Abstract
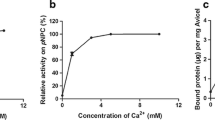
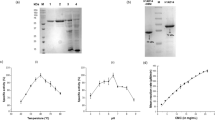
Cytophaga hutchinsonii is an aerobic cellulolytic soil bacterium that rapidly digests crystalline cellulose. The predicted mechanism by which C. hutchinsonii digests cellulose differs from that of other known cellulolytic bacteria and fungi. The genome of C. hutchinsonii contains 22 glycoside hydrolase (GH) genes, which may be involved in cellulose degradation. One predicted GH with uncertain specificity, CHU_0961, is a modular enzyme with several modules. In this study, phylogenetic tree of the catalytic modules of the GH9 enzymes showed that CHU_0961 and its homologues formed a new group (group C) of GH9 enzymes. The catalytic module of CHU_0961 (CHU_0961B) was identified as a 1,4-β-d-glucan glucohydrolase (EC 3.2.1.74) that has unique properties compared with known GH9 cellulases. CHU_0961B showed highest activity against barley glucan, but low activity against other polysaccharides. Interestingly, CHU_0961B showed similar activity against ρ-nitrophenyl β-d-cellobioside (ρ-NPC) and ρ-nitrophenyl β-d-glucopyranoside. CHU_0961B released glucose from the nonreducing end of cello-oligosaccharides, ρ-NPC, and barley glucan in a nonprocessive exo-type mode. CHU_0961B also showed same hydrolysis mode against deacetyl-chitooligosaccharides as against cello-oligosaccharides. The kcat/Km values for CHU_0961B against cello-oligosaccharides increased as the degree of polymerization increased, and its kcat/Km for cellohexose was 750 times higher than that for cellobiose. Site-directed mutagenesis showed that threonine 321 in CHU_0961 played a role in hydrolyzing cellobiose to glucose. CHU_0961 may act synergistically with other cellulases to convert cellulose to glucose on the bacterial cell surface. The end product, glucose, may initiate cellulose degradation to provide nutrients for bacterial proliferation in the early stage of C. hutchinsonii growth.
Key points
• CHU_0961 and its homologues formed a novel group (group C) of GH9 enzymes.
• CHU_0961 was identified as a 1,4-β- d -glucan glucohydrolase with unique properties.
• CHU_0961 may play an important role in the early stage of C. hutchinsonii growth.






Similar content being viewed by others
References
Artzi L, Bayer EA, Morais S (2017) Cellulosomes: bacterial nanomachines for dismantling plant polysaccharides. Nat Rev Microbiol 15:83–95. https://doi.org/10.1038/nrmicro.2016.164
Bai XF, Wang XF, Wang S, Ji XF, Guan ZW, Zhang WC, Lu XM (2017) Functional studies of β-glucosidases of Cytophaga hutchinsonii and their effects on cellulose degradation. Front Microbiol 8:140. https://doi.org/10.3389/fmicb.2017.00140
Bayer EA, Shimon LJW, Shoham Y, Lamed R (1998) Cellulosomes - Structure and ultrastructure. J Struct Biol 124:221–234. https://doi.org/10.1006/jsbi.1998.4065
Bycroft M, Bateman A, Clarke J, Hamill SJ, Sandford R, Thomas RL, Chothia C (1999) The structure of a PKD domain from polycystin-1: implications for polycystic kidney disease. EMBO J 18:297–305. https://doi.org/10.1093/emboj/18.2.297
Chojnacki S, Cowley A, Lee J, Foix A, Lopez R (2017) Programmatic access to bioinformatics tools from EMBL-EBI update: 2017. Nucleic Acids Res 45(W1):W550–W553. https://doi.org/10.1093/nar/gkx273
Duan CJ, Feng YL, Cao QL, Huang MY, Feng JX (2016) Identification of a novel family of carbohydrate-binding modules with broad ligand specificity. Sci Rep 6:19392. https://doi.org/10.1038/srep19392
Duan CJ, Huang MY, Pang H, Zhao J, Wu CX, Feng JX (2017) Characterization of a novel theme C glycoside hydrolase family 9 cellulase and its CBM-chimeric enzymes. Appl Microbiol Biotechnol 101:5723–5737. https://doi.org/10.1007/s00253-017-8320-7
Finn RD, Coggill P, Eberhardt RY, Eddy SR, Mistry J, Mitchell AL, Potter SC, Punta M, Qureshi M, Sangrador-Vegas A, Salazar GA, Tate J, Bateman A (2016) The Pfam protein families database: towards a more sustainable future. Nucleic Acids Res 44(D1):D279–D285. https://doi.org/10.1093/nar/gkv1344
Gebler J, Gilkes NR, Claeyssens M, Wilson DB, Beguin P, Wakarchuk WW, Kilburn DG, Miller RC Jr, Warren RA, Withers SG (1992) Stereoselective hydrolysis catalyzed by related β-1,4-glucanases and β-1,4-xylanases. J Biol Chem 267:12559–12561. https://doi.org/10.1016/0092-8674(92)90644-R
Gilad R, Rabinovich L, Yaron S, Bayer EA, Lamed R, Gilbert HJ, Shoham Y (2003) CelI, a noncellulosomal family 9 enzyme from Clostridium thermocellum, is a processive endoglucanase that degrades crystalline cellulose. J Bacteriol 185:391–398. https://doi.org/10.1128/JB.185.2.391-398.2003
Himmel ME, Ding SY, Johnson DK, Adney WS, Nimlos MR, Brady JW, Foust TD (2007) Biomass recalcitrance: engineering plants and enzymes for biofuels production. Science 315:804–807. https://doi.org/10.1126/science.1137016
Ji X, Wang Y, Zhang C, Bai X, Zhang W, Lu X (2014) Novel outer membrane protein involved in cellulose and cellooligosaccharide degradation by Cytophaga hutchinsonii. Appl Environ Microbiol 80:4511–4518. https://doi.org/10.1128/AEM.00687-14
Kataeva IA, Uversky VN, Brewer JM, Schubot F, Rose JP, Wang BC, Ljungdahl LG (2004) Interactions between immunoglobulin-like and catalytic modules in Clostridium thermocellum cellulosomal cellobiohydrolase CbhA. Protein Eng Des Sel 17:759–769. https://doi.org/10.1093/protein/gzh094
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Lasica AM, Ksiazek M, Madej M, Potempa J (2017) The type IX secretion system (T9SS): highlights and recent insights into its structure and function. Front Cell Infect Microbiol 7:215. https://doi.org/10.3389/fcimb.2017.00215
Li Y, Irwin DC, Wilson DB (2007) Processivity, substrate binding, and mechanism of cellulose hydrolysis by Thermobifida fusca Cel9A. Appl Environ Microbiol 73:3165–3172. https://doi.org/10.1128/AEM.02960-06
Louime C, Abazinge M, Johnson E, Latinwo L, Ikediobi C, Clark AM (2007) Molecular cloning and biochemical characterization of a family-9 endoglucanase with an unusual structure from the gliding bacteria Cytophaga hutchinsonii. Appl Biochem Biotechnol 141(1):127–138. https://doi.org/10.1007/s12010-007-9215-3
Lynd LR, Weimer PJ, van Zyl WH, Pretorius IS (2002) Microbial cellulose utilization: fundamentals and biotechnology. Microbiol Mol Biol Rev 66:506–577. https://doi.org/10.1128/mmbr.66.3.506-577.2002
Miller GL (1959) Use of dinitrosalicyclic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Odoux E, Escoute J, Verdiel JL, Brillouet JM (2003) Localization of β-D-glucosidase activity and glucovanillin in vanilla bean (Vanilla planifolia Andrews). Ann Bot 92:437–444. https://doi.org/10.1093/aob/mcg150
Park JK, Wang LX, Patel HV, Roseman S (2002) Molecular cloning and characterization of a unique β-glucosidase from Vibrio cholerae. J Biol Chem 277(33):29555–29560. https://doi.org/10.1074/jbc.M202978200
Qi M, Jun HS, Forsberg CW (2008) Cel9D, an atypical 1,4-β-D-glucan glucohydrolase from Fibrobacter succinogenes: characteristics, catalytic residues, and synergistic interactions with other cellulases. J Bacteriol 190:1976–1984. https://doi.org/10.1128/JB.01667-07
Ravachol J, Borne R, Tardif C, de Philip P, Fierobe HP (2014) Characterization of all family-9 glycoside hydrolases synthesized by the cellulosome-producing bacterium Clostridium cellulolyticum. J Biol Chem 289:7335–7348. https://doi.org/10.1074/jbc.M113.545046
Taillefer M, Arntzen MO, Henrissat B, Pope PB, Larsbrink J (2018) Proteomic dissection of the cellulolytic machineries used by soil-dwelling Bacteroidetes. mSystems 3:e00240–e00218. https://doi.org/10.1128/mSystems.00240-18
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599. https://doi.org/10.1093/molbev/msm092
Veith PD, Nor Muhammad NA, Dashper SG, Likic VA, Gorasia DG, Chen D, Byrne SJ, Catmull DV, Reynolds EC (2013) Protein substrates of a novel secretion system are numerous in the Bacteroidetes phylum and have in common a cleavable C-terminal secretion signal, extensive post-translational modification, and cell-surface attachment. J Proteome Res 12:4449–4461. https://doi.org/10.1021/pr400487b
Wang S, Zhao D, Bai X, Zhang W, Lu X (2017) Identification and characterization of a large protein essential for degradation of the crystalline region of cellulose by Cytophaga hutchinsonii. Appl Environ Microbiol 83:e02270–e02216. https://doi.org/10.1128/AEM.02270-16
Wilson DB (2008) Three microbial strategies for plant cell wall degradation. Ann N Y Acad Sci 1125:289–297. https://doi.org/10.1196/annals.1419.026
Wilson DB (2009) Evidence for a novel mechanism of microbial cellulose degradation. Cellulose 16:723–727. https://doi.org/10.1007/s10570-009-9326-9
Xie G, Bruce DC, Challacombe JF, Chertkov O, Detter JC, Gilna P, Han CS, Lucas S, Misra M, Myers GL, Richardson P, Tapia R, Thayer N, Thompson LS, Brettin TS, Henrissat B, Wilson DB, McBride MJ (2007) Genome sequence of the cellulolytic gliding bacterium Cytophaga hutchinsonii. Appl Environ Microbiol 73:3536–3546. https://doi.org/10.1128/Aem.00225-07
Zhang YH, Cui J, Lynd LR, Kuang LR (2006) A transition from cellulose swelling to cellulose dissolution by o-phosphoric acid: evidence from enzymatic hydrolysis and supramolecular structure. Biomacromolecules 7:644–648. https://doi.org/10.1021/bm050799c
Zhang C, Wang Y, Li Z, Zhou XR, Zhang WC, Zhao Y, Lu XM (2014) Characterization of a multi-function processive endoglucanase CHU_2103 from Cytophaga hutchinsonii. Appl Microbiol Biotechnol 98:6679–6687. https://doi.org/10.1007/s00253-014-5640-8
Zhang C, Zhang WC, Lu XM (2015) Expression and characteristics of a Ca2+-dependent endoglucanase from Cytophaga hutchinsonii. Appl Microbiol Biotechnol 99(22):9617–9623. https://doi.org/10.1007/s00253-015-6746-3
Zhou W, Irwin DC, Escovar-Kousen J, Wilson DB (2004) Kinetic studies of Thermobifida fusca Cel9A active site mutant enzymes. Biochemistry 43:9655–9663. https://doi.org/10.1021/bi049394n
Zhu YT, McBride MJ (2017) The unusual cellulose utilization system of the aerobic soil bacterium Cytophaga hutchinsonii. Appl Microbiol Biotechnol 101:7113–7127. https://doi.org/10.1007/s00253-017-8467-2
Zhu YT, Zhou H, Bi YL, Zhang WX, Chen GJ, Liu WF (2013) Characterization of a family 5 glycoside hydrolase isolated from the outer membrane of cellulolytic Cytophaga hutchinsonii. Appl Microbiol Biotechnol 97(9):3925–3937. https://doi.org/10.1007/s00253-012-4259-x
Zhu Y, Han L, Hefferon KL, Silvaggi NR, Wilson DB, McBride MJ (2016) Periplasmic Cytophaga hutchinsonii endoglucanases are required for use of crystalline cellulose as the sole source of carbon and energy. Appl Environ Microbiol 82:4835–4845. https://doi.org/10.1128/AEM.01298-16
Funding
This work was financially supported by grant from the National Natural Science Foundation of China (grant no. 31560019) and the Guangxi Natural Science Foundation (grant no. 2018GXNSFAA138053).
Author information
Authors and Affiliations
Contributions
C-JD and J-XF conceived and designed research. NJ, X-DM, and L-HF conducted experiments. C-XL contributed bioinformational analysis. C-JD and NJ analyzed data. C-JD and NJ wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Ethical statement
This article does not contain any studies with human participants and/or animals performed by any of the authors
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 1290 kb).
Rights and permissions
About this article
Cite this article
Jiang, N., Ma, XD., Fu, LH. et al. Identification of a unique 1,4-β-d-glucan glucohydrolase of glycoside hydrolase family 9 from Cytophaga hutchinsonii. Appl Microbiol Biotechnol 104, 7051–7066 (2020). https://doi.org/10.1007/s00253-020-10731-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10731-8