Abstract
Purpose
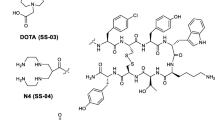
Peptide receptor targeting has become an increasingly attractive method to target tumors diagnostically and radiotherapeutically. Peptides linked to a variety of chelators have been developed for this purpose. They have, however, rarely been tested for their agonistic or antagonistic properties. We report here on a somatostatin antagonist that switched to an agonist upon coupling to a DOTA chelator.
Methods
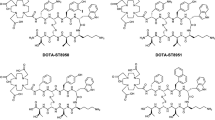
Two novel somatostatin analogs, 406-040-15 and its DOTA-coupled counterpart 406-051-20, with and without cold Indium labeling, were tested for their somatostatin receptor subtypes 1–5 (sst1–sst5) binding affinity using receptor autoradiography. Moreover, they were tested functionally for their ability to affect sst2 and sst3 internalization in vitro in HEK293 cells stably expressing the human sst2 or sst3 receptor, using an immunofluorescence microscopy-based internalization assay.
Results
All three compounds were characterized as pan-somatostatin analogs having a high affinity for all five sst. In the sst2 internalization assay, all three compounds showed an identical behavior, namely, a weak agonistic effect complemented by a weak antagonistic effect, compatible with the behavior of a partial agonist. Conversely, in the sst3 internalization assay, 406-040-15 was a full antagonist whereas its DOTA-coupled counterpart, 406-051-20, with and without Indium labeling, switched to a full agonist.
Conclusion
Adding the DOTA chelator to the somatostatin analog 406-040-15 triggers a switch at sst3 receptor from an antagonist to an agonist. This indicates that potential radioligands for tumor targeting should always be tested functionally before further development, in particular if a chelator is added.




Similar content being viewed by others
References
Reubi JC. Peptide receptors as molecular targets for cancer diagnosis and therapy. Endocr Rev 2003;24:389–427.
van Essen M, Krenning EP, Kam BL, de Jong M, Valkema R, Kwekkeboom DJ. Peptide-receptor radionuclide therapy for endocrine tumors. Nat Rev Endocrinol 2009;5:382–93.
Rufini V, Calcagni ML, Baum RP. Imaging of neuroendocrine tumors. Semin Nucl Med 2006;36:228–47.
Reubi JC, Maecke HR. Peptide-based probes for cancer imaging. J Nucl Med 2008;49:1735–8.
Nock B, Nikolopoulou A, Chiotellis E, Loudos G, Maintas D, Reubi JC, et al. [99mTc]Demobesin 1, a novel potent bombesin analogue for GRP receptor-targeted tumour imaging. Eur J Nucl Med Mol Imaging 2003;30:247–58.
Behr TM, Jenner N, Béhé M, Angerstein C, Gratz S, Raue F, et al. Radiolabeled peptides for targeting cholecystokinin-B/gastrin receptor-expressing tumors. J Nucl Med 1999;40:1029–44.
Gotthardt M, Fischer M, Naeher I, Holz JB, Jungclas H, Fritsch HW, et al. Use of the incretin hormone glucagon-like peptide-1 (GLP-1) for the detection of insulinomas: initial experimental results. Eur J Nucl Med Mol Imaging 2002;29:597–606.
Wild D, Schmitt JS, Ginj M, Mäcke HR, Bernard BF, Krenning E, et al. DOTA-NOC, a high-affinity ligand of somatostatin receptor subtypes 2, 3 and 5 for labelling with various radiometals. Eur J Nucl Med Mol Imaging 2003;30:1338–47.
Zhang H, Schuhmacher J, Waser B, Wild D, Eisenhut M, Reubi JC, et al. DOTA-PESIN, a DOTA-conjugated bombesin derivative designed for the imaging and targeted radionuclide treatment of bombesin receptor-positive tumours. Eur J Nucl Med Mol Imaging 2007;34:1198–208.
Cescato R, Schulz S, Waser B, Eltschinger V, Rivier J, Wester HJ, et al. Internalization of sst2, sst3 and sst5 receptors: effects of somatostatin agonists and antagonists. J Nucl Med 2006;47:502–11.
Ginj M, Zhang H, Waser B, Cescato R, Wild D, Wang X, et al. Radiolabeled somatostatin receptor antagonists are preferable to agonists for in vivo peptide receptor targeting of tumors. Proc Natl Acad Sci USA 2006;103:16436–41.
Waser B, Tamma ML, Cescato R, Maecke HR, Reubi JC. Highly efficient in vivo agonist-induced internalization of sst2 receptors in somatostatin target tissues. J Nucl Med 2009;50:936–41.
Cescato R, Maina T, Nock B, Nikolopoulou A, Charalambidis D, Piccand V, et al. Bombesin receptor antagonists may be preferable to agonists for tumor targeting. J Nucl Med 2008;49:318–26.
Cescato R, Erchegyi J, Waser B, Piccand V, Maecke HR, Rivier JE, et al. Design and in vitro characterization of highly sst2-selective somatostatin antagonists suitable for radiotargeting. J Med Chem 2008;51:4030–7. doi:10.1021/jm701618q10.1021/jm701618q.
Mansi R, Wang X, Forrer F, Kneifel S, Tamma ML, Waser B, et al. Evaluation of a 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid-conjugated bombesin-based radioantagonist for the labeling with single-photon emission computed tomography, positron emission tomography, and therapeutic radionuclides. Clin Cancer Res 2009;15:5240–9.
Edreira M, Melendez-Alafort L, Mather SJ. Optimization of the small-scale synthesis of DOTA-Tyr3-octreotide. Nucl Med Commun 2002;23:493–9.
Reubi JC, Schär JC, Waser B, Wenger S, Heppeler A, Schmitt J, et al. Affinity profiles for human somatostatin receptor subtypes sst1-sst5 of somatostatin radiotracers selected for scintigraphic and radiotherapeutic use. Eur J Nucl Med 2000;27:273–82.
Reubi JC, Kvols LK, Waser B, Nagorney D, Heitz PU, Charboneau JW, et al. Detection of somatostatin receptors in surgical and percutaneous needle biopsy samples of carcinoids and islet cell carcinomas. Cancer Res 1990;50:5969–77.
Erchegyi J, Cescato R, Grace CR, Waser B, Piccand V, Hoyer D, et al. Novel, potent, and radio-iodinatable somatostatin receptor 1 (sst1) selective analogues. J Med Chem 2009;52:2733–46.
Olias G, Viollet C, Kusserow H, Epelbaum J, Meyerhof W. Regulation and function of somatostatin receptors. J Neurochem 2004;89:1057–91.
Ginj M, Zhang H, Eisenwiener KP, Wild D, Schulz S, Rink H, et al. New pansomatostatin ligands and their chelated versions: affinity profile, agonist activity, internalization, and tumor targeting. Clin Cancer Res 2008;14:2019–27.
Bass RT, Buckwalter BL, Patel BP, Pausch MH, Price LA, Strnad J, et al. Identification and characterization of novel somatostatin antagonists [published erratum appears in Mol Pharmacol 1997 Jan;51(1):170]. Mol Pharmacol 1996;50:709–15.
Schlag BD, Lou Z, Fennell M, Dunlop J. Ligand dependency of 5-hydroxytryptamine 2C receptor internalization. J Pharmacol Exp Ther 2004;310:865–70.
Corbani M, Gonindard C, Meunier JC. Ligand-regulated internalization of the opioid receptor-like 1: a confocal study. Endocrinology 2004;145:2876–85.
Liu Q, Cescato R, Dewi DA, Rivier J, Reubi JC, Schonbrunn A. Receptor signaling and endocytosis are differentially regulated by somatostatin analogs. Mol Pharmacol 2005;68:90–101.
Edwards WB, Xu B, Akers W, Cheney PP, Liang K, Rogers BE, et al. Agonist-antagonist dilemma in molecular imaging: evaluation of a monomolecular multimodal imaging agent for the somatostatin receptor. Bioconjug Chem 2008;19:192–200.
Acknowledgments
The project described was supported in part by a grant Number 3200B0-105726/1 from the Swiss National Science Foundation and by Award Number DK059953 from the National Institute of Diabetes and Digestive and Kidney Diseases. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Diabetes and Digestive and Kidney Diseases or the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reubi, J.C., Erchegyi, J., Cescato, R. et al. Switch from antagonist to agonist after addition of a DOTA chelator to a somatostatin analog. Eur J Nucl Med Mol Imaging 37, 1551–1558 (2010). https://doi.org/10.1007/s00259-010-1445-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-010-1445-x