Abstract
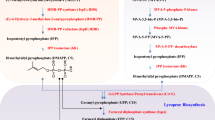
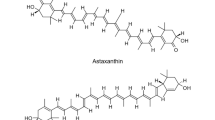
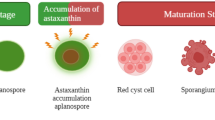
Astaxanthin is a natural pigment, known for its strong antioxidant activity and numerous health benefits to human and animals. Its antioxidant activity is known to be substantially greater than β-carotene and about a thousand times more effective than vitamin E. The potential health benefits have generated a growing commercial interest, and the escalating demand has prompted the exploration of alternative supply chain. Astaxanthin naturally occurs in many sea creatures such as trout, shrimp, and microalgae, some fungi, bacteria, and flowering plants, acting to protect hosts against environmental stress and adverse conditions. Due to the rapid growth and simple growth medium requirement, microbes, such as the microalga, Haematococcus pluvialis, and the fungus Xanthophyllomyces dendrorhous, have been developed to produce astaxanthin. With advances in metabolic engineering, non-carotenogenic microbes, such as Escherichia coli and Saccharomyces cerevisiae, have been purposed to produce astaxanthin and significant progress has been achieved. Here, we review the recent achievements in microbial astaxanthin biosynthesis (with reference to metabolic engineering strategies) and extraction methods, current challenges (technical and regulatory), and commercialization outlook. Due to greenness, sustainability, and dramatic cost reduction, we envision microbial synthesis of astaxanthin offers an alternative means of production (e.g. chemical synthesis) in the near future.




Similar content being viewed by others
References
Ajikumar PK, Tyo K, Carlsen S, Mucha O, Phon TH, Stephanopoulos G (2008) Terpenoids: opportunities for biosynthesis of natural product drugs using engineered microorganisms. Mol Pharm 5(2):167–190. https://doi.org/10.1021/mp700151b
Alvarez V, Rodriguez-Saiz M, de la Fuente JL, Gudina EJ, Godio RP, Martin JF, Barredo JL (2006) The crtS gene of Xanthophyllomyces dendrorhous encodes a novel cytochrome-P450 hydroxylase involved in the conversion of beta-carotene into astaxanthin and other xanthophylls. Fungal Genet Biol 43(4):261–272. https://doi.org/10.1016/j.fgb.2005.12.004
Ambati RR, Phang SM, Ravi S, Aswathanarayana RG (2014) Astaxanthin: sources, extraction, stability, biological activities and its commercial applications--a review. Mar Drugs 12(1):128–152. https://doi.org/10.3390/md12010128
Barredo JL, García-Estrada C, Kosalkova K, Barreiro C (2017) Biosynthesis of astaxanthin as a main carotenoid in the heterobasidiomycetous yeast Xanthophyllomyces dendrorhous. Journal of Fungi 3(3):44
Bauer A, Minceva M (2019) Direct extraction of astaxanthin from the microalgae Haematococcus pluvialis using liquid–liquid chromatography. RSC Advances 9(40):22779–22789. https://doi.org/10.1039/c9ra03263k
Breitenbach J, Misawa N, Kajiwara S, Sandmann G (1996) Expression in Escherichia coli and properties of the carotene ketolase from Haematococcus pluvialis. FEMS Microbiol Lett 140(2–3):241–246. https://doi.org/10.1016/0378-1097(96)00187-5
Capelli B, Jenkins U, Cysewski GR (2013) Role of astaxanthin in sports nutrition. In: Bagchi D, Nair S, Sen CK (eds) Nutrition and enhanced sports performance. Academic Press, San Diego, pp 465–471
Chen X, Zhang C, Lindley ND (2020) Metabolic engineering strategies for sustainable terpenoid flavor and fragrance synthesis. J Agric Food Chem. https://doi.org/10.1021/acs.jafc.9b06203
Chi S, He Y, Ren J, Su Q, Liu X, Chen Z, Wang M, Li Y, Li J (2015) Overexpression of a bifunctional enzyme, CrtS, enhances astaxanthin synthesis through two pathways in Phaffia rhodozyma. Microb Cell Fact 14:90. https://doi.org/10.1186/s12934-015-0279-4
Choi SK, Harada H, Matsuda S, Misawa N (2007a) Characterization of two beta-carotene ketolases, CrtO and CrtW, by complementation analysis in Escherichia coli. Appl Microbiol Biotechnol 75(6):1335–1341. https://doi.org/10.1007/s00253-007-0967-z
Choi SK, Kim JH, Park YS, Kim YJ, Chang HI (2007b) An efficient method for the extraction of astaxanthin from the red yeast Xanthophyllomyces dendrorhous. J Microbiol Biotechnol 17(5):847–852
Christian D, Zhang J, Sawdon AJ, Peng CA (2018) Enhanced astaxanthin accumulation in Haematococcus pluvialis using high carbon dioxide concentration and light illumination. Bioresour Technol 256:548–551. https://doi.org/10.1016/j.biortech.2018.02.074
Clomburg JM, Crumbley AM, Gonzalez R (2017) Industrial biomanufacturing: the future of chemical production. Science 355(6320):aag0804. https://doi.org/10.1126/science.aag0804
Cunningham FX Jr, Gantt E (2011) Elucidation of the pathway to astaxanthin in the flowers of Adonis aestivalis. The Plant cell 23(8):3055–3069. https://doi.org/10.1105/tpc.111.086827
Cunningham FX Jr, Pogson B, Sun Z, McDonald KA, DellaPenna D, Gantt E (1996) Functional analysis of the beta and epsilon lycopene cyclase enzymes of Arabidopsis reveals a mechanism for control of cyclic carotenoid formation. The Plant cell 8(9):1613–1626. https://doi.org/10.1105/tpc.8.9.1613
Denby CM, Li RA, Vu VT, Costello Z, Lin W, Chan LJG, Williams J, Donaldson B, Bamforth CW, Petzold CJ, Scheller HV, Martin HG, Keasling JD (2018) Industrial brewing yeast engineered for the production of primary flavor determinants in hopped beer. Nat Commun 9(1):965. https://doi.org/10.1038/s41467-018-03293-x
Ding W, Zhao Y, Xu JW, Zhao P, Li T, Ma H, Reiter RJ, Yu X (2018) Melatonin: a multifunctional molecule that triggers defense responses against high light and nitrogen starvation stress in Haematococcus pluvialis. J Agric Food Chem 66(29):7701–7711. https://doi.org/10.1021/acs.jafc.8b02178
Fakhri S, Abbaszadeh F, Dargahi L, Jorjani M (2018) Astaxanthin: a mechanistic review on its biological activities and health benefits. Pharmacol Res 136:1–20. https://doi.org/10.1016/j.phrs.2018.08.012
Gao S, Tong Y, Zhu L, Ge M, Zhang Y, Chen D, Jiang Y, Yang S (2017) Iterative integration of multiple-copy pathway genes in Yarrowia lipolytica for heterologous beta-carotene production. Metab Eng 41:192–201. https://doi.org/10.1016/j.ymben.2017.04.004
Gassel S, Breitenbach J, Sandmann G (2014) Genetic engineering of the complete carotenoid pathway towards enhanced astaxanthin formation in Xanthophyllomyces dendrorhous starting from a high-yield mutant. Appl Microbiol Biotechnol 98(1):345–350. https://doi.org/10.1007/s00253-013-5358-z
Grenfell-Lee D, Zeller S, Cardoso R, Pucaj K (2014) The safety of beta-carotene from Yarrowia lipolytica. Food Chem Toxicol 65:1–11. https://doi.org/10.1016/j.fct.2013.12.010
Han D, Li Y, Hu Q (2013) Astaxanthin in microalgae: pathways, functions and biotechnological implications. Algae 28(2):131–147. https://doi.org/10.4490/algae.2013.28.2.131
Hasunuma T, Takaki A, Matsuda M, Kato Y, Vavricka CJ, Kondo A (2019) Single-stage astaxanthin production enhances the nonmevalonate pathway and photosynthetic central metabolism in Synechococcus sp. PCC 7002. ACS Synth Biol 8(12):2701–2709. https://doi.org/10.1021/acssynbio.9b00280
Henke NA, Heider SA, Peters-Wendisch P, Wendisch VF (2016) Production of the marine carotenoid astaxanthin by metabolically engineered Corynebacterium glutamicum. Mar Drugs 14(7):124–121. https://doi.org/10.3390/md14070124
Hong ME, Choi YY, Sim SJ (2016) Effect of red cyst cell inoculation and iron(II) supplementation on autotrophic astaxanthin production by Haematococcus pluvialis under outdoor summer conditions. J Biotechnol 218:25–33. https://doi.org/10.1016/j.jbiotec.2015.11.019
Hong ME, Hwang SK, Chang WS, Kim BW, Lee J, Sim SJ (2015) Enhanced autotrophic astaxanthin production from Haematococcus pluvialis under high temperature via heat stress-driven Haber-Weiss reaction. Appl Microbiol Biotechnol 99(12):5203–5215. https://doi.org/10.1007/s00253-015-6440-5
Ide T, Hoya M, Tanaka T, Harayama S (2012) Enhanced production of astaxanthin in Paracoccus sp. strain N-81106 by using random mutagenesis and genetic engineering. Biochem Eng J 65:37–43. https://doi.org/10.1016/j.bej.2012.03.015
Jin J, Wang Y, Yao M, Gu X, Li B, Liu H, Ding M, Xiao W, Yuan Y (2018) Astaxanthin overproduction in yeast by strain engineering and new gene target uncovering. Biotechnol Biofuels 11(1):230. https://doi.org/10.1186/s13068-018-1227-4
Khoo KS, Lee SY, Ooi CW, Fu X, Miao X, Ling TC, Show PL (2019) Recent advances in biorefinery of astaxanthin from Haematococcus pluvialis. Bioresour Technol 288:121606. https://doi.org/10.1016/j.biortech.2019.121606
Kildegaard KR, Adiego-Perez B, Domenech Belda D, Khangura JK, Holkenbrink C, Borodina I (2017) Engineering of Yarrowia lipolytica for production of astaxanthin. Synth Syst Biotechnol 2(4):287–294. https://doi.org/10.1016/j.synbio.2017.10.002
Kirby J, Keasling JD (2009) Biosynthesis of plant isoprenoids: perspectives for microbial engineering. Annu Rev Plant Biol 60(1):335–355. https://doi.org/10.1146/annurev.arplant.043008.091955
Koller M, Muhr A, Braunegg G (2014) Microalgae as versatile cellular factories for valued products. Algal Research 6:52–63. https://doi.org/10.1016/j.algal.2014.09.002
Krause W, Henrich K, Paust J, Ernst H (1997) Preparation of astaxanthin. Available online at: https://www.google.com/patents/US5654488.
Krichnavaruk S, Shotipruk A, Goto M, Pavasant P (2008) Supercritical carbon dioxide extraction of astaxanthin from Haematococcus pluvialis with vegetable oils as co-solvent. Bioresour Technol 99(13):5556–5560. https://doi.org/10.1016/j.biortech.2007.10.049
Lane MM, Morrissey JP (2010) Kluyveromyces marxianus: a yeast emerging from its sister’s shadow. Fungal Biol Rev 24(1–2):17–26. https://doi.org/10.1016/j.fbr.2010.01.001
Larroude M, Celinska E, Back A, Thomas S, Nicaud JM, Ledesma-Amaro R (2018) A synthetic biology approach to transform Yarrowia lipolytica into a competitive biotechnological producer of beta-carotene. Biotechnol Bioeng 115(2):464–472. https://doi.org/10.1002/bit.26473
Lemuth K, Steuer K, Albermann C (2011) Engineering of a plasmid-free Escherichia coli strain for improved in vivo biosynthesis of astaxanthin. Microb Cell Fact 10(1):29. https://doi.org/10.1186/1475-2859-10-29
Liao JC, Mi L, Pontrelli S, Luo S (2016) Fuelling the future: microbial engineering for the production of sustainable biofuels. Nat Rev Microbiol 14(5):288–304. https://doi.org/10.1038/nrmicro.2016.32
Lin YJ, Chang JJ, Lin HY, Thia C, Kao YY, Huang CC, Li WH (2017) Metabolic engineering a yeast to produce astaxanthin. Bioresour Technol 245(Pt A):899–905. https://doi.org/10.1016/j.biortech.2017.07.116
Liu YH, Alimujiang A, Wang X, Luo SW, Balamurugan S, Yang WD, Liu JS, Zhang L, Li HY (2019) Ethanol induced jasmonate pathway promotes astaxanthin hyperaccumulation in Haematococcus pluvialis. Bioresour Technol 289:121720. https://doi.org/10.1016/j.biortech.2019.121720
Mano J, Liu N, Hammond JH, Currie DH, Stephanopoulos G (2020) Engineering Yarrowia lipolytica for the utilization of acid whey. Metab Eng 57:43–50. https://doi.org/10.1016/j.ymben.2019.09.010
Matthaus F, Ketelhot M, Gatter M, Barth G (2014) Production of lycopene in the non-carotenoid-producing yeast Yarrowia lipolytica. Appl Environ Microbiol 80(5):1660–1669. https://doi.org/10.1128/AEM.03167-13
Meadows AL, Hawkins KM, Tsegaye Y, Antipov E, Kim Y, Raetz L, Dahl RH, Tai A, Mahatdejkul-Meadows T, Xu L, Zhao L, Dasika MS, Murarka A, Lenihan J, Eng D, Leng JS, Liu CL, Wenger JW, Jiang H, Chao L, Westfall P, Lai J, Ganesan S, Jackson P, Mans R, Platt D, Reeves CD, Saija PR, Wichmann G, Holmes VF, Benjamin K, Hill PW, Gardner TS, Tsong AE (2016) Rewriting yeast central carbon metabolism for industrial isoprenoid production. Nature 537(7622):694–697. https://doi.org/10.1038/nature19769
Misawa N, Satomi Y, Kondo K, Yokoyama A, Kajiwara S, Saito T, Ohtani T, Miki W (1995) Structure and functional analysis of a marine bacterial carotenoid biosynthesis gene cluster and astaxanthin biosynthetic pathway proposed at the gene level. J Bacteriol 177(22):6575–6584. https://doi.org/10.1128/jb.177.22.6575-6584.1995
Molino A, Rimauro J, Casella P, Cerbone A, Larocca V, Chianese S, Karatza D, Mehariya S, Ferraro A, Hristoforou E, Musmarra D (2018) Extraction of astaxanthin from microalga Haematococcus pluvialis in red phase by using generally recognized as safe solvents and accelerated extraction. J Biotechnol 283:51–61. https://doi.org/10.1016/j.jbiotec.2018.07.010
Ng QX, De Deyn MLZQ, Loke W, Foo NX, Chan HW, Yeo WS (2020) Effects of astaxanthin supplementation on skin health: a systematic review of clinical studies. Journal of Dietary Supplements:1–14 doi:https://doi.org/10.1080/19390211.2020.1739187
Nobre B, Marcelo F, Passos R, Beirão L, Palavra A, Gouveia L, Mendes R (2006) Supercritical carbon dioxide extraction of astaxanthin and other carotenoids from the microalga Haematococcus pluvialis. Eur Food Res Technol 223(6):787–790. https://doi.org/10.1007/s00217-006-0270-8
Nouchi R, Suiko T, Kimura E, Takenaka H, Murakoshi M, Uchiyama A, Aono M, Kawashima R (2020) Effects of lutein and astaxanthin intake on the improvement of cognitive functions among healthy adults: a systematic review of randomized controlled trials. Nutrients 12(3). https://doi.org/10.3390/nu12030617
Novoveská L, Ross ME, Stanley MS, Pradelles R, Wasiolek V, Sassi J-F (2019) Microalgal carotenoids: a review of production, current markets, regulations, and future direction. Mar Drugs 17(11):640. https://doi.org/10.3390/md17110640
Panis G, Carreon JR (2016) Commercial astaxanthin production derived by green alga Haematococcus pluvialis: a microalgae process model and a techno-economic assessment all through production line. Algal Research 18:175–190. https://doi.org/10.1016/j.algal.2016.06.007
Park SY, Binkley RM, Kim WJ, Lee MH, Lee SY (2018) Metabolic engineering of Escherichia coli for high-level astaxanthin production with high productivity. Metab Eng 49:105–115. https://doi.org/10.1016/j.ymben.2018.08.002
Rajesh K, Rohit MV, Venkata Mohan S (2017) Chapter 7 - Microalgae-based carotenoids production. In: Rastogi RP, Madamwar D, Pandey A (eds) Algal green chemistry. Elsevier, Amsterdam, pp 139–147
Sanzo GD, Mehariya S, Martino M, Larocca V, Casella P, Chianese S, Musmarra D, Balducchi R, Molino A (2018) Supercritical carbon dioxide extraction of astaxanthin, lutein, and fatty acids from Haematococcus pluvialis microalgae. Mar Drugs 16(9):334. https://doi.org/10.3390/md16090334
Scaife MA, Burja AM, Wright PC (2009) Characterization of cyanobacterial beta-carotene ketolase and hydroxylase genes in Escherichia coli, and their application for astaxanthin biosynthesis. Biotechnol Bioeng 103(5):944–955. https://doi.org/10.1002/bit.22330
Scaife MA, Ma CA, Ninlayarn T, Wright PC, Armenta RE (2012) Comparative analysis of beta-carotene hydroxylase genes for astaxanthin biosynthesis. J Nat Prod 75(6):1117–1124. https://doi.org/10.1021/np300136t
Schmidt I, Schewe H, Gassel S, Jin C, Buckingham J, Humbelin M, Sandmann G, Schrader J (2011) Biotechnological production of astaxanthin with Phaffia rhodozyma/Xanthophyllomyces dendrorhous. Appl Microbiol Biotechnol 89(3):555–571. https://doi.org/10.1007/s00253-010-2976-6
Schwartz C, Frogue K, Misa J, Wheeldon I (2017) Host and pathway engineering for enhanced lycopene biosynthesis in Yarrowia lipolytica. Front Microbiol 8:2233. https://doi.org/10.3389/fmicb.2017.02233
Sekova VY, Isakova EP, Deryabina YI (2015) Biotechnological applications of the extremophilic yeast Yarrowia lipolytica (review). Appl Biochem Microbiol 51(3):278–291. https://doi.org/10.1134/S0003683815030151
Shah MM, Liang Y, Cheng JJ, Daroch M (2016) Astaxanthin-producing green microalga Haematococcus pluvialis: From Single Cell to High Value Commercial Products. Front Plant Sci 7:531. https://doi.org/10.3389/fpls.2016.00531
Stanbury PF, Whitaker A, Hall SJ (2017) Chapter 10 - The recovery and purification of fermentation products. In: Stanbury PF, Whitaker A, Hall SJ (eds) Principles of fermentation technology, Third edn. Butterworth-Heinemann, Oxford, pp 619–686
Tramontin LRR, Kildegaard KR, Sudarsan S, Borodina I (2019) Enhancement of astaxanthin biosynthesis in oleaginous yeast Yarrowia lipolytica via microalgal pathway. Microorganisms 7(10):472–417. https://doi.org/10.3390/microorganisms7100472
Ukibe K, Hashida K, Yoshida N, Takagi H (2009) Metabolic engineering of Saccharomyces cerevisiae for astaxanthin production and oxidative stress tolerance. Appl Environ Microbiol 75(22):7205–7211. https://doi.org/10.1128/AEM.01249-09
Ukibe K, Katsuragi T, Tani Y, Takagi H (2008) Efficient screening for astaxanthin-overproducing mutants of the yeast Xanthophyllomyces dendrorhous by flow cytometry. FEMS Microbiol Lett 286(2):241–248. https://doi.org/10.1111/j.1574-6968.2008.01278.x
Verdoes JC, Sandmann G, Visser H, Diaz M, van Mossel M, van Ooyen AJ (2003) Metabolic engineering of the carotenoid biosynthetic pathway in the yeast Xanthophyllomyces dendrorhous (Phaffia rhodozyma). Appl Environ Microbiol 69(7):3728–3738. https://doi.org/10.1128/aem.69.7.3728-3738.2003
Visser H, van Ooyen AJ, Verdoes JC (2003) Metabolic engineering of the astaxanthin-biosynthetic pathway of Xanthophyllomyces dendrorhous. FEMS Yeast Res 4(3):221–231. https://doi.org/10.1016/S1567-1356(03)00158-2
Wang B, Pan X, Jia J, Xiong W, Manirafasha E, Ling X, Yinghua L (2019a) Strategy and regulatory mechanisms of glutamate feeding to enhance astaxanthin yield in Xanthophyllomyces dendrorhous. Enzyme Microb Technol 125:45–52. https://doi.org/10.1016/j.enzmictec.2019.02.010
Wang F, Gao B, Wu M, Huang L, Zhang C (2019b) A novel strategy for the hyper-production of astaxanthin from the newly isolated microalga Haematococcus pluvialis JNU35. Algal Research 39. https://doi.org/10.1016/j.algal.2019.101466
Wang R, Gu X, Yao M, Pan C, Liu H, Xiao W, Wang Y, Yuan Y (2017) Engineering of β-carotene hydroxylase and ketolase for astaxanthin overproduction in Saccharomyces cerevisiae. Front Chem Sci Eng 11(1):89–99. https://doi.org/10.1007/s11705-017-1628-0
Watanabe K, Arafiles KHV, Higashi R, Okamura Y, Tajima T, Matsumura Y, Nakashimada Y, Matsuyama K, Aki T (2018) Isolation of high carotenoid-producing Aurantiochytrium sp. mutants and improvement of astaxanthin productivity using metabolic information. Journal of oleo science 67(5):571–578. https://doi.org/10.5650/jos.ess17230
Yamamoto K, Hara KY, Morita T, Nishimura A, Sasaki D, Ishii J, Ogino C, Kizaki N, Kondo A (2016) Enhancement of astaxanthin production in Xanthophyllomyces dendrorhous by efficient method for the complete deletion of genes. Microb Cell Fact 15(1):155. https://doi.org/10.1186/s12934-016-0556-x
Yamashita E (2015) Let astaxanthin be thy medicine. PharmaNutrition 3(4):115–122. https://doi.org/10.1016/j.phanu.2015.09.001
Yokoyama A, Shizuri Y, Misawa N (1998) Production of new carotenoids, astaxanthin glucosides, by Escherichia coli transformants carrying carotenoid biosynthesis genes. Tetrahedron Lett 39(22):3709–3712. https://doi.org/10.1016/s0040-4039(98)00542-5
Zelcbuch L, Antonovsky N, Bar-Even A, Levin-Karp A, Barenholz U, Dayagi M, Liebermeister W, Flamholz A, Noor E, Amram S, Brandis A, Bareia T, Yofe I, Jubran H, Milo R (2013) Spanning high-dimensional expression space using ribosome-binding site combinatorics. Nucleic Acids Res 41(9):e98. https://doi.org/10.1093/nar/gkt151
Zhang C (2018) Biosynthesis of carotenoids and apocarotenoids by microorganisms and their industrial potential. Progress in Carotenoid Research:85
Zhang C, Chen X, Lindley ND, Too HP (2018a) A “plug-n-play” modular metabolic system for the production of apocarotenoids. Biotechnol Bioeng 115(1):174–183. https://doi.org/10.1002/bit.26462
Zhang C, Chen X, Stephanopoulos G, Too HP (2016) Efflux transporter engineering markedly improves amorphadiene production in Escherichia coli. Biotechnol Bioeng 113(8):1755–1763. https://doi.org/10.1002/bit.25943
Zhang C, Chen X, Zou R, Zhou K, Stephanopoulos G, Too HP (2013) Combining genotype improvement and statistical media optimization for isoprenoid production in E. coli. PLoS ONE 8(10):e75164. https://doi.org/10.1371/journal.pone.0075164
Zhang C, Seow VY, Chen X, Too HP (2018b) Multidimensional heuristic process for high-yield production of astaxanthin and fragrance molecules in Escherichia coli. Nat Commun 9(1):1858. https://doi.org/10.1038/s41467-018-04211-x
Zhang C, Too HP (2019) Revalorizing lignocellulose for the production of natural pharmaceuticals and other high value bioproducts. Curr Med Chem 26(14):2475–2484. https://doi.org/10.2174/0929867324666170912095755
Zhang Z, Sun D, Zhang Y, Chen F (2019) Glucose triggers cell structure changes and regulates astaxanthin biosynthesis in Chromochloris zofingiensis. Algal Research 39:101455. https://doi.org/10.1016/j.algal.2019.101455
Zhou P, Li M, Shen B, Yao Z, Bian Q, Ye L, Yu H (2019) Directed coevolution of beta-carotene ketolase and hydroxylase and its application in temperature-regulated biosynthesis of astaxanthin. J Agric Food Chem 67(4):1072–1080. https://doi.org/10.1021/acs.jafc.8b05003
Zhou P, Xie W, Li A, Wang F, Yao Z, Bian Q, Zhu Y, Yu H, Ye L (2017) Alleviation of metabolic bottleneck by combinatorial engineering enhanced astaxanthin synthesis in Saccharomyces cerevisiae. Enzyme Microb Technol 100:28–36. https://doi.org/10.1016/j.enzmictec.2017.02.006
Zhou P, Ye L, Xie W, Lv X, Yu H (2015) Highly efficient biosynthesis of astaxanthin in Saccharomyces cerevisiae by integration and tuning of algal crtZ and bkt. Appl Microbiol Biotechnol 99(20):8419–8428. https://doi.org/10.1007/s00253-015-6791-y
Author information
Authors and Affiliations
Contributions
C.Z. and X.C. proposed the initial outline of the manuscript. C.Z. and H.P.T. wrote and reviewed the manuscript. All authors contributed to the discussion and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This mini-review does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Points
• Growing commercial interests and demand of astaxanthin due to its health benefits
• Recent achievements in astaxanthin biosynthesis and extraction in microbes
• A summary of metabolic engineering strategies in astaxanthin biosynthesis
• Discussion about current challenges and outlook of microbial-derived astaxanthin
Rights and permissions
About this article
Cite this article
Zhang, C., Chen, X. & Too, HP. Microbial astaxanthin biosynthesis: recent achievements, challenges, and commercialization outlook. Appl Microbiol Biotechnol 104, 5725–5737 (2020). https://doi.org/10.1007/s00253-020-10648-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10648-2