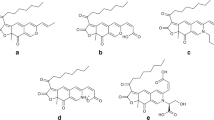
Abstract
The replacement of synthetic colors in food products by natural alternatives has been boosted by consumers willing to pay more for healthier products. However, the success of microbial colorants depends not only on its acceptability on the market but also its production costs. Talaromyces species can produce water-soluble red colorants induced by glucose and monosodium glutamate (MSG). In this study, the influence of several conditions was evaluated to produce natural red colorants by submerged culture of Talaromyces amestolkiae. Under optimal conditions (g/L: glucose 10, MSG 25, MgSO4 0.012, FeSO4 0.01, CaCl2 0.015; and initial pH of 5.0), a 30-fold increase in the production was achieved, reaching a red colorant production of 13.44 UA500nm. Depending on the initial pH, colorants with different hues and chroma values were obtained. Deep yellow colorants were derived from neutral and basic pH, while deep red colors were derived from acidic pH. The fluorescence spectrum of culture broth obtained before and after complexation with salts presented red colorants with yellow fluorescence spectra. The information generated in this study would be useful for the formulation of industrial media for large-scale cultivation of T. amestolkiae, which have the potential to produce Talaromyces fermented colorants for use in health foods and pharmaceutics.





Similar content being viewed by others
References
Arai T, Koganei K, Umemura S, Kojima R, Kato J, Kasumi T, Ogihara J (2013) Importance of the ammonia assimilation by Penicillium purpurogenum in amino derivative Monascus pigment, PP-V, production. AMB Express 3:19
Bibb MJ (2005) Regulation of secondary metabolism in streptomycetes. Curr Opin Microbiol 8(2):208–215. https://doi.org/10.1016/j.mib.2005.02.016
Blanch HW, Bhavaraju SM (1976) Non-Newtonian fermentation broths: rheology and mass transfer. Biotechnol Bioeng 18(6):745–790. https://doi.org/10.1002/bit.260180602
Carels M, Shepherd D (1977) The effect of different nitrogen sources on pigment production and sporulation of Monascus species in submerged, shaken culture. Can J Microbiol 23(10):1360–1372. https://doi.org/10.1139/m77-205
Chen M-H, Johns MR (1993) Effect of pH and nitrogen source on pigment production by Monascus purpureus. Appl Microbiol Biotechnol 40(1):132–138
Chen W, Chen R, Liu Q, He Y, He K, Ding X, Kang L, Guo X, Xie N, Zhou Y, Lu Y, Cox RJ, Molnár I, Li M, Shao Y, Chen F (2017) Orange, red, yellow: biosynthesis of azaphilone pigments in Monascus fungi. Chem Sci 8:4917–4925. https://doi.org/10.1039/c7sc00475c
Dufossé L (2017) Pigments, microbial. In: Reference module in life sciences. Elsevier, Oxford, pp 1–16. https://doi.org/10.1016/B978-0-12-809633-8.13091-2
Dufossé L (2018) Red colourants from filamentous fungi: are they ready for the food industry? J Food Compos Anal 69:156–161. https://doi.org/10.1016/j.jfca.2017.11.002
Evans PJ, Wang HY (1984) Pigment production from immobilized Monascus spp utilizing polymeric resin adsorption. App Environ Microbiol 47(6):1323–1326
Galaffu N, Bortlik K, Michel M (2015) An industry perspective on natural food colour stability. In: Colour Additives for Foods and Beverages, Woodhead publishing, pp 91–130. https://doi.org/10.1016/C2013-0-16427-6
Gao JM, Yang SX, Qin JC (2013) Azaphilonoids: chemistry and biology. Chem Rev 113:4755–4811
Hajjaj H, Klaebe A, Loret MO, Tzedakis T, Goma G, Blanc PJ (1997) Production and identification of N-glucosylrubropunctamine and Nglucosylmonascorubramine from Monascus ruber and the occurrence of electron donor-acceptor complexes in these red pigments. Appl Environ Microbiol 63:2671–2678
Hajjaj H, Klaébé A, Goma G, Blanc PJ, Barbier E, François J (2000) Medium-chain fatty acids affect Citrinin production in the filamentous fungus Monascus ruber. Appl Environ Microbiol 66(3):1120–1125
Hsu YW, Hsu LC, Liang YH, Kuo YH, Pan TM (2011) New bioactive orange pigments with yellow fluorescence from Monascus-fermented dioscorea. J Agric Food Chem 59:4512–4518
Huang Z, Zhang S, Xu Y, Li L, Li Y (2014) Structural characterization of two new orange pigments with strong yellow fluorescence. Phytochem Lett 10:140–144. https://doi.org/10.1016/j.phytol.2014.08.020
Huang T, Tan H, Chen G, Wang L, Wu Z (2017) Rising temperature stimulates the biosynthesis of water-soluble fluorescent yellow pigments and gene expression in Monascus ruber CGMCC10910. AMB Express 7(134):1–10. https://doi.org/10.1186/s13568-017-0441-y
Ismael R, Schwander H, Hendrix P (2013) Fluorescent dyes and pigments. In: Ullmann’s encyclopedia of industrial chemistry. Wiley Online Library, pp 1–22
Jůzlová P, Martínková L, Křen V (1996) Secondary metabolites of the fungus Monascus: a review. J Ind Microbiol 16(3):163–170
Kang B, Zhang X, Wu Z, Wang Z, Park S (2014) Production of citrinin-free Monascus pigments by submerged culture at low pH. Enzym Microbial Technol 55:50–57. https://doi.org/10.1016/j.enzmictec.2013.12.007
Kim C, Jung H, Kim JH, Shin CS (2006) Effect of Monascus pigment derivatives on the electrophoretic mobility of bacteria, and the cell adsorbtion and antibacterial activities of pigments. Colloid Surf B 47:153–159. https://doi.org/10.1016/j.colsurfb.2005.12.009
Krairak S, Yamamura K, Irie R, Nakajima M, Shimizu H, Chim-Anage P, Yongsmith B, Shioya S (2000) Maximizing yellow pigment production in fed-batch culture of Monascus sp. J Biosci Bioeng 90(4):363–367. https://doi.org/10.1016/S1389-1723(01)80002-5
Lakowicz JR (2006) Chapter sixteen – protein fluorescence. In: Principles of fluorescence spectroscopy, 3rd edn. Springer, New York, pp 529–569
Lee B-K, Park N-H, Piao HY, Chung W-J (2001) Production of red pigments by Monascus purpureus in submerged culture. Biotechnol Bioprocess Eng 6(5):341–346
Lehto S, Buchweitz M, Klimm A, Straßburger R, Bechtold C, Ulberth F (2017) Comparison of food colour regulations in the EU and the US: a review of current provisions. Food Addit Contam A 34(3):335–355. https://doi.org/10.1080/19440049.2016.1274431
Lin TF, Demain AL (1991) Effect of nutrition of Monascus sp. on formation of red pigments. Appl Microbiol Biotechnol 36(1):70–75
Lin TF, Demain AL (1994) Leucine interference in the production of water-soluble red Monascus pigments. Arch Microbiol 162(1–2):114–119
Loret MO, Morel S (2010) Isolation and structural characterization of two new metabolites from Monascus. J Agric Food Chem 58:1800–1803
Lv J, Zhang B-B, Liu X-D, Zhang C, Chen L, Xu G-R, Cheung PCK (2017) Enhanced production of natural yellow pigments from Monascus purpureus by liquid culture: the relationship between fermentation conditions and mycelial morphology. J Biosci Bioeng 124(4):452–458. https://doi.org/10.1016/j.jbiosc.2017.05.010
Mapari SAS, Meyer AS, Thrane U (2006) Colorimetric characterization for comparative analysis of fungal pigments and natural food colorants. J Agric Food Chem 54(19):7027–7035. https://doi.org/10.1021/jf062094n
Mapari SAS, Thrane U, Meyer AS (2010) Fungal polyketide azaphilone pigments as future natural food colorants? Trends Biotechnol 28:300–307. https://doi.org/10.1016/j.tibtech.2010.03.004
Orozco SFB, Kilikian BV (2008) Effect of pH on citrinin and red pigments production by Monascus purpureus CCT3802. World J Microb Biotechnol 24(2):263–268. https://doi.org/10.1007/s11274-007-9465-9
Plackett RL, Burman JP (1946) The Design of Optimum Multifactorial Experiments. Biometrika 33(4):305–325
Sá M, Monte J, Brazinha C, Galinha CF, Crespo JG (2017) 2D fluorescence spectroscopy for monitoring Dunaliella salina concentration and integrity during membrane harvesting. Algal Res 24:325–332. https://doi.org/10.1016/j.algal.2017.04.013
Santos-Ebinuma VC, Teixeira MFS, Pessoa A Jr (2013) Submerged culture conditions for the production of alternative natural colorants by a newly isolated Penicillium purpurogenum DPUA 1275. J Microbiol Biotechnol 23:802–810. https://doi.org/10.4014/jmb.1211.11057
Shi K, Song D, Chen G, Pistolozzi M, Wu Z, Quan L (2015) Controlling composition and color characteristics of Monascus pigments by pH and nitrogen sources in submerged fermentation. J Biosci Bioeng 120(2):145–154. https://doi.org/10.1016/j.jbiosc.2015.01.001
Simonot L, Thoury M, Delaney J (2011) Extension of the Kubelka–Munk theory for fluorescent turbid media to a nonopaque layer on a background. J Opt Soc Am A 28(7):1349–1357. https://doi.org/10.1364/JOSAA.28.001349
Singh S (2006) Impact of color on marketing. Manag Decis 44(6):783–789. https://doi.org/10.1108/00251740610673332
Stowe RA, Mayer RP (1966) Efficient screening of process variables. Ind Eng Chem 58(2):36–40
Terán-Hilares R, de Souza RA, Marcelino PF, da Silva SS, Dragone G, Mussatto SI, Santos JC (2018) Sugarcane bagasse hydrolysate as a potential feedstock for red pigment production by Monascus ruber. Food Chem 245:786–791. https://doi.org/10.1016/j.foodchem.2017.11.111
Torres FAE, Zaccarim BR, Novaes LCL, Jozala AF, Santos CA, Teixeira MFS, Santos-Ebinuma VC (2016) Natural colorants from filamentous fungi. Appl Microbiol Biotechnol 100(6):2511–2521. https://doi.org/10.1007/s00253-015-7274-x
Vendruscolo F, Schmidell W, Moritz DE, Bühler RMM, de Oliveira D, Ninow JL (2016) Isoelectric point of amino acid: importance for Monascus pigment production. Biocatal Agric Biotechnol 5:179–185. https://doi.org/10.1016/j.bcab.2015.12.006
Venkatachalam M, Zelena M, Cacciola F, Ceslova L, Girard-Valenciennes E, Clerc P, Dugo P, Mondello L, Fouillaud M, Rotondo A, Giuffrida D, Dufossé L (2018) Partial characterization of the pigments produced by the marine-derived fungus Talaromyces albobiverticillius 30548. Towards a new fungal red colorant for the food industry. J Food Compos Anal 67:38–47. https://doi.org/10.1016/j.jfca.2017.12.036
Ventura SPM, Santos-Ebinuma VC, Pereira JFB, Teixeira MFS, Pessoa A Jr, Coutinho JAP (2013) Isolation of natural red colorants from fermented broth using ionic liquid-based aqueous two-phase systems. J Ind Microbiol Biotechnol 40:507–516. https://doi.org/10.1007/s10295-013-1237-y
Vinha AF, Rodrigues F, Nunes MA, Oliveira MBPP (2018) Natural pigments and colorants in foods and beverages. In: Polyphenols: properties, recovery, and applications. Woodhead Publishing, pp 363–391
Wei WG, Yao ZJ (2005) Synthesis studies toward chloroazaphilone and vinylogous γ -pyridones: two common natural product core structures. J Org Chem 70:4585–4590
Weinberg ED (1989) Chapter seven - roles of micronutrients in secondary metabolism of Actinomycetes. In: Shapiro S (ed) Regulation of Secondary Metabolism in Actinomycetes, 3rd edn. CRC Press, Boca Raton, pp 239–261
Yilmaz N, Houbraken J, Hoekstra ES, Frisvad JC, Visagie CM, Samson RA (2012) Delimitation and characterisation of Talaromyces purpurogenus and related species. Persoonia 29(1):39–54
Zaccarim BR, de Oliveira F, Passarini MRZ, Duarte AWF, Sette LD, Jozala AF, Santos Ebinuma VC (2018) Sequencing and phylogenetic analyses of Talaromyces amestolkiae from the Amazon, a producer of natural colorants. Biotechnol Prog. https://doi.org/10.1002/btpr.2684
Zollinger H (2003) Color of organic compounds. In: Color chemistry: syntheses, properties, and applications of organic dyes and pigments. Wiley-VHCA and Wiley-VCH, Zurich and Weinheim, p 51
Funding
This study was supported by the São Paulo Research Foundation (FAPESP) – Brazil [Grant no. 2014/01580-3], by the Conselho Nacional de Desenvolvimento Científico CNPq-Brazil [Grant no. 155317/2016-4], and partly by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 585 kb)
Rights and permissions
About this article
Cite this article
de Oliveira, F., Pedrolli, D.B., Teixeira, M.F.S. et al. Water-soluble fluorescent red colorant production by Talaromyces amestolkiae. Appl Microbiol Biotechnol 103, 6529–6541 (2019). https://doi.org/10.1007/s00253-019-09972-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-09972-z