Abstract
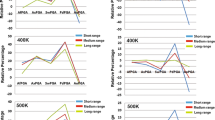
Penicillin G acylase (PGA) catalyzes the hydrolysis of penicillin G to 6-aminopenicillanic acid and phenylacetic acid, which provides the precursor for most semisynthetic penicillins. Most applications rely on PGAs from Gram-negative bacteria. Here we describe the first three crystal structures for PGAs from Gram-positive Bacilli and their utilization in protein engineering experiments for the manipulation of their thermostability. PGAs from Bacillus megaterium (BmPGA, Tm = 56.0 °C), Bacillus thermotolerans (BtPGA, Tm = 64.5 °C), and Bacillus sp. FJAT-27231 (FJAT-PGA, Tm = 74.3 °C) were recombinantly produced with B. megaterium, secreted, purified to apparent heterogeneity, and crystallized. Structures with resolutions of 2.20 Å (BmPGA), 2.27 Å (BtPGA), and 1.36 Å (FJAT-PGA) were obtained. They revealed high overall similarity, reflecting the high identity of up to approx. 75%. Notably, the active center displays a deletion of more than ten residues with respect to PGAs from Gram-negatives. This enlarges the substrate binding site and may indicate a different substrate spectrum. Based on the structures, ten single-chain FJAT-PGAs carrying artificial linkers were produced. However, in all cases, complete linker cleavage was observed. While thermostability remained in the wild-type range, the enzymatic activity dropped between 30 and 60%. Furthermore, four hybrid PGAs carrying subunits from two different enzymes were successfully produced. Their thermostabilities mostly lay between the values of the two mother enzymes. For one PGA increased, enzyme activity was observed. Overall, the three novel PGA structures combined with initial protein engineering experiments provide the basis for establishment of new PGA-based biotechnological processes.







Similar content being viewed by others
References
Adams PD, Afonine PV, Bunkóczi G, Chen VB, Davis IW, Echols N, Headd JJ, Hung LW, Kapral GJ, Grosse-Kunstleve RW, McCoy AJ, Moriarty NW, Oeffner R, Read RJ, Richardson DC, Richardson JS, Terwilliger TC, Zwart PH (2010) PHENIX: a comprehensive python-based system for macromolecular structure solution. Acta Crystallogr Sect D Biol Crystallogr 66:213–221. https://doi.org/10.1107/S0907444909052925
Afonine PV, Grosse-Kunstleve RW, Echols N, Headd JJ, Moriarty NW, Mustyakimov M, Terwilliger TC, Urzhumtsev A, Zwart PH, Adams PD (2012) Towards automated crystallographic structure refinement with phenix.refine. Acta Crystallogr Sect D Biol Crystallogr 68:352–367. https://doi.org/10.1107/S0907444912001308
Berman HM, Westbrook J, Feng Z, Gilliland G, Bhat TN, Weissig H, Shindyalov IN, Bourne PE (2000) The Protein Data Bank. Nucleic Acids Res 28:235–242. https://doi.org/10.1093/nar/28.1.235
Biedendieck R, Borgmeier C, Bunk B, Stammen S, Scherling C, Meinhardt F, Wittmann C, Jahn D (2011) Systems biology of recombinant protein production using Bacillus megaterium, 1st edn. Elsevier Inc
Bradford MM (1976) A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye-binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Burkhardt A, Pakendorf T, Reime B, Meyer J, Fischer P, Stübe N, Panneerselvam S, Lorbeer O, Stachnik K, Warmer M, Rödig P, Göries D, Meents A (2016) Status of the crystallography beamlines at PETRA III. Eur Phys J Plus 131:0–8. doi: https://doi.org/10.1140/epjp/i2016-16056-0
Chand D, Varshney N, Ramasamy S, Panigrahi P, Brannigan JA, Wilkinson AJ, Suresh CG (2015) Structure mediation in substrate binding and post-translational processing of penicillin acylases: information from mutant structures of Kluyvera citrophila penicillin G acylase. Protein Sci 24:1660–1670. https://doi.org/10.1002/pro.2761
Chen VB, Arendall WB, Headd JJ, Keedy DA, Immormino RM, Kapral GJ, Murray LW, Richardson JS, Richardson DC (2010) MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr Sect D Biol Crystallogr 66:12–21. https://doi.org/10.1107/S0907444909042073
Chiang C, Bennett RE (1967) Purification and properties of penicillin amidase from Bacillus megaterium. J Bacteriol 93:302–308
Daumy GO, Danley D, McColl AS, Apostolakos D, Vinick FJ (1985) Experimental evolution of penicillin G acylases from Escherichia coli and Proteus rettgeri. J Bacteriol 163:925–932
Dodson G, Wlodawer A (1998) Catalytic triads and their relatives. Trends Biochem Sci 23:347–352. https://doi.org/10.1016/S0968-0004(98)01254-7
Duggleby H, Tolley S, Hill C, Dodson E (1995) Penicillin acylase has a single-amino-acid catalytic centre. Nature 373:264–268
Edman P, Begg G (1967) A protein sequenator. Eur J Biochem 1:80–91. https://doi.org/10.1285/i20705948v4n2p203
Emsley P, Lohkamp B, Scott WG, Cowtan K (2010) Features and development of coot. Acta Crystallogr Sect D Biol Crystallogr 66:486–501. https://doi.org/10.1107/S0907444910007493
Evans PR, Murshudov GN (2013) How good are my data and what is the resolution? Acta Crystallogr Sect D Biol Crystallogr 69:1204–1214. https://doi.org/10.1107/S0907444913000061
Flores G, Soberón X, Osuna J (2004) Production of a fully functional, permuted single-chain penicillin G acylase. Protein Sci 13:1677–1683. https://doi.org/10.1110/ps.03436604
Fu LL, Xu ZR, Li WF, Shuai JB, Lu P, Hu CX (2007) Protein secretion pathways in Bacillus subtilis: implication for optimization of heterologous protein secretion. Biotechnol Adv 25:1–12. https://doi.org/10.1016/j.biotechadv.2006.08.002
Grote A, Hiller K, Scheer M, Münch R, Nörtemann B, Hempel DC, Jahn D (2005) JCat: a novel tool to adapt codon usage of a target gene to its potential expression host. Nucleic Acids Res 33:526–531. https://doi.org/10.1093/nar/gki376
Hewitt L, Kasche V, Lummer K, Lewis RJ, Murshudov GN, Verma CS, Dodson GG, Wilson KS (2000) Structure of a slow processing precursor penicillin acylase from Escherichia coli reveals the linker peptide blocking the active-site cleft. J Mol Biol 302:887–898. https://doi.org/10.1006/jmbi.2000.4105
Ignatova Z, Hörnle C, Kasche V, Nurk A (2002) Unusual signal peptide directs penicillin amidase from Escherichia coli to the tat translocation machinery. Biochem Biophys Res Commun 291:146–149. https://doi.org/10.1006/bbrc.2002.6420
Jurrus E, Engel D, Star K, Monson K, Brandi J, Felberg LE, Brookes DH, Wilson L, Chen J, Liles K, Chun M, Li P, Gohara DW, Dolinsky T, Konecny R, Koes DR, Nielsen JE, Head-Gordon T, Geng W, Krasny R, Wei GW, Holst MJ, McCammon JA, Baker NA (2018) Improvements to the APBS biomolecular solvation software suite. Protein Sci 27:112–128. https://doi.org/10.1002/pro.3280
Kabsch W (2010) XDS. Acta Crystallogr Sect D Biol Crystallogr 66:125–132. https://doi.org/10.1107/S0907444909047337
Kim HS, Kang TS, Hyun JS, Kang HS (2004) Regulation of penicillin G acylase gene expression in Escherichia coli by repressor PaaX and the cAMP-cAMP receptor protein complex. J Biol Chem 279:33253–33262. https://doi.org/10.1074/jbc.M404348200
Kleywegt GJ (1997) Validation of protein models from C(α) coordinates alone. J Mol Biol 273:371–376. https://doi.org/10.1006/jmbi.1997.1309
Lakowitz A, Krull R, Biedendieck R (2017) Recombinant production of the antibody fragment D1.3 scFv with different Bacillus strains. Microb Cell Factories 16:1–18. https://doi.org/10.1186/s12934-017-0625-9
Marešová H, Plačková M, Grulich M, Kyslík P (2014) Current state and perspectives of penicillin G acylase-based biocatalyses. Appl Microbiol Biotechnol 98:2867–2879. https://doi.org/10.1007/s00253-013-5492-7
McDonough M, Klei HE, Kelly J (1999) Crystal structure of penicillin G acylase from the Bro1 mutant strain of Providencia rettgeri. Protein Sci 8:1971–1981. https://doi.org/10.1110/ps.8.10.1971
McCoy AJ, Grosse-Kunstleve RW, Adams PD, Winn MD, Storoni LC, Read RJ (2007) Phaser crystallographic software. J Appl Crystallogr 40:658–674. https://doi.org/10.1107/S0021889807021206
McVey CE, M a W, Dodson GG, Wilson KS, J a B (2001) Crystal structures of penicillin acylase enzyme-substrate complexes: structural insights into the catalytic mechanism. J Mol Biol 313:139–150. https://doi.org/10.1006/jmbi.2001.5043
Merino E, Balbás P, Recillas F, Becerril B, Valle F, Bolivar F (1992) Carbon regulation and the role in nature of the Escherichia coli penicillin acylase (pac) gene. Mol Microbiol 6:2175–2182. https://doi.org/10.1111/j.1365-2958.1992.tb01391.x
Panbangred W, Weeradechapon K, Udomvaraphant S, Fujiyama K, Meevootisom V (2000) High expression of the penicillin G acylase gene (pac) from Bacillus megaterium UN1 in its own pac minus mutant. J Appl Microbiol 89:152–157. https://doi.org/10.1046/j.1365-2672.2000.01093.x
Pantoliano MW, Petrella EC, Kwasnoski JD, Lobanov VS, Myslik J, Graf E, Carver T, Asel E, Springer BA, Lane P, Salemme FR (2001) High-density miniaturized thermal shift assays as a general strategy for drug discovery. J Biomol Screen 6:429–440
Parashar D, Satyanarayana T (2016) A chimeric α-amylase engineered from Bacillus acidicola and Geobacillus thermoleovorans with improved thermostability and catalytic efficiency. J Ind Microbiol Biotechnol 43:473–484. https://doi.org/10.1007/s10295-015-1721-7
Petersen TN, Brunak S, Von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8:785–786. https://doi.org/10.1038/nmeth.1701
Rajendran K, Sekar S, Mahadevan S, Kumar Shanmugam B, Jeyaprakash R, Paramasamy G, Mandal AB (2014) Biological real-time reaction calorimeter studies for the production of penicillin G acylase from Bacillus badius. Appl Biochem Biotechnol 172:3736–3747. https://doi.org/10.1007/s12010-014-0800-y
Retnoningrum DS, Rahayu AP, Mulyanti D, Dita A, Valerius O, Ismaya WT (2016) Unique characteristics of recombinant hybrid manganese superoxide dismutase from Staphylococcus equorum and S. saprophyticus. Protein J 35:136–144. https://doi.org/10.1007/s10930-016-9650-5
Righetti P, Gianazza E, Gelfi C, Chairi M (1990) In: Hames B, Rickwood D (eds) 2nd editioGel electrophoresis of proteins: a practical approach. IRL Press - Oxford University Press, New York, pp 149–214
Rojviriya C, Pratumrat T, Saper MA, Yuvaniyama J (2011) Improved X-ray diffraction from Bacillus megaterium penicillin G acylase crystals through long cryosoaking dehydration. Acta Crystallogr Sect F Struct Biol Cryst Commun 67:1570–1574. https://doi.org/10.1107/S1744309111040462
Sasajima Y, Kohama Y, Kojima-Misaizu M, Kurokawa N, Hara Y, Dong J, Ihara M, Ueda H (2014) Simultaneous retention of thermostability and specific activity in chimeric human alkaline phosphatases. Mol Biotechnol 56:953–961. https://doi.org/10.1007/s12033-014-9774-9
Schomaker V, Trueblood KN (1968) On the rigid-body motion of molecules in crystals. Acta Crystallogr Sect B Struct Crystallogr Cryst Chem 24:63–76. https://doi.org/10.1107/S0567740868001718
Sheng Y, Li S, Gou X, Kong X, Wang X, Sun Y, Zhang J (2005) The hybrid enzymes from α-aspartyl dipeptidase and l-aspartase. Biochem Biophys Res Commun 331:107–112. https://doi.org/10.1016/J.BBRC.2005.03.140
Srirangan K, Orr V, Akawi L, Westbrook A, Moo-Young M, Chou CP (2013) Biotechnological advances on penicillin G acylase: pharmaceutical implications, unique expression mechanism and production strategies. Biotechnol Adv 31:1319–1332. https://doi.org/10.1016/j.biotechadv.2013.05.006
Stammen S, Müller BK, Korneli C, Biedendieck R, Gamer M, Franco-Lara E, Jahn D (2010) High-yield intra- and extracellular protein production using Bacillus megaterium. Appl Environ Microbiol 76:4037–4046. https://doi.org/10.1128/AEM.00431-10
Tickle IJ, Flensburg C, Keller P, Paciorek W, Sharff A, Vonrhein C, Bricogne G (2018) STARANISO
Tjalsma H, Antelmann H, Jongbloed JDH, Braun PG, Darmon E, Dorenbos R, Dubois F, Westers H, Zanen G, Quax WJ, Kuipers OP, Bron S, Hecker M, Dubois JF, Van DJM (2004) Proteomics of protein secretion by Bacillus subtilis : separating the “secrets” of the secretome. Microbiol Mol Biol Rev 2(68):207–233. https://doi.org/10.1128/MMBR.68.2.207
Valle F, Balbás P, Merino E, Bollvar F (1991) The role of penicillin amidases in nature and in industry. Trends Biochem Sci 16:36–40
Varshney NK, Suresh Kumar R, Ignatova Z, Prabhune A, Pundle A, Dodson E, Suresh CG (2012) Crystallization and X-ray structure analysis of a thermostable penicillin G acylase from Alcaligenes faecalis. Acta Crystallogr Sect F Struct Biol Cryst Commun 68:273–277. https://doi.org/10.1107/S1744309111053930
Voutilainen SP, Nurmi-Rantala S, Penttilä M, Koivula A (2014) Engineering chimeric thermostable GH7 cellobiohydrolases in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 98:2991–3001. https://doi.org/10.1007/s00253-013-5177-2
Wang J, Zhang Q, Huang H, Yuan Z, Ding D, Yang S, Jiang W (2007) Increasing synthetic performance of penicillin G acylase from Bacillus megaterium by site-directed mutagenesis. Appl Microbiol Biotechnol 74:1023–1030. https://doi.org/10.1007/s00253-006-0752-4
Winn MD, Ballard CC, Cowtan KD, Dodson EJ, Emsley P, Evans PR, Keegan RM, Krissinel EB, Leslie AGW, McCoy A, McNicholas SJ, Murshudov GN, Pannu NS, Potterton EA, Powell HR, Read RJ, Vagin A, Wilson KS (2011) Overview of the CCP4 suite and current developments. Acta Crystallogr Sect D Biol Crystallogr 67:235–242. https://doi.org/10.1107/S0907444910045749
Wittchen KD, Meinhardt F (1995) Inactivation of the major extracellular protease from Bacillus megaterium DSM319 by gene replacement. Appl Microbiol Biotechnol 42:871–877. https://doi.org/10.1007/BF00191184
Xu G, Zhao Q, Huang B, Zhou J, Cao F (2018) Directed evolution of a penicillin V acylase from Bacillus sphaericus to improve its catalytic efficiency for 6-APA production. Enzym Microb Technol 119:65–70. https://doi.org/10.1016/j.enzmictec.2018.08.006
Yang S, Huang H, Li S, Ye Y, Wan L, Zhang F, Yuan Z (2000) Enhancing penicillin G acylase stability by site-directed mutagenesis. Sheng Wu Hua Xue Yu Sheng Wu Wu Li Xue Bao (Shanghai) 32(6):581–585
Yang Y, Biedendieck R, Wang W, Gamer M, Malten M, Jahn D, Deckwer W-D (2006) High yield recombinant penicillin G amidase production and export into the growth medium using Bacillus megaterium. Microb Cell Factories 5:36. https://doi.org/10.1186/1475-2859-5-36
Yang Y, Malten M, Grote A, Jahn D, Deckwer W-D (2007) Codon optimized Thermobifida fusca hydrolase secreted by Bacillus megaterium. Biotechnol Bioeng 96:780–794. https://doi.org/10.1002/bit
Zhang Q, Zhang L, Han H, Zhang Y (1986) A method for screening penicillin G acylase-producing bacteria by means of 2-nitro-5-phenylacetaminobenzoic acid test paper. Anal Biochem 156:413–416. https://doi.org/10.1016/0003-2697(86)90274-5
Zheng F, Vermaas JV, Zheng J, Wang Y, Tu T, Wang X, Xie X, Yao B, Beckham GT, Luo H (2018) Characterizing activity and thermostability of GH5 endoglucanase chimeras from mesophilic and thermophilic parents. Appl Environ Microbiol 85. https://doi.org/10.1128/AEM.02079-18
Acknowledgments
We would like to thank Beate Jaschok-Kentner (HZI, Braunschweig, Germany) for N-terminal protein sequencing. The Swiss Light Source (SLS, Paul Scherrer Institute, Villigen, Switzerland) and the PETRAIII synchrotron (DESY, Hamburg, Germany) are acknowledged for providing access to their facilities.
Funding
This work was funded by the German Research Foundation (DFG), within the priority program, SPP1934 “Dispersitäts-, Struktur- und Phasenänderungen von Proteinen und biologischen Agglomeraten in biotechnologischen Prozessen.”
Author information
Authors and Affiliations
Contributions
J.M., W.B., D.J., and R.B. conceived of the study and wrote the paper. J.M., G.G., C.M., A.L., T.K., and R.B. carried out experiments and analyzed the results. J.P. and W.B. conducted crystal x-ray measurements and solved the structure of the three PGAs. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 1995 kb)
Rights and permissions
About this article
Cite this article
Mayer, J., Pippel, J., Günther, G. et al. Crystal structures and protein engineering of three different penicillin G acylases from Gram-positive bacteria with different thermostability. Appl Microbiol Biotechnol 103, 7537–7552 (2019). https://doi.org/10.1007/s00253-019-09977-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-09977-8