Abstract
Purpose
CD38 is considered a potential biomarker for multiple myeloma (MM) and has shown a strong link with chronic lymphocytic leukemia due to high and uniform expression on plasma cells. In vivo evaluation of CD38 expression may provide useful information about lesion detection and prognosis of treatment in MM. In this study, immunoPET imaging with 89Zr-labeled daratumumab was used for differentiation of CD38 expression in murine lymphoma models to provide a potential non-invasive method for monitoring CD38 in the clinic.
Methods
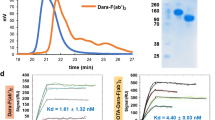
Daratumumab was radiolabeled with 89Zr (t1/2 = 78.4 h) via conjugation with desferrioxamine (Df). After Western blot (WB) was used to screen CD38 expression in five lymphoma cell lines, flow cytometry and cellular binding assays were performed to test the binding ability of labeled or conjugated daratumumab with CD38 in vitro. PET imaging and biodistribution studies were performed to evaluate CD38 expression after injection of 89Zr-Df-daratumumab. 89Zr-Df-IgG was also evaluated as a non-specific control group in the Ramos model. Finally, CD38 expression in tumor tissues was verified by histological analysis.
Results
Using WB screening, the Ramos cell line was found to express the highest level of CD38 while the HBL-1 cell line had the lowest expression. Df-conjugated and 89Zr-labeled daratumumab displayed similar high binding affinities with Ramos cells. PET imaging of 89Zr-Df-daratumumab showed a high tumor uptake of up to 26.6 ± 8.0 %ID/g for Ramos at 120 h post-injection, and only up to 6.6 ± 2.9 %ID/g for HBL-1 (n = 4). Additionally, 89Zr-Df-IgG demonstrated a low tumor uptake in the Ramos model (only 4.3 ± 0.8 %ID/g at 120 h post-injection). Ex vivo biodistribution studies showed similar trends with imaging results. Immunofluorescence staining of tumor tissues verified higher CD38 expression of Ramos than that of HBL-1.
Conclusions
The role of 89Zr-Df-daratumumab was investigated for evaluating CD38 expression in lymphoma models non-invasively and was found to be to a promising imaging agent of CD38-positive hematological diseases such as MM in future clinical applications.





Similar content being viewed by others
References
Swerdlow SH, Campo E, Pileri SA, Harris NL, Stein H, Siebert R, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127:2375–90. https://doi.org/10.1182/blood-2016-01-643569.
Regelink JC, Minnema MC, Terpos E, Kamphuis MH, Raijmakers PG, Pieters-van den Bos IC, et al. Comparison of modern and conventional imaging techniques in establishing multiple myeloma-related bone disease: a systematic review. Br J Haematol. 2013;162:50–61. https://doi.org/10.1111/bjh.12346.
Ehlerding EB, England CG, McNeel DG, Cai W. Molecular imaging of immunotherapy targets in cancer. J Nucl Med. 2016;57:1487–92. https://doi.org/10.2967/jnumed.116.177493.
van Dongen GA, Visser GW, Lub-de Hooge MN, de Vries EG, Perk LR. Immuno-PET: a navigator in monoclonal antibody development and applications. Oncologist. 2007;12:1379–89. https://doi.org/10.1634/theoncologist.12-12-1379.
Malavasi F, Deaglio S, Funaro A, Ferrero E, Horenstein AL, Ortolan E, et al. Evolution and function of the ADP ribosyl cyclase/CD38 gene family in physiology and pathology. Physiol Rev. 2008;88:841–86. https://doi.org/10.1152/physrev.00035.2007.
Deaglio S, Mehta K, Malavasi F. Human CD38: a (r)evolutionary story of enzymes and receptors. Leuk Res. 2001;25:1–12.
Sanchez L, Wang Y, Siegel DS, Wang ML. Daratumumab: a first-in-class CD38 monoclonal antibody for the treatment of multiple myeloma. J Hematol Oncol. 2016;9:51. https://doi.org/10.1186/s13045-016-0283-0.
Malavasi F, Deaglio S, Damle R, Cutrona G, Ferrarini M, Chiorazzi N. CD38 and chronic lymphocytic leukemia: a decade later. Blood. 2011;118:3470–8. https://doi.org/10.1182/blood-2011-06-275610.
Amaya-Chanaga CI, Rassenti LZ. Biomarkers in chronic lymphocytic leukemia: clinical applications and prognostic markers. Best Pract Res Clin Haematol. 2016;29:79–89. https://doi.org/10.1016/j.beha.2016.08.005.
Lokhorst HM, Plesner T, Laubach JP, Nahi H, Gimsing P, Hansson M, et al. Targeting CD38 with Daratumumab Monotherapy in multiple myeloma. N Engl J Med. 2015;373:1207–19. https://doi.org/10.1056/NEJMoa1506348.
de Weers M, Tai YT, van der Veer MS, Bakker JM, Vink T, Jacobs DC, et al. Daratumumab, a novel therapeutic human CD38 monoclonal antibody, induces killing of multiple myeloma and other hematological tumors. J Immunol. 2011;186:1840–8. https://doi.org/10.4049/jimmunol.1003032.
Overdijk MB, Verploegen S, Bogels M, van Egmond M, Lammerts van Bueren JJ, Mutis T, et al. Antibody-mediated phagocytosis contributes to the anti-tumor activity of the therapeutic antibody daratumumab in lymphoma and multiple myeloma. MAbs. 2015;7:311–21. https://doi.org/10.1080/19420862.2015.1007813.
Overdijk MB, Jansen JH, Nederend M, Lammerts van Bueren JJ, Groen RW, Parren PW, et al. The therapeutic CD38 monoclonal antibody Daratumumab induces programmed cell death via Fcgamma receptor-mediated cross-linking. J Immunol. 2016;197:807–13. https://doi.org/10.4049/jimmunol.1501351.
Ehlerding EB, England CG, Jiang D, Graves SA, Kang L, Lacognata S, et al. CD38 as a PET imaging target in lung cancer. Mol Pharm. 2017;14:2400–6. https://doi.org/10.1021/acs.molpharmaceut.7b00298.
Hong H, Severin GW, Yang Y, Engle JW, Zhang Y, Barnhart TE, et al. Positron emission tomography imaging of CD105 expression with 89Zr-Df-TRC105. Eur J Nucl Med Mol Imaging. 2012;39:138–48. https://doi.org/10.1007/s00259-011-1930-x.
England CG, Ehlerding EB, Hernandez R, Rekoske BT, Graves SA, Sun H, et al. Preclinical pharmacokinetics and biodistribution studies of 89Zr-Labeled Pembrolizumab. J Nucl Med. 2017;58:162–8. https://doi.org/10.2967/jnumed.116.177857.
Abou DS, Ku T, Smith-Jones PM. In vivo biodistribution and accumulation of 89Zr in mice. Nucl Med Biol. 2011;38:675–81. https://doi.org/10.1016/j.nucmedbio.2010.12.011.
Bailly C, Clery PF, Faivre-Chauvet A, Bourgeois M, Guerard F, Haddad F, et al. Immuno-PET for clinical Theranostic approaches. Int J Mol Sci. 2016;18 https://doi.org/10.3390/ijms18010057.
Wright BD, Lapi SE. Designing the magic bullet? The advancement of immuno-PET into clinical use. J Nucl Med. 2013;54:1171–4. https://doi.org/10.2967/jnumed.113.126086.
Zojer N, Kirchbacher K, Vesely M, Hubl W, Ludwig H. Rituximab treatment provides no clinical benefit in patients with pretreated advanced multiple myeloma. Leuk Lymphoma. 2006;47:1103–9. https://doi.org/10.1080/10428190600564803.
Dimopoulos M, Terpos E, Comenzo RL, Tosi P, Beksac M, Sezer O, et al. International myeloma working group consensus statement and guidelines regarding the current role of imaging techniques in the diagnosis and monitoring of multiple myeloma. Leukemia. 2009;23:1545–56. https://doi.org/10.1038/leu.2009.89.
Dimopoulos MA, Hillengass J, Usmani S, Zamagni E, Lentzsch S, Davies FE, et al. Role of magnetic resonance imaging in the management of patients with multiple myeloma: a consensus statement. J Clin Oncol. 2015;33:657–64. https://doi.org/10.1200/JCO.2014.57.9961.
Bartel TB, Haessler J, Brown TL, Shaughnessy JD Jr, van Rhee F, Anaissie E, et al. F18-fluorodeoxyglucose positron emission tomography in the context of other imaging techniques and prognostic factors in multiple myeloma. Blood. 2009;114:2068–76. https://doi.org/10.1182/blood-2009-03-213280.
de Waal EG, Glaudemans AW, Schroder CP, Vellenga E, Slart RH. Nuclear medicine imaging of multiple myeloma, particularly in the relapsed setting. Eur J Nucl Med Mol Imaging. 2017;44:332–41. https://doi.org/10.1007/s00259-016-3576-1.
Rajkumar SV, Harousseau JL, Durie B, Anderson KC, Dimopoulos M, Kyle R, et al. Consensus recommendations for the uniform reporting of clinical trials: report of the international myeloma workshop consensus panel 1. Blood. 2011;117:4691–5. https://doi.org/10.1182/blood-2010-10-299487.
Ak I, Gulbas Z. F-18 FDG uptake of bone marrow on PET/CT scan: it’s correlation with CD38/CD138 expressing myeloma cells in bone marrow of patients with multiple myeloma. Ann Hematol. 2011;90:81–7. https://doi.org/10.1007/s00277-010-1037-7.
Ak I, Aslan V, Vardareli E, Gulbas Z. Tc-99m methoxyisobutylisonitrile bone marrow imaging for predicting the levels of myeloma cells in bone marrow in multiple myeloma: correlation with CD38/CD138 expressing myeloma cells. Ann Hematol. 2003;82:88–92. https://doi.org/10.1007/s00277-002-0600-2.
Chang X, Yue L, Liu W, Wang Y, Wang L, Xu B, et al. CD38 and E2F transcription factor 2 have uniquely increased expression in rheumatoid arthritis synovial tissues. Clin Exp Immunol. 2014;176:222–31. https://doi.org/10.1111/cei.12268.
Pavon EJ, Zumaquero E, Rosal-Vela A, Khoo KM, Cerezo-Wallis D, Garcia-Rodriguez S, et al. Increased CD38 expression in T cells and circulating anti-CD38 IgG autoantibodies differentially correlate with distinct cytokine profiles and disease activity in systemic lupus erythematosus patients. Cytokine. 2013;62:232–43. https://doi.org/10.1016/j.cyto.2013.02.023.
Lins L, Farias E, Brites-Alves C, Torres A, Netto EM, Brites C. Increased expression of CD38 and HLADR in HIV-infected patients with oral lesion. J Med Virol. 2017;89:1782–7. https://doi.org/10.1002/jmv.24852.
Dentone C, Fenoglio D, Schenone E, Cenderello G, Prinapori R, Signori A, et al. Increased CD38 expression on T lymphocytes as a marker of HIV dissemination into the central nervous system. HIV Clin Trials. 2015;16:190–6. https://doi.org/10.1179/1945577115Y.0000000005.
Boix F, Millan O, San Segundo D, Mancebo E, Rimola A, Fabrega E, et al. High expression of CD38, CD69, CD95 and CD154 biomarkers in cultured peripheral T lymphocytes correlates with an increased risk of acute rejection in liver allograft recipients. Immunobiology. 2016;221:595–603. https://doi.org/10.1016/j.imbio.2016.01.008.
Shallis RM, Terry CM, Lim SH. The multi-faceted potential of CD38 antibody targeting in multiple myeloma. Cancer Immunol Immunother. 2017;66:697–703. https://doi.org/10.1007/s00262-017-1990-2.
Kraeber-Bodere F, Bailly C, Cherel M, Chatal JF. ImmunoPET to help stratify patients for targeted therapies and to improve drug development. Eur J Nucl Med Mol Imaging. 2016;43:2166–8. https://doi.org/10.1007/s00259-016-3458-6.
Funaro A, Horenstein AL, Calosso L, Morra M, Tarocco RP, Franco L, et al. Identification and characterization of an active soluble form of human CD38 in normal and pathological fluids. Int Immunol. 1996;8:1643–50.
Luo H, Hernandez R, Hong H, Graves SA, Yang Y, England CG, et al. Noninvasive brain cancer imaging with a bispecific antibody fragment, generated via click chemistry. Proc Natl Acad Sci U S A. 2015;112:12806–11. https://doi.org/10.1073/pnas.1509667112.
Lam K, Chan C, Reilly RM. Development and preclinical studies of 64Cu-NOTA-pertuzumab F(ab’)2 for imaging changes in tumor HER2 expression associated with response to trastuzumab by PET/CT. MAbs. 2017;9:154–64. https://doi.org/10.1080/19420862.2016.1255389.
Zhang Y, Hong H, Orbay H, Valdovinos HF, Nayak TR, Theuer CP, et al. PET imaging of CD105/endoglin expression with a (6)(1)/(6)(4)cu-labeled Fab antibody fragment. Eur J Nucl Med Mol Imaging. 2013;40:759–67. https://doi.org/10.1007/s00259-012-2334-2.
Houghton JL, Zeglis BM, Abdel-Atti D, Sawada R, Scholz WW, Lewis JS. Pretargeted Immuno-PET of pancreatic cancer: overcoming circulating antigen and internalized antibody to reduce radiation doses. J Nucl Med. 2016;57:453–9. https://doi.org/10.2967/jnumed.115.163824.
Witzig TE, Gordon LI, Cabanillas F, Czuczman MS, Emmanouilides C, Joyce R, et al. Randomized controlled trial of yttrium-90-labeled ibritumomab tiuxetan radioimmunotherapy versus rituximab immunotherapy for patients with relapsed or refractory low-grade, follicular, or transformed B-cell non-Hodgkin’s lymphoma. J Clin Oncol. 2002;20:2453–63. https://doi.org/10.1200/JCO.2002.11.076.
Green DJ, Orgun NN, Jones JC, Hylarides MD, Pagel JM, Hamlin DK, et al. A preclinical model of CD38-pretargeted radioimmunotherapy for plasma cell malignancies. Cancer Res. 2014;74:1179–89. https://doi.org/10.1158/0008-5472.CAN-13-1589.
Teiluf K, Seidl C, Blechert B, Gaertner FC, Gilbertz KP, Fernandez V, et al. Alpha-Radioimmunotherapy with (2)(1)(3)bi-anti-CD38 immunoconjugates is effective in a mouse model of human multiple myeloma. Oncotarget. 2015;6:4692–703. https://doi.org/10.18632/oncotarget.2986.
Acknowledgments
This work was supported by the University of Wisconsin - Madison, the National Institutes of Health (NIBIB/NCI 1R01CA169365, 1R01EB021336, P30CA014520, T32CA009206), the American Cancer Society (125246-RSG-13-099-01-CCE), the National Natural Science Foundation of China (81672602, 81472589, 31771036, 51703132, 51573096), the Beijing Nova Program (Z141102001814055, Z171100001117024), the Beijing Capital Special Development Application Program (Z141107002514159), PKU medicine-engineer collaborative seed program (BMU20160574), Logistics Scientific Research Project (BWS16J010) and the Basic Research Program of Shenzhen (JCYJ20170412111100742, JCYJ20160422091238319)
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Kang, L., Jiang, D., England, C.G. et al. ImmunoPET imaging of CD38 in murine lymphoma models using 89Zr-labeled daratumumab. Eur J Nucl Med Mol Imaging 45, 1372–1381 (2018). https://doi.org/10.1007/s00259-018-3941-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-018-3941-3