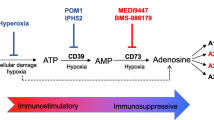
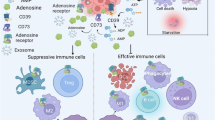
The A2A adenosine receptor plays a critical and non-redundant role in suppressing inflammation at sites of hypoxia and tissue damage. The tumor microenvironment has high levels of adenosine as a result of hypoxia and ectopic expression of enzymes responsible for the generation of extracellular adenosine. Thus, we sought to determine the ability of A2A receptor null mice to immunologically reject tumors. We observed that mice lacking the A2A adenosine receptor showed significantly delayed growth of lymphoma cells when compared to WT mice. Furthermore, when immunized with a low dose of tumor or with an irradiated GM-CSF–secreting tumor vaccine, A2A receptor null mice showed significantly enhanced protection from a subsequent high-dose challenge from both immunogenic and poorly immunogenic tumor lines. This increase in protection was accompanied by an increase in the number of tumor-antigen-specific CD8 T cells at the vaccine-site draining lymph node. Finally, we found that A2A receptor null mice displayed more robust anti-tumor responses than WT mice when they were treated with a soluble B7-DC/Fc fusion protein designed to antagonize B7-H1-mediated co-inhibition. This combinatorial immunotherapy strategy could also be recapitulated with pharmacological A2A receptor blockade paired with B7-DC/Fc administration. In light of these data, we believe that blockade of the A2A adenosine receptor is an attractive target for tumor immunotherapy that synergizes with other immunomodulatory approaches currently in clinical trials.





Similar content being viewed by others
References
Bilzer M, Gerbes AL (2002) Role of G-protein-coupled adenosine receptors in downregulation of inflammation and protection from tissue damage. Z Gastroenterol 40(7):543–544. doi:10.1055/s-2002-32802
Blay J, White TD, Hoskin DW (1997) The extracellular fluid of solid carcinomas contains immunosuppressive concentrations of adenosine. Cancer Res 57(13):2602–2605
Zou AP, Nithipatikom K, Li PL, Cowley AW Jr (1999) Role of renal medullary adenosine in the control of blood flow and sodium excretion. Am J Physiol 276(3 Pt 2):R790–R798
Linden J (2001) Molecular approach to adenosine receptors: receptor-mediated mechanisms of tissue protection. Annu Rev Pharmacol Toxicol 41:775–787
Van Belle H, Goossens F, Wynants J (1987) Formation and release of purine catabolites during hypoperfusion, anoxia, and ischemia. Am J Physiol 252(5 Pt 2):H886–H893
Filippini A, Taffs RE, Sitkovsky MV (1990) Extracellular ATP in T-lymphocyte activation: possible role in effector functions. Proc Nat Acad Sci USA 87(21):8267–8271
Resta R, Yamashita Y, Thompson LF (1998) Ecto-enzyme and signaling functions of lymphocyte CD73. Immunol Rev 161:95–109
Zimmermann H (1999) Two novel families of ectonucleotidases: molecular structures, catalytic properties and a search for function. Trends Pharmacol Sci 20(6):231–236
Olah ME, Stiles GL (1995) Adenosine receptor subtypes: characterization and therapeutic regulation. Annu Rev Pharmacol Toxicol 35:581–606. doi:10.1146/annurev.pa.35.040195.003053
Cronstein BN (1994) Adenosine, an endogenous anti-inflammatory agent. J Appl Physiol 76(1):5–13
Robeva AS, Woodard RL, Jin XW, Gao ZH, Bhattacharya S, Taylor HE, Rosin DL, Linden J (1996) Molecular characterization of recombinant human adenosine receptors. Drug Dev Res 39(3–4):243–252
Furlong TJ, Pierce KD, Selbie LA, Shine J (1992) Molecular characterization of a human brain adenosine A2 receptor. Brain Res Mol Brain Res 15(1–2):62–66
Olah ME, Stiles GL (1995) Adenosine receptor subtypes: characterization and therapeutic regulation. Annu Rev Pharmacol Toxicol 35:581–606
Montesinos MC, Gadangi P, Longaker M, Sung J, Levine J, Nilsen D, Reibman J, Li M, Jiang CK, Hirschhorn R, Recht PA, Ostad E, Levin RI, Cronstein BN (1997) Wound healing is accelerated by agonists of adenosine A(2) (G(alpha s)-linked) receptors. J Exp Med 186(9):1615–1620
Erdmann AA, Gao ZG, Jung U, Foley J, Borenstein T, Jacobson KA, Fowler DH (2005) Activation of Th1 and Tc1 cell adenosine A2A receptors directly inhibits IL-2 secretion in vitro and IL-2-driven expansion in vivo. Blood 105(12):4707–4714
Ohta A, Sitkovsky M (2001) Role of G-protein-coupled adenosine receptors in downregulation of inflammation and protection from tissue damage. Nature 414(6866):916–920. doi:10.1038/414916a
Raskovalova T, Lokshin A, Huang X, Jackson EK, Gorelik E (2006) Adenosine-mediated inhibition of cytotoxic activity and cytokine production by IL-2/NKp46-activated NK cells: involvement of protein kinase A isozyme I (PKA I). Immunol Res 36(1–3):91–99
Schnurr M, Toy T, Shin A, Hartmann G, Rothenfusser S, Soellner J, Davis ID, Cebon J, Maraskovsky E (2004) Role of adenosine receptors in regulating chemotaxis and cytokine production of plasmacytoid dendritic cells. Blood 103(4):1391–1397
Visser SS, Theron AJ, Ramafi G, Ker JA, Anderson R (2000) Apparent involvement of the A(2A) subtype adenosine receptor in the anti-inflammatory interactions of CGS 21680, cyclopentyladenosine, and IB-MECA with human neutrophils. Biochem Pharmacol 60(7):993–999
Scheibner KA, Boodoo S, Collins S, Black KE, Chan-Li Y, Zarek P, Powell JD, Horton MR (2009) The adenosine a2a receptor inhibits matrix-induced inflammation in a novel fashion. Am J Respir Cell Mol Biol 40(3):251–259. doi:10.1165/rcmb.2008-0168OC
Raskovalova T, Lokshin A, Huang X, Su Y, Mandic M, Zarour HM, Jackson EK, Gorelik E (2007) Inhibition of cytokine production and cytotoxic activity of human antimelanoma specific CD8+ and CD4+ T lymphocytes by adenosine-protein kinase A type I signaling. Cancer Res 67(12):5949–5956
Zarek PE, Huang CT, Lutz ER, Kowalski J, Horton MR, Linden J, Drake CG, Powell JD (2008) A2A receptor signaling promotes peripheral tolerance by inducing T-cell anergy and the generation of adaptive regulatory T cells. Blood 111(1):251–259. doi:10.1182/blood-2007-03-081646
Deaglio S, Dwyer KM, Gao W, Friedman D, Usheva A, Erat A, Chen JF, Enjyoji K, Linden J, Oukka M, Kuchroo VK, Strom TB, Robson SC (2007) Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J Exp Med 204(6):1257–1265
Odashima M, Bamias G, Rivera-Nieves J, Linden J, Nast CC, Moskaluk CA, Marini M, Sugawara K, Kozaiwa K, Otaka M, Watanabe S, Cominelli F (2005) Activation of A2A adenosine receptor attenuates intestinal inflammation in animal models of inflammatory bowel disease. Gastroenterology 129(1):26–33
Ohta A, Gorelik E, Prasad SJ, Ronchese F, Lukashev D, Wong MK, Huang X, Caldwell S, Liu K, Smith P, Chen JF, Jackson EK, Apasov S, Abrams S, Sitkovsky M (2006) A2A adenosine receptor protects tumors from antitumor T cells. Proc Nat Acad Sci USA 103(35):13132–13137
Pieters R, Thompson LF, Broekema GJ, Huismans DR, Peters GJ, Pals ST, Horst E, Hahlen K, Veerman AJP (1991) Expression of 5′-Nucleotidase (Cd73) Related to Other Differentiation Antigens in Leukemias of B-Cell Lineage. Blood 78(2):488–492
Jin DC, Fan J, Wang L, Thompson LF, Liu AJ, Daniel BJ, Shin T, Curiel TJ, Zhang B (2010) CD73 on Tumor Cells Impairs Antitumor T-Cell Responses: A Novel Mechanism of Tumor-Induced Immune Suppression. Cancer Res 70(6):2245–2255. doi:10.1158/0008-5472.Can-09-3109
Chen JF, Huang ZH, Ma JY, Zhu JM, Moratalla R, Standaert D, Moskowitz MA, Fink JS, Schwarzschild MA (1999) A(2A) adenosine receptor deficiency attenuates brain injury induced by transient focal ischemia in mice. J Neurosci 19(21):9192–9200
Dranoff G (1995) Hot papers—vaccinology—vaccination with irradiated tumor-cells engineered to secrete murine granulocyte-macrophage colony-stimulating factor stimulates potent, specific, and long-lasting antitumor immunity Dranoff G, Jaffee E, Lazenby A, Golumbek P, Levitsky H, Brose K, Jackson V, Hamada H, Pardoll D, Mulligan Rc - Comments Sci 9 (14):15
Borrello I, Sotomayor EM, Cooke S, Levitsky HI (1999) A universal granulocyte-macrophage colony-stimulating factor-producing bystander cell line for use in the formulation of autologous tumor cell-based vaccines. Hum Gene Ther 10(12):1983–1991
Celik C, Lewis DA, Goldrosen MH (1983) Demonstration of immunogenicity with the poorly immunogenic B16 melanoma. Cancer Res 43(8):3507–3510
Blank C, Mackensen A (2007) Contribution of the PD-L1/PD-1 pathway to T-cell exhaustion: an update on implications for chronic infections and tumor evasion. Cancer Immunol Immunother 56(5):739–745
Dong HD, Zhu GF, Tamada K, Chen LP (1999) B7–H1, a third member of the B7 family, co-stimulates T-cell proliferation and interleukin-10 secretion. Nat Med 5(12):1365–1369
Tseng SY, Otsuji M, Gorski K, Huang X, Slansky JE, Pai SI, Shalabi A, Shin T, Pardoll DM, Tsuchiya H (2001) B7-DC, a new dendritic cell molecule with potent costimulatory properties for T cells. J Exp Med 193(7):839–845
Shin T, Kennedy G, Gorski K, Tsuchiya H, Koseki H, Azuma M, Yagita H, Chen LP, Powell J, Pardoll D, Housseau F (2003) Cooperative B7–1/2 (CD80/CD86) and B7-DC costimulation of CD4(+) T cells independent of the PD-1 receptor. J Exp Med 198(1):31–38. doi:10.1084/Jem.200030242
Dong HD, Strome SE, Salomao DR, Tamura H, Hirano F, Flies DB, Roche PC, Lu J, Zhu GF, Tamada K, Lennon VA, Celis E, Chen LP (2002) Tumor-associated B7–H1 promotes T-cell apoptosis: A potential mechanism of immune evasion. Nat Med 8(8):793–800. doi:10.1038/Nm730
Salamone JD (2010) Preladenant, a novel adenosine A2A receptor antagonist for the potential treatment of parkinsonism and other disorders. IDrugs 13(10):723–731
Kantoff PW, Higano CS, Shore ND, Berger ER, Small EJ, Penson DF, Redfern CH, Ferrari AC, Dreicer R, Sims RB, Xu Y, Frohlich MW, Schellhammer PF (2010) Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med 363(5):411–422. doi:10.1056/NEJMoa1001294
Lassi K, Dawson NA (2010) Update on castrate-resistant prostate cancer: 2010. Curr Opin Oncol 22(3):263–267. doi:10.1097/CCO.0b013e3283380939
Nemunaitis J, Jahan T, Ross H, Sterman D, Richards D, Fox B, Jablons D, Aimi J, Lin A, Hege K (2006) Phase 1/2 trial of autologous tumor mixed with an allogeneic GVAX (R) vaccine in advanced-stage non-small-cell lung cancer. Cancer Gene Ther 13(6):555–562. doi:10.1038/Sj.Cgt.7700922
Curran MA, Montalvo W, Yagita H, Allison JP (2010) PD-1 and CTLA-4 combination blockade expands infiltrating T cells and reduces regulatory T and myeloid cells within B16 melanoma tumors. Proc Nat Acad Sci USA 107(9):4275–4280. doi:10.1073/Pnas.0915174107
Iwahashi M, Katsuda M, Nakamori M, Nakamura M, Naka T, Ojima T, Iida T, Yamaue H (2010) Vaccination with peptides derived from cancer-testis antigens in combination with CpG-7909 elicits strong specific CD8+ T cell response in patients with metastatic esophageal squamous cell carcinoma. Cancer Sci. doi:10.1111/j.1349-7006.2010.01732.x
Hashii Y, Sato E, Ohta H, Oka Y, Sugiyama H, Ozono K (2010) WT1 peptide immunotherapy for cancer in children and young adults. Pediatr Blood Cancer 55(2):352–355. doi:10.1002/pbc.22522
Flavell RA, Sanjabi S, Wrzesinski SH, Licona-Limon P (2010) The polarization of immune cells in the tumour environment by TGFbeta. Nat Rev Immunol 10(8):554–567. doi:10.1038/nri2808
Strauss L, Bergmann C, Szczepanski M, Gooding W, Johnson JT, Whiteside TL (2007) A unique subset of CD4+ CD25 highFoxp3+ T cells secreting interleukin-10 and transforming growth factor-beta1 mediates suppression in the tumor microenvironment. Clin Cancer Res 13(151):4345–4354. doi:10.1158/1078-0432.CCR-07-0472
Deepak P, Kumar S, Acharya A (2007) Interteukin-13-induced type II polarization of inflammatory macrophages is mediated through suppression of nuclear factor-kappa B and preservation of I kappa B alpha in a T cell lymphoma. Clin Exp Immunol 149(2):378–386. doi:10.1111/J.1365-2249.2007.03427.X
Bergmann C, Strauss L, Zeidler R, Lang S, Whiteside TL (2007) Expansion and characteristics of human T regulatory type 1 cells in co-cultures simulating tumor microenvironment. Cancer Immunol Immunother 56(9):1429–1442. doi:10.1007/S00262-007-0280-9
Kusmartsev S, Gabrilovich DI (2002) Immature myeloid cells and cancer-associated immune suppression. Cancer Immunol Immunother 51(6):293–298. doi:10.1007/S00262-002-0280-8
Lappas CM, Rieger JM, Linden J (2005) A2A adenosine receptor induction inhibits IFN-gamma production in murine CD4+ T cells. J Immunol 174(2):1073–1080
Naganuma M, Wiznerowicz EB, Lappas CM, Linden J, Worthington MT, Ernst PB (2006) Cutting Edge: Critical Role for A2A Adenosine Receptors in the T Cell-Mediated Regulation of Colitis. J Immunol 177(5):2765–2769
Sevigny CP, Li L, Awad AS, Huang L, McDuffie M, Linden J, Lobo PI, Okusa MD (2007) Activation of adenosine 2A receptors attenuates allograft rejection and alloantigen recognition. J Immunol 178(7):4240–4249
Sitkovsky MV, Lukashev D, Apasov S, Kojima H, Koshiba M, Caldwell C, Ohta A, Thiel M (2004) Physiological control of immune response and inflammatory tissue damage by hypoxia-inducible factors and adenosine A2A receptors. Annu Rev Immunol 22:657–682. doi:10.1146/annurev.immunol.22.012703.104731
Mangsbo SM, Sandin LC, Anger K, Korman AJ, Loskog A, Totterman TH (2010) Enhanced tumor eradication by combining CTLA-4 or PD-1 blockade with CpG therapy. J Immunother 33(3):225–235. doi:10.1097/CJI.0b013e3181c01fcb
Hernandez J, Ko A, Sherman LA (2001) CTLA-4 blockade enhances the CTL responses to the p53 self-tumor antigen. J Immunol 166(6):3908–3914
Carthon BC, Wolchok JD, Yuan J, Kamat A, Ng Tang DS, Sun J, Ku G, Troncoso P, Logothetis CJ, Allison JP, Sharma P (2010) Preoperative CTLA-4 blockade: tolerability and immune monitoring in the setting of a presurgical clinical trial. Clin Cancer Res 16(10):2861–2871. doi:10.1158/1078-0432.CCR-10-0569
Merighi S, Mirandola P, Milani D, Varani K, Gessi S, Klotz KN, Leung E, Baraldi PG, Morea PA (2002) Adenosine receptors as mediators of both cell proliferation and cell death of cultured human melanoma cells. J Invest Dermatol 119(4):923–933
Ryzhov S, Novitskiy SV, Zaynagetdinov R, Goldstein AE, Carbone DP, Biaggioni I, Dikov MM, Feoktiztov I (2008) Host A(2B) adenosine receptor promotes carcinoma growth. Neoplasia 10(9):986–995
Acknowledgments
We thank Amplimmune for their generous gift of B7-DC/FC for use in this study, and to Charles Drake and Ivan Borrello for their critical reviews. Funding for this study was provided by the NIH grant R01CA114227.
Conflict of interest
Jonathan Powell is a Scientific Founder of Amplimmune, Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Waickman, A.T., Alme, A., Senaldi, L. et al. Enhancement of tumor immunotherapy by deletion of the A2A adenosine receptor. Cancer Immunol Immunother 61, 917–926 (2012). https://doi.org/10.1007/s00262-011-1155-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-011-1155-7