Abstract
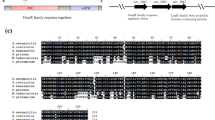
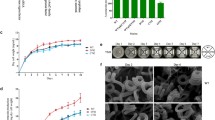
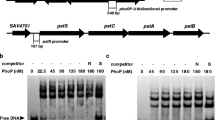
Streptomyces avermitilis is well known as the producer of anthelmintic agent avermectins, which are widely used in agriculture, veterinary medicine, and human medicine. aveI encodes a TetR-family regulator, which is the homolog of AtrA. It was reported that deletion of aveI caused enhanced avermectin production. In this study, we investigated the regulatory function of the AveI in S. avermitilis. By binding to the 15-nt palindromic sequence in the promoter regions, AveI directly regulates at least 35 genes. AveI represses avermectin production by directly regulating the transcription of the cluster-situated regulator gene aveR and structural genes aveA1, aveA3, and aveD. AveI represses oligomycin production by repressing the CSR gene olmRII and structural genes olmC. AveI activates melanin biosynthesis by activating the expression of melC1C2 operon. AveI activates morphological differentiation by activating the expression of ssgR and ssgD genes, repressing the expression of wblI gene. Besides, AveI regulates many genes involved in primary metabolism, including substrates transport, the metabolism of amino acids, lipids, and carbohydrates. Therefore, AveI functions as a global regulator in S. avermitilis, controls not only secondary metabolism and morphological differentiation, but also primary metabolism.








Similar content being viewed by others
References
Chen L, Lu Y, Chen J, Zhang W, Shu D, Qin Z, Yang S, Jiang W (2008) Characterization of a negative regulator AveI for avermectin biosynthesis in Streptomyces avermitilis NRRL8165. Appl Microbiol Biotechnol 80(2):277–286
Chen L, Chen J, Jiang Y, Zhang W, Jiang W, Lu Y (2009) Transcriptomics analyses reveal global roles of the regulator AveI in Streptomyces avermitilis. FEMS Microbiol Lett 298(2):199–207
Choudoir MJ, Pepe-Ranney C, Buckley DH (2018) Diversification of secondary metabolite biosynthetic gene clusters coincides with lineage divergence in Streptomyces. Antibiotics (Basel) 7(1):12
Craney A, Ahmed S, Nodwell J (2013) Towards a new science of secondary metabolism. J Antibiot 66(7):387–400
Duong CT, Lee HN, Choi SS, Lee SY, Kim ES (2009) Functional expression of SAV3818, a putative TetR-family transcriptional regulatory gene from Streptomyces avermitilis, stimulates antibiotic production in Streptomyces species. J Microbiol Biotechnol 19(2):136–139
Fowler-Goldsworthy K, Gust B, Mouz S, Chandra G, Findlay KC, Chater KF (2011) The actinobacteria-specific gene wblA controls major developmental transitions in Streptomyces coelicolor A3(2). Microbiol 157(Pt 5):1312–1328
Guo J, Zhao J, Li L, Chen Z, Wen Y, Li J (2010) The pathway-specific regulator AveR from Streptomyces avermitilis positively regulates avermectin production while it negatively affects oligomycin biosynthesis. Mol Gen Genet 283(2):123–133
Guo J, Zhang X, Luo S, He F, Chen Z, Wen Y, Li J (2013) A novel TetR family transcriptional regulator, SAV576, negatively controls avermectin biosynthesis in Streptomyces avermitilis. PLoS One 8(8):e71330
Guo J, Zhang X, Chen Z, Wen Y, Li J (2014) Two adjacent and similar TetR family transcriptional regulator genes, SAV577 and SAV576, co-regulate avermectin production in Streptomyces avermitilis. PLoS One 9(6):e99224
He JM, Zhu H, Zheng GS, Liu PP, Wang J, Zhao GP, Zhu GQ, Jiang WH, Lu YH (2016) Direct involvement of the master nitrogen metabolism regulator GlnR in antibiotic biosynthesis in Streptomyces. J Biol Chem 291(51):26443–26454
Hirano S, Tanaka K, Ohnishi Y, Horinouchi S (2008) Conditionally positive effect of the TetR-family transcriptional regulator AtrA on streptomycin production by Streptomyces griseus. Microbiol 154(Pt 3):905–914
Ikeda H, Omura S (1997) Avermectin biosynthesis. Chem Rev 97(7):2591–2610
Ikeda H, Kazuo SY, Omura S (2014) Genome mining of the Streptomyces avermitilis genome and development of genome-minimized hosts for heterologous expression of biosynthetic gene clusters. J Ind Microbiol Biotechnol 41(2):233–250
Jiang L, Liu Y, Wang P, Wen Y, Song Y, Chen Z, Li J (2011) Inactivation of the extracytoplasmic function sigma factor Sig6 stimulates avermectin production in Streptomyces avermitilis. Biotechnol Lett 33(10):1955–1961
Kim SH, Traag BA, Hasan AH, McDowall KJ, Kim BG, van Wezel GP (2015) Transcriptional analysis of the cell division-related ssg genes in Streptomyces coelicolor reveals direct control of ssgR by AtrA. Antonie Van Leeuwenhoek 108(1):201–213
Kitani S, Ikeda H, Sakamoto T, Noguchi S, Nihira T (2009) Characterization of a regulatory gene, aveR, for the biosynthesis of avermectin in Streptomyces avermitilis. Appl Microbiol Biotechnol 82(6):1089–1096
Li X, Yu T, He Q, McDowall KJ, Jiang B, Jiang Z, Wu L, Li G, Li Q, Wang S, Shi Y, Wang L, Hong B (2015) Binding of a biosynthetic intermediate to AtrA modulates the production of lidamycin by Streptomyces globisporus. Mol Microbiol 96(6):1257–1271
Liu G, Chater KF, Chandra G, Niu G, Tan H (2013a) Molecular regulation of antibiotic biosynthesis in Streptomyces. Microbiol Mol Biol Rev 77(1):112–143
Liu Y, Yan T, Jiang L, Wen Y, Song Y, Chen Z, Li J (2013b) Characterization of SAV7471, a TetR-family transcriptional regulator involved in the regulation of coenzyme A metabolism in Streptomyces avermitilis. J Bacteriol 195(19):4365–4372
Liu W, Zhang Q, Guo J, Chen Z, Li J, Wen Y (2015) Increasing avermectin production in Streptomyces avermitilis by manipulating the expression of a novel TetR-family regulator and its target gene product. Appl Environ Microbiol 81(15):5157–5173
Liu X, Cheng Y, Lyu M, Wen Y, Song Y, Chen Z, Li J (2017) Redox-sensing regulator Rex regulates aerobic metabolism, morphological differentiation, and avermectin production in Streptomyces avermitilis. Sci Rep 7:44567
Luo S, Sun D, Zhu J, Chen Z, Wen Y, Li J (2014) An extracytoplasmic function sigma factor, sigma(25), differentially regulates avermectin and oligomycin biosynthesis in Streptomyces avermitilis. Appl Microbiol Biotechnol 98(16):7097–7112
Macneil DJ, Klapko LM (1987) Transformation of Streptomyces avermitilis by plasmid DNA. J Ind Microbiol 2(4):209–218
Mao XM, Luo S, Zhou RC, Wang F, Yu P, Sun N, Chen XX, Tang Y, Li YQ (2015) Transcriptional regulation of the daptomycin gene cluster in Streptomyces roseosporus by an autoregulator, AtrA. J Biol Chem 290(12):7992–8001
Molle V, Palframan WJ, Findlay KC, Buttner MJ (2000) WhiD and WhiB, homologous proteins required for different stages of sporulation in Streptomyces coelicolor A3(2). J Bacteriol 182(5):1286–1295
Noens EE, Mersinias V, Traag BA, Smith CP, Koerten HK, van Wezel GP (2005) SsgA-like proteins determine the fate of peptidoglycan during sporulation of Streptomyces coelicolor. Mol Microbiol 58(4):929–944
Noens EE, Mersinias V, Willemse J, Traag BA, Laing E, Chater KF, Smith CP, Koerten HK, van Wezel GP (2007) Loss of the controlled localization of growth stage-specific cell-wall synthesis pleiotropically affects developmental gene expression in an ssgA mutant of Streptomyces coelicolor. Mol Microbiol 64(5):1244–1259
Nothaft H, Rigali S, Boomsma B, Swiatek M, McDowall KJ, van Wezel GP, Titgemeyer F (2010) The permease gene nagE2 is the key to N-acetylglucosamine sensing and utilization in Streptomyces coelicolor and is subject to multi-level control. Mol Microbiol 75(5):1133–1144
Sun D, Zhu J, Chen Z, Li J, Wen Y (2016) SAV742, a novel AraC-family regulator from Streptomyces avermitilis, controls avermectin biosynthesis, cell growth and development. Sci Rep 6:36915
Traag BA, Kelemen GH, Van Wezel GP (2004) Transcription of the sporulation gene ssgA is activated by the IclR-type regulator SsgR in a whi-independent manner in Streptomyces coelicolor A3(2). Mol Microbiol 53(3):985–1000
Uguru GC, Stephens KE, Stead JA, Towle JE, Baumberg S, McDowall KJ (2005) Transcriptional activation of the pathway-specific regulator of the actinorhodin biosynthetic genes in Streptomyces coelicolor. Mol Microbiol 58(1):131–150
van Wezel GP, McDowall KJ (2011) The regulation of the secondary metabolism of Streptomyces: new links and experimental advances. Nat Prod Rep 28(7):1311–1333
Wang W, Tian J, Li L, Ge M, Zhu H, Zheng G, Huang H, Ruan L, Jiang W, Lu Y (2015) Identification of two novel regulatory genes involved in pristinamycin biosynthesis and elucidation of the mechanism for AtrA-p-mediated regulation in Streptomyces pristinaespiralis. Appl Microbiol Biotechnol 99(17):7151–7164
Xu LH, Fushinobu S, Takamatsu S, Wakagi T, Ikeda H, Shoun H (2010) Regio- and stereospecificity of filipin hydroxylation sites revealed by crystal structures of cytochrome P450 105P1 and 105D6 from Streptomyces avermitilis. J Biol Chem 285(22):16844–16853
Yang R, Liu X, Wen Y, Song Y, Chen Z, Li J (2015) The PhoP transcription factor negatively regulates avermectin biosynthesis in Streptomyces avermitilis. Appl Microbiol Biotechnol 99(24):10547–10557
Zhu J, Sun D, Liu W, Chen Z, Li J, Wen Y (2016) AvaR2, a pseudo gamma-butyrolactone receptor homologue from Streptomyces avermitilis, is a pleiotropic repressor of avermectin and avenolide biosynthesis and cell growth. Mol Microbiol 102(4):562–578
Zhu J, Chen Z, Li J, Wen Y (2017) AvaR1, a butenolide-type autoregulator receptor in Streptomyces avermitilis, directly represses avenolide and avermectin biosynthesis and multiple physiological responses. Front Microbiol 8:2577
Funding
This study was supported by grants from the National Natural Science Foundation of China (No. 31470190) and the project for Extramural Scientists of the State Key Laboratory of Agrobiotechnology (No. 2019SKLAB6-5).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 755 kb)
Rights and permissions
About this article
Cite this article
Liu, L., Cheng, Y., Lyu, M. et al. AveI, an AtrA homolog of Streptomyces avermitilis, controls avermectin and oligomycin production, melanogenesis, and morphological differentiation. Appl Microbiol Biotechnol 103, 8459–8472 (2019). https://doi.org/10.1007/s00253-019-10062-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-10062-3